Advanced Enzymatic Synthesis of Darunavir Key Intermediates for Commercial Scale Production
Advanced Enzymatic Synthesis of Darunavir Key Intermediates for Commercial Scale Production
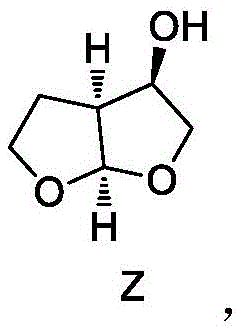
The pharmaceutical industry continuously seeks robust, scalable, and economically viable pathways for the synthesis of complex antiretroviral agents, particularly for the production of Darunavir, a potent HIV-1 protease inhibitor. A pivotal breakthrough in this domain is documented in Chinese Patent CN110372641B, which discloses a highly efficient preparation method for hexahydrofurofuranol derivatives, specifically the key intermediate (3R, 3aS, 6aR)-hexahydrofuro[2,3-b]furan-3-ol. This patent represents a significant technological leap by transitioning from traditional chemical chirality construction to a sophisticated enzyme-mediated reduction process. The innovation lies in the utilization of a specific aldehyde ketone reductase derived from Saccharomyces kudriavzevii, which enables the construction of multiple chiral centers with exceptional stereocontrol. For global procurement leaders and R&D directors, this technology offers a compelling alternative to legacy methods, promising not only superior product quality but also a streamlined manufacturing workflow that aligns with modern green chemistry principles and cost-reduction mandates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the darunavir core structure has relied heavily on chemical methodologies that often involve cumbersome steps and expensive reagents. For instance, prior art from manufacturers like Sumitomo Chemical utilized chiral ligand catalysts to establish the necessary stereochemistry, a approach that, while effective, introduces significant cost burdens due to the high price of ligands and the complexity of removing metal residues from the final API. Similarly, other routes described in European patents involved carbonyl reductase polypeptides but often lacked the optimized reaction conditions or the specific enzyme variants required to achieve industrial-grade efficiency. These conventional pathways frequently suffer from moderate diastereoselectivity, necessitating rigorous and yield-loss-inducing purification steps such as repeated crystallizations or chromatography. Furthermore, the use of stoichiometric amounts of hazardous reducing agents like borohydrides or aluminum hydrides poses safety risks and generates substantial chemical waste, creating environmental compliance challenges that modern supply chains are eager to avoid.
The Novel Approach
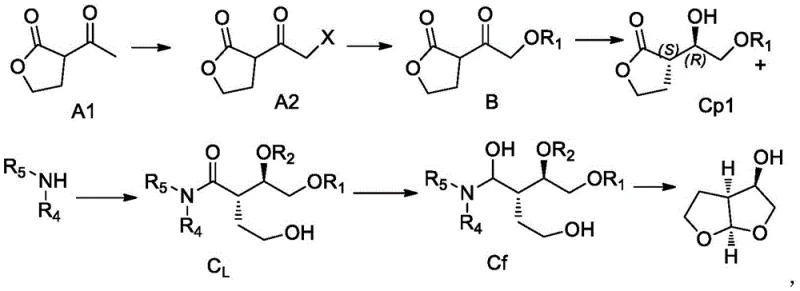
In stark contrast, the methodology outlined in CN110372641B introduces a paradigm shift by employing a genetically engineered aldehyde ketone reductase to drive the critical reduction step. This biological catalyst is not merely a substitute but a superior tool that operates under mild physiological conditions, typically between 25°C and 37°C, thereby reducing energy consumption and thermal stress on sensitive intermediates. The process begins with a halogenation and acylation sequence to prepare the ketone substrate, followed by the enzymatic reduction which constructs the chiral configuration with remarkable precision. By integrating a glucose dehydrogenase system for cofactor regeneration, the process ensures that the expensive NADP+ cofactor is recycled in situ, minimizing reagent costs. This novel approach effectively bypasses the need for external chiral auxiliaries, simplifying the synthetic tree and enhancing the overall atom economy. The result is a pathway that is not only chemically elegant but also commercially attractive for reliable pharmaceutical intermediate supplier networks seeking to optimize their portfolio.
Mechanistic Insights into Aldehyde Ketone Reductase Catalyzed Stereoselective Reduction
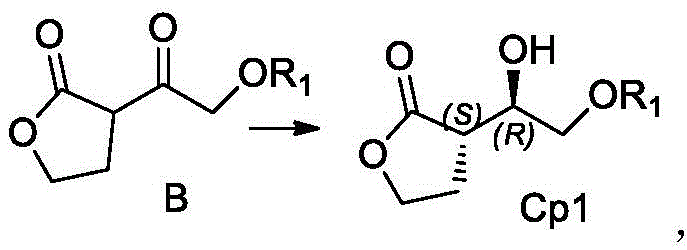
The core of this technological advancement resides in the specific catalytic mechanism of the aldehyde ketone reductase encoded by SEQ ID NO:1. This enzyme, derived from Saccharomyces kudriavzevii, exhibits a unique active site geometry that preferentially binds the prochiral ketone substrate in a conformation that favors the formation of the (3R, 3aS, 6aR) stereoisomer. During the reaction, the enzyme facilitates the hydride transfer from the reduced nicotinamide adenine dinucleotide phosphate (NADPH) to the carbonyl carbon of the substrate with extreme facial selectivity. The patent data indicates that this biocatalytic step achieves a conversion rate of up to 97.8%, with a diastereomeric excess (de) value exceeding 96% and an enantiomeric excess (ee) value greater than 99%. Such high selectivity is critical for R&D teams focused on impurity control, as it drastically reduces the formation of unwanted stereoisomers that could complicate downstream processing or compromise the safety profile of the final drug product. The enzyme's stability in the presence of organic co-solvents like DMSO or butyl acetate further enhances its utility, allowing for higher substrate loading and improved reaction kinetics without denaturation.
Furthermore, the integration of a cofactor regeneration system is a masterstroke of process engineering that directly impacts the economic feasibility of the route. In traditional stoichiometric reductions, the cost of reducing equivalents can be prohibitive at scale. However, by coupling the primary reductase with glucose dehydrogenase, the system continuously oxidizes glucose to gluconolactone while regenerating NADPH from NADP+. This cyclic process means that only catalytic amounts of the expensive cofactor are required, effectively decoupling the cost of chirality from the scale of production. The patent specifies that the enzyme can be utilized in various forms, including whole cells, broken cell lysates, or immobilized preparations, offering flexibility for different manufacturing setups. This mechanistic robustness ensures that the process remains consistent even when scaling from laboratory shake flasks to industrial fermenters, providing supply chain heads with the confidence needed for long-term capacity planning and risk mitigation.
How to Synthesize (3R, 3aS, 6aR)-hexahydrofuro[2,3-b]furan-3-ol Efficiently
The synthesis of this high-value intermediate follows a logical progression designed to maximize yield and purity while minimizing operational complexity. The process initiates with the halogenation of a gamma-lactone derivative, followed by acylation to install the necessary side chain functionality. Once the ketone precursor is prepared, the mixture is subjected to the biocatalytic reduction described previously, which sets the critical stereocenters. Subsequent steps involve coupling with an amine component, such as N-methylaniline, to form an amide linkage, followed by deprotection and a final cyclization event to close the bis-tetrahydrofuran ring system. Each step has been optimized in the patent examples to ensure high recovery and minimal byproduct formation. For detailed operational parameters, including specific solvent ratios, temperature profiles, and workup procedures, please refer to the standardized synthesis guide below which encapsulates the critical process parameters defined in the intellectual property.
- Perform halogenation of the starting ketone followed by acylation to prepare the substrate for biocatalysis.
- Execute the critical stereoselective reduction using aldehyde ketone reductase (SEQ ID NO: 1) with glucose dehydrogenase for cofactor regeneration.
- Complete the synthesis through amine coupling, deprotection, and acid-catalyzed ring closure to form the final bicyclic structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this enzymatic route translates into tangible competitive advantages that extend beyond simple chemistry. The elimination of expensive chiral ligands and transition metal catalysts results in a direct reduction in raw material expenditure, which is a primary driver for cost reduction in API manufacturing. Moreover, the mild reaction conditions reduce the energy load on production facilities and lower the requirements for specialized corrosion-resistant equipment, further decreasing capital expenditure (CAPEX) and operational expenditure (OPEX). The high selectivity of the enzyme minimizes the generation of difficult-to-separate impurities, which simplifies the purification train and reduces solvent consumption, aligning with sustainability goals and waste disposal regulations. This efficiency allows for a more agile supply chain capable of responding to market demands without the bottlenecks associated with complex chemical resolutions.
- Cost Reduction in Manufacturing: The substitution of stoichiometric chemical reducing agents and chiral auxiliaries with a catalytic enzymatic system fundamentally alters the cost structure of the synthesis. By utilizing a renewable biocatalyst that can be produced via fermentation, the dependency on fluctuating markets for precious metals and synthetic ligands is removed. The in-situ regeneration of the NADPH cofactor ensures that the cost per kilogram of product remains low even at massive scales. Additionally, the high conversion rates mean that less starting material is wasted, improving the overall mass balance and yield of the process. These factors combine to create a manufacturing protocol that is inherently more economical, allowing suppliers to offer competitive pricing while maintaining healthy margins.
- Enhanced Supply Chain Reliability: Reliance on a biologically derived catalyst that can be stored and transported as a stable powder or immobilized bead enhances supply security. Unlike some sensitive chemical catalysts that require strict cold-chain logistics or inert atmosphere handling, the engineered whole cells or enzyme preparations described in the patent demonstrate robust stability. This resilience reduces the risk of supply disruptions due to transportation delays or storage failures. Furthermore, the use of commodity chemicals for the upstream preparation of the substrate ensures that the supply chain is not vulnerable to the scarcity of exotic reagents. This reliability is crucial for maintaining continuous production schedules for life-saving antiretroviral medications, ensuring that patients have uninterrupted access to therapy.
- Scalability and Environmental Compliance: The process is explicitly designed with industrial scale-up in mind, as evidenced by the successful demonstration in multi-liter reactors with high substrate loading. The aqueous nature of the biocatalytic step reduces the volume of organic solvents required compared to purely chemical routes, significantly lowering the facility's environmental footprint. The waste stream is predominantly biological and biodegradable, simplifying wastewater treatment and reducing the burden of hazardous waste disposal. This alignment with green chemistry principles not only satisfies regulatory requirements but also enhances the corporate social responsibility profile of the manufacturing entity. The ability to scale this process from grams to metric tons without losing efficiency makes it an ideal candidate for meeting the growing global demand for HIV treatments.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. They are derived from a careful analysis of the experimental data and the comparative advantages highlighted in the patent specification. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer or licensing. The answers provided reflect the specific capabilities of the aldehyde ketone reductase system and its performance metrics relative to industry standards.
Q: What is the primary advantage of the enzymatic method in CN110372641B over traditional chemical reduction?
A: The enzymatic method utilizing aldehyde ketone reductase derived from Saccharomyces kudriavzevii achieves significantly higher optical purity (ee > 99%) and diastereoselectivity (de > 96%) under mild conditions (25-37°C), eliminating the need for expensive chiral ligands or harsh reducing agents required in conventional chemical routes.
Q: How does this process impact the cost structure for Darunavir intermediate manufacturing?
A: By replacing precious metal catalysts and complex chiral auxiliaries with a recyclable biocatalytic system, the process drastically reduces raw material costs and simplifies downstream purification, leading to substantial overall cost savings in large-scale API production.
Q: Is this synthetic route suitable for industrial scale-up?
A: Yes, the patent explicitly demonstrates high conversion rates (up to 97.8%) in large-volume reactors (e.g., 5L beakers) using whole-cell biocatalysts, indicating robust scalability and operational stability suitable for commercial metric-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Hexahydrofuro[2,3-b]furan-3-ol Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of securing a stable and high-quality supply of critical antiretroviral intermediates. As a premier CDMO partner, we possess the technical expertise and infrastructure to translate complex laboratory innovations like CN110372641B into commercial reality. Our facilities are equipped to handle diverse synthetic pathways, ranging from traditional organic synthesis to advanced biocatalytic processes. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, which utilize state-of-the-art analytical instrumentation to verify identity, potency, and impurity profiles against the highest international standards.
We invite you to collaborate with us to leverage this cutting-edge enzymatic technology for your Darunavir supply chain. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments. By partnering with NINGBO INNO PHARMCHEM, you gain access to a resilient supply network dedicated to advancing global health through superior chemical manufacturing excellence.
