Scalable Synthesis of Cholic Acid Intermediate A2 for Virus-Free API Manufacturing
Scalable Synthesis of Cholic Acid Intermediate A2 for Virus-Free API Manufacturing
The pharmaceutical industry is currently witnessing a paradigm shift in the sourcing of critical bile acid intermediates, driven by stringent safety regulations and supply chain volatility. Patent CN115611961A introduces a groundbreaking chemical synthesis method for producing Cholic Acid Intermediate A2, a pivotal precursor in the manufacturing of Cholic Acid (CA). Unlike traditional methods that rely on the extraction of bile acids from animal viscera, which poses significant risks of viral contamination and batch-to-batch inconsistency, this novel approach utilizes a fully synthetic route starting from common plant-source compounds. By leveraging a mild acid-catalyzed elimination reaction, the technology enables the production of high-purity intermediates under controlled conditions, effectively mitigating the biological safety hazards associated with animal-derived raw materials while offering a robust pathway for commercial scale-up.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the global supply of Cholic Acid has been heavily dependent on the extraction and refinement from the internal organs of cattle or sheep. This biological sourcing model presents inherent and unavoidable limitations that threaten both product safety and supply continuity. The primary concern is the risk of virus infection and prion transmission, such as Bovine Spongiform Encephalopathy (BSE), which necessitates complex and costly viral clearance validation processes for any resulting Active Pharmaceutical Ingredient (API). Furthermore, the availability of raw animal organs is subject to seasonal fluctuations, regulatory restrictions on animal slaughter, and geopolitical trade barriers, leading to unpredictable pricing and potential supply shortages. The purification of natural extracts is also chemically challenging due to the presence of structurally similar bile acids like chenodeoxycholic acid and deoxycholic acid, often requiring extensive chromatography that limits overall yield and increases manufacturing costs significantly.
The Novel Approach
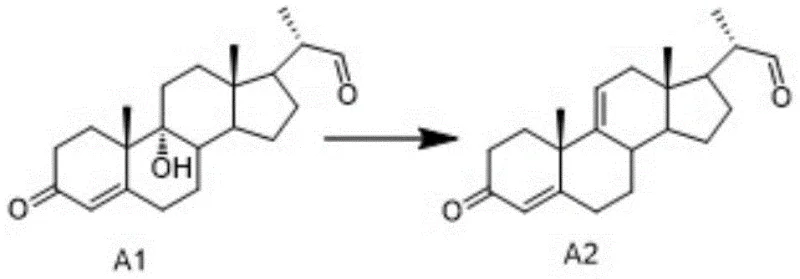
In stark contrast, the methodology disclosed in CN115611961A offers a chemically defined, virus-free alternative that transforms the production landscape for bile acid derivatives. The core innovation lies in the efficient conversion of Compound A1 to Intermediate A2 through a controlled elimination reaction, which serves as the gateway to the final Cholic Acid structure. This synthetic strategy bypasses the need for animal tissues entirely, utilizing 9α-hydroxy BA (Compound SM) as a starting material, which can be sourced from stable plant-based supplies or synthesized via established organic pathways. The process is characterized by mild reaction conditions, typically operating between 20-50°C, which preserves the integrity of the sensitive steroid skeleton while ensuring high conversion rates. By establishing a fully synthetic supply chain, manufacturers can achieve superior control over impurity profiles, eliminate biological contaminants, and secure a more reliable long-term supply of this critical medical intermediate.
Mechanistic Insights into Acid-Catalyzed Elimination Reaction
The heart of this patented technology is the precise execution of the elimination reaction that converts Compound A1 into the key Intermediate A2. This transformation involves the dehydration of the steroid backbone under acidic conditions, a step that requires meticulous control to prevent side reactions such as double bond migration or skeletal rearrangement. The patent details two distinct solvent systems to achieve this: one utilizing water as a solvent with concentrated sulfuric acid and glacial acetic acid, and another employing dichloromethane or trichloromethane with acetic anhydride and sulfuric acid. In the aqueous system, the reaction proceeds at temperatures between 20-50°C, where the sulfuric acid acts as a potent proton donor to facilitate the departure of the hydroxyl group, while the acetic acid modulates the acidity to prevent over-reaction. This balance is critical for maintaining the stereochemical integrity of the molecule, ensuring that the resulting double bond is formed at the desired position without compromising the adjacent chiral centers essential for biological activity.
Impurity control is further enhanced by the specific post-treatment protocols outlined in the patent, which are designed to remove residual acids and byproducts efficiently. Following the reaction, the mixture is quenched in an ice bath, and the precipitate is washed to neutrality before being dissolved in dichloromethane for phase separation. This workup procedure is vital for removing trace sulfuric acid and acetic acid residues that could catalyze degradation during subsequent storage or processing steps. Additionally, the use of methanol replacement during concentration helps to strip away high-boiling impurities and ensures the final product is obtained in a viscous state suitable for direct use in the next synthetic step. The alternative organic solvent system using acetic anhydride offers similar benefits, providing a homogeneous reaction environment that can improve mass transfer and reaction kinetics, thereby reducing the overall reaction time to between 3-5 hours while maintaining high selectivity for the target Intermediate A2.
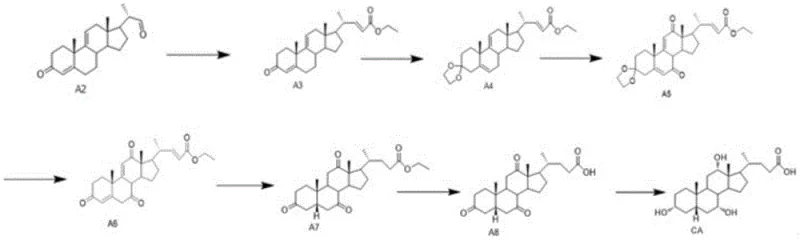
How to Synthesize Cholic Acid Intermediate A2 Efficiently
The synthesis of Cholic Acid Intermediate A2 is a multi-stage process that demands rigorous adherence to reaction parameters to ensure optimal yield and purity. The protocol begins with the oxidation of the starting material SM to generate Compound A1, followed by the critical elimination step to produce A2, and concludes with a series of downstream transformations including Wittig olefination, ketal protection, and selective reductions to arrive at the final Cholic Acid. Each step in this sequence has been optimized to minimize waste and maximize throughput, making it an attractive candidate for industrial adoption. The detailed standardized synthesis steps, including specific reagent ratios, temperature profiles, and workup procedures, are provided in the technical guide below to assist process chemists in replicating this high-efficiency route.
- Oxidize 9α-hydroxy BA (SM) to Intermediate A1 using TEMPO/NaClO or Jones reagent.
- Perform acid-catalyzed elimination on A1 using sulfuric acid and acetic acid/anhydride to yield Intermediate A2.
- Convert A2 to Cholic Acid through Wittig reaction, ketal protection, oxidation, hydrolysis, hydrogenation, and selective reduction.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from animal extraction to the synthetic route described in CN115611961A represents a strategic opportunity to de-risk the supply of critical bile acid intermediates. The traditional reliance on animal organs introduces variables that are difficult to control, such as disease outbreaks in livestock and changing agricultural regulations, which can cause sudden spikes in raw material costs or complete supply interruptions. By adopting this synthetic methodology, organizations can decouple their production from biological volatility, securing a more predictable and stable supply chain. Furthermore, the elimination of viral risks simplifies the regulatory compliance landscape, potentially reducing the time and cost associated with viral clearance testing and validation, which are mandatory for animal-derived APIs. This shift not only enhances supply security but also aligns with growing consumer and regulatory preferences for synthetic, cruelty-free, and contaminant-free pharmaceutical ingredients.
- Cost Reduction in Manufacturing: The synthetic route offers significant potential for cost optimization by streamlining the purification process. Unlike natural extracts which contain a complex mixture of structurally similar bile acids requiring expensive chromatographic separation, the synthetic intermediate A2 is generated with high specificity, reducing the burden on downstream purification units. Additionally, the reaction conditions are mild and utilize common industrial reagents like sulfuric acid and acetic acid, avoiding the need for exotic catalysts or extreme temperature controls that drive up utility costs. The ability to use standard stainless steel reactors without specialized linings for corrosive biological fluids further contributes to lower capital expenditure and operational overhead, resulting in a more economically viable manufacturing process overall.
- Enhanced Supply Chain Reliability: Sourcing raw materials from plant-derived or synthetic precursors provides a level of supply chain resilience that animal extraction cannot match. The starting material, 9α-hydroxy BA, can be produced consistently in large quantities independent of seasonal slaughtering cycles or geographic limitations on livestock. This stability allows for better long-term planning and inventory management, reducing the need for safety stock buffers that tie up working capital. Moreover, a synthetic supply chain is less susceptible to trade restrictions on animal products, facilitating smoother international logistics and ensuring continuous delivery to global manufacturing sites even during regional agricultural disruptions.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing unit operations such as liquid-liquid extraction, crystallization, and filtration that are well-understood and easily expanded from pilot to commercial scale. The mild reaction temperatures (20-50°C) reduce energy consumption compared to high-temperature processes, contributing to a lower carbon footprint. Furthermore, the synthetic route generates a more defined waste stream compared to biological processing, simplifying wastewater treatment and disposal. The absence of animal byproducts eliminates the need for specialized biohazard waste handling, streamlining environmental compliance and reducing the associated disposal costs, making the process more sustainable and environmentally friendly.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of Cholic Acid Intermediate A2. These answers are derived directly from the technical specifications and beneficial effects outlined in patent CN115611961A, providing clarity on the feasibility and advantages of this synthetic approach for potential adopters.
Q: Why is synthetic Cholic Acid preferred over animal-extracted sources?
A: Animal extraction carries inherent risks of virus and prion contamination (e.g., BSE). Synthetic routes from plant-derived precursors like 9α-hydroxy BA eliminate these biological safety hazards and ensure consistent purity.
Q: What are the critical reaction conditions for synthesizing Intermediate A2?
A: The patent specifies mild elimination conditions between 20-50°C using concentrated sulfuric acid with either glacial acetic acid/water or acetic anhydride/dichloromethane, ensuring high selectivity without degrading the steroid backbone.
Q: Is this synthetic route scalable for commercial API production?
A: Yes, the process utilizes standard organic synthesis unit operations (oxidation, elimination, hydrogenation) and avoids extreme cryogenic or high-pressure conditions, making it highly suitable for scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cholic Acid Intermediate A2 Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing high-quality, virus-free intermediates for the pharmaceutical industry. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex synthetic routes like the one for Cholic Acid Intermediate A2 can be successfully transferred to large-scale manufacturing. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch meets the highest standards required for API synthesis. We are committed to supporting our partners in navigating the transition from traditional extraction to advanced chemical synthesis, providing the technical expertise and capacity needed to bring safer, more reliable medicines to market.
We invite you to contact our technical procurement team to discuss how we can support your specific project requirements. Whether you need a Customized Cost-Saving Analysis for switching to synthetic intermediates or require specific COA data and route feasibility assessments for your pipeline, our experts are ready to assist. Let us partner with you to optimize your supply chain and ensure the uninterrupted production of high-value pharmaceutical products.
