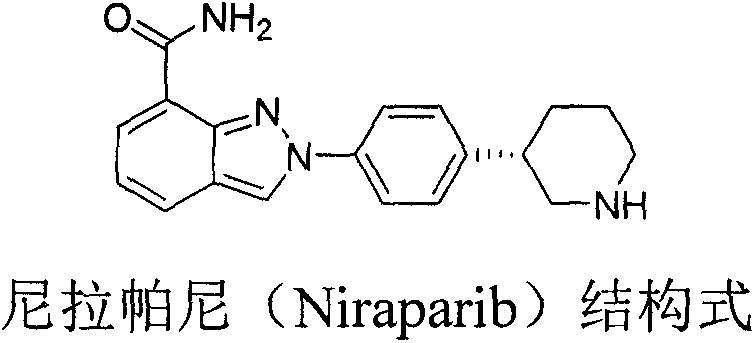
Advanced Chemical Synthesis of Niraparib Chiral Intermediates for Commercial Scale Production
The global demand for Poly (ADP-ribose) polymerase (PARP) inhibitors has surged following the clinical success of drugs like Niraparib in treating ovarian and breast cancers. As the market expands, the pressure on the supply chain to deliver high-purity active pharmaceutical ingredients (APIs) and their precursors has never been greater. Patent CN107311911B introduces a transformative chemical methodology for producing the critical chiral piperidine intermediate of Niraparib, addressing long-standing bottlenecks in cost and scalability. This technical insight report analyzes the proprietary synthesis route disclosed in the patent, highlighting its potential to redefine the manufacturing landscape for this high-value oncology drug. By shifting away from biological catalysis and chiral resolution towards a robust chemical asymmetric synthesis, this innovation offers a compelling value proposition for pharmaceutical manufacturers seeking reliability and efficiency.
In the context of modern pharmaceutical manufacturing, the transition from discovery-scale chemistry to commercial production often reveals hidden inefficiencies in legacy synthetic routes. Historically, the preparation of Niraparib intermediates has relied on methodologies that, while effective on a small scale, present significant hurdles for industrial application. One prominent prior art route, described in WO2009087381, utilizes a Suzuki coupling reaction followed by hydrogenation and, critically, chiral column separation to isolate the desired enantiomer. This reliance on chiral chromatography is a major economic and logistical burden, as it typically suffers from low throughput, high solvent consumption, and substantial product loss, making it ill-suited for multi-ton production campaigns.
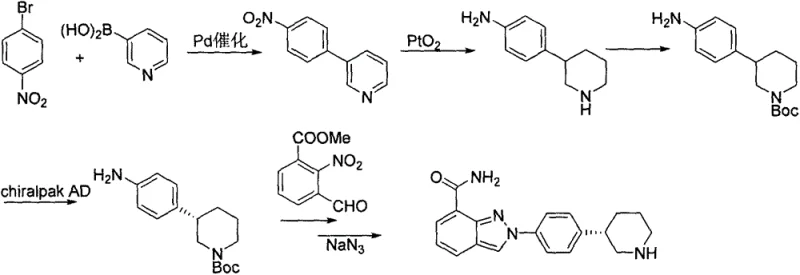
Furthermore, alternative approaches such as those detailed in WO2014088983 have attempted to utilize enzymatic catalysis to establish chirality. While biocatalysis is often touted for its specificity, in this specific application, it introduces dependencies on expensive enzyme preparations and requires stringent control over reaction parameters that can complicate process validation. The cumulative effect of these legacy methods is a supply chain vulnerable to cost volatility and capacity constraints. The industry urgently requires a synthetic strategy that bypasses these limitations by utilizing stable, commodity-grade starting materials and standard unit operations that can be easily scaled in existing multipurpose chemical plants without specialized infrastructure.
The novel approach disclosed in patent CN107311911B fundamentally reimagines the construction of the chiral piperidine core by employing a chiral auxiliary strategy reminiscent of Evans aldol chemistry, adapted here for a Michael addition framework. Instead of resolving a racemic mixture or relying on fragile biological catalysts, this route installs chirality at the outset using a chiral oxazolidinone auxiliary coupled with 4-bromophenylacetic acid. This strategic decision allows for the precise control of stereochemistry during the carbon-carbon bond-forming step, effectively rendering the subsequent chiral separation steps obsolete. The result is a linear, convergent synthesis that maximizes atom economy and minimizes waste generation, directly addressing the economic pain points identified in previous manufacturing attempts.
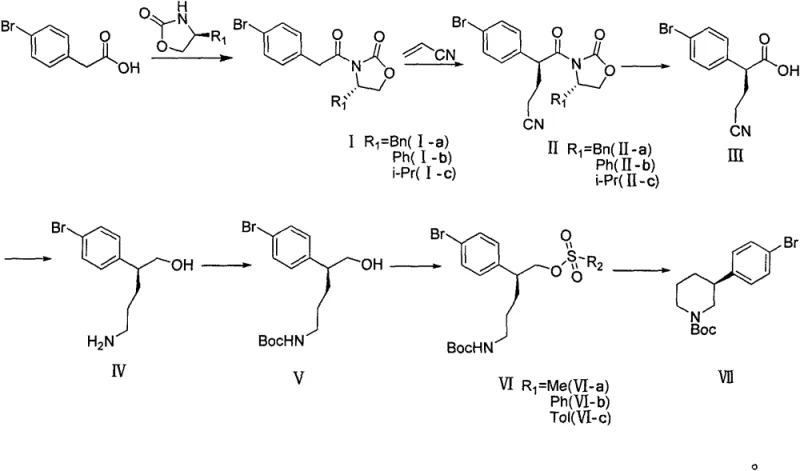
At the heart of this mechanistic breakthrough is the titanium-catalyzed Michael addition of acrylonitrile to the chiral imide intermediate. The chiral oxazolidinone moiety acts as a temporary stereochemical director, shielding one face of the enolate equivalent and forcing the incoming nucleophile to attack from the less hindered trajectory. This steric differentiation is critical for achieving the high diastereomeric excess required for pharmaceutical grade intermediates. Following the establishment of the stereocenter, the auxiliary is cleaved via hydrolysis under mild oxidative conditions, releasing the chiral acid without racemization. This sequence demonstrates a sophisticated understanding of physical organic chemistry, leveraging well-understood principles of asymmetric induction to solve a complex manufacturing problem with elegant simplicity.
From an impurity control perspective, this route offers distinct advantages over transition-metal heavy cross-coupling strategies. By avoiding palladium-catalyzed couplings in the late stages of the synthesis, the risk of heavy metal contamination in the final API is significantly mitigated, simplifying the purification train and reducing the load on quality control laboratories. Furthermore, the intermediates generated throughout this seven-step sequence, including the protected amino alcohol and the mesylate precursor, are chemically stable and isolable. This stability provides process engineers with valuable flexibility, allowing for the stocking of key intermediates to buffer against supply chain disruptions and enabling a modular approach to production scheduling that enhances overall operational resilience.
How to Synthesize Niraparib Chiral Intermediate Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for executing this transformation in a pilot or production setting. The process begins with the activation of 4-bromophenylacetic acid and its subsequent coupling with the chiral auxiliary, followed by the critical stereoselective alkylation. Each step has been optimized for yield and purity, utilizing common organic solvents such as dichloromethane, tetrahydrofuran, and toluene, which facilitates solvent recovery and recycling. The detailed standardized synthesis steps below outline the specific reagents and conditions required to replicate this high-efficiency pathway.
- Condense 4-bromophenylacetic acid with a chiral oxazolidinone auxiliary using pivaloyl chloride and a base to form the amide intermediate.
- Perform a titanium-catalyzed Michael addition with acrylonitrile to establish the stereocenter with high diastereoselectivity.
- Execute hydrolysis to remove the auxiliary, followed by reduction, Boc protection, mesylation, and final intramolecular cyclization to close the piperidine ring.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route represents a strategic opportunity to optimize the cost structure and reliability of the Niraparib supply chain. By eliminating the dependency on specialized chiral columns and expensive enzymatic reagents, the manufacturing process becomes significantly more robust and predictable. The shift towards commodity chemicals means that raw material sourcing is less susceptible to the volatility often seen with specialized fine chemical reagents, thereby stabilizing the cost of goods sold (COGS) over the long term. This stability is crucial for long-term supply agreements and budget forecasting in the highly competitive oncology market.
- Cost Reduction in Manufacturing: The most immediate financial impact of this technology is the removal of costly purification steps. Traditional routes relying on chiral HPLC consume vast amounts of solvent and stationary phase, driving up operational expenditures. By achieving chirality through chemical synthesis rather than physical separation, this method drastically reduces solvent usage and waste disposal costs. Additionally, the use of inexpensive starting materials like 4-bromophenylacetic acid and acrylonitrile ensures that the raw material bill remains low, allowing for substantial margin improvement or competitive pricing strategies for the final API.
- Enhanced Supply Chain Reliability: Supply continuity is paramount for life-saving medications. This synthetic route relies on reagents that are widely available from multiple global suppliers, reducing the risk of single-source bottlenecks. Unlike enzymatic processes that may require custom fermentation runs or specific storage conditions, the chemical reagents used here are shelf-stable and easy to transport. This inherent robustness translates to shorter lead times and a more agile response to fluctuations in market demand, ensuring that patients receive their medication without interruption.
- Scalability and Environmental Compliance: Scaling a chemical process often introduces new challenges, but this route is designed with industrial practicality in mind. The reactions proceed under mild temperatures and pressures, minimizing energy consumption and safety risks associated with high-pressure hydrogenation or cryogenic conditions. Furthermore, the reduction in solvent intensity and the avoidance of heavy metal catalysts align with modern green chemistry principles, simplifying environmental permitting and waste treatment compliance. This makes the technology not only economically attractive but also environmentally sustainable for large-scale manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis technology. These answers are derived directly from the technical specifications and beneficial effects described in the patent documentation, providing clarity for stakeholders evaluating the feasibility of this manufacturing route. Understanding these details is essential for making informed decisions about technology transfer and process validation.
Q: How does this new synthesis route improve upon previous enzymatic methods?
A: Unlike enzymatic routes which require expensive biocatalysts and complex operational conditions, this chemical route utilizes robust, commercially available reagents like 4-bromophenylacetic acid and chiral oxazolidinones, significantly lowering production costs and simplifying scale-up.
Q: What is the primary advantage regarding chirality control in this process?
A: The process employs a chiral auxiliary strategy (Evans auxiliary logic) during the Michael addition step, which induces high stereoselectivity without the need for costly and low-yield chiral column chromatography separation used in earlier generations of synthesis.
Q: Is this manufacturing process suitable for large-scale industrial production?
A: Yes, the patent explicitly states the method is suitable for industrial production due to the use of standard organic solvents, mild reaction conditions, and the avoidance of specialized equipment like chiral HPLC systems, ensuring high yield and operational stability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Niraparib Intermediate Supplier
As the pharmaceutical industry continues to evolve, the ability to adapt and implement advanced synthetic methodologies is what separates leading CDMOs from the rest. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, possessing the technical expertise to translate complex patent literature into viable commercial processes. We understand that the synthesis of chiral intermediates requires precision and rigorous quality control. Our facilities are equipped to handle diverse synthetic pathways, ranging from small-scale optimization to full commercial production. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and reliability. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch against the highest industry standards.
We invite you to explore how our technical capabilities can enhance your supply chain for Niraparib and other critical oncology intermediates. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a supply chain partner dedicated to innovation, efficiency, and the uninterrupted delivery of life-saving medicines.
