Advanced Biocatalytic Synthesis of Niraparib Intermediates for Commercial Scale-up
Advanced Biocatalytic Synthesis of Niraparib Intermediates for Commercial Scale-up
The pharmaceutical industry is constantly seeking more efficient and sustainable pathways for the production of complex active pharmaceutical ingredients (APIs). A significant breakthrough in this domain is documented in patent CN116179503A, which discloses a novel recombinant aminotransferase and its application in the preparation of chiral intermediates for Niraparib. Niraparib, chemically known as 2-[4-((3S)-3-piperidinyl)phenyl]-2H-indole-7-carboxamide, is a potent poly(ADP-ribose) polymerase (PARP) inhibitor widely utilized in the maintenance treatment of recurrent ovarian cancer. 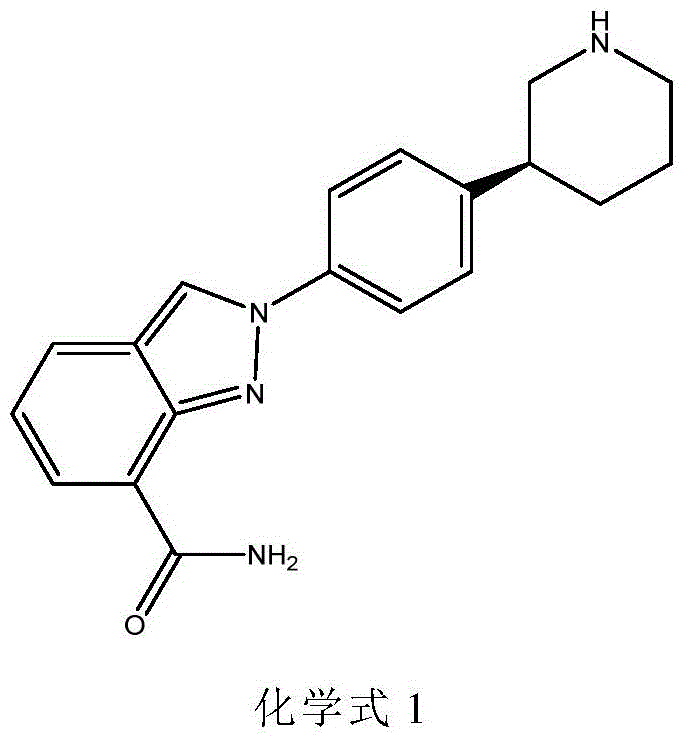 The structural complexity of this molecule, particularly the chiral piperidine moiety, has historically posed significant challenges for synthetic chemists aiming for high purity and yield. This patent introduces a biocatalytic solution that leverages an aminotransferase derived from Arthrobacter sp. KNK168, offering a transformative approach that bypasses the limitations of conventional chemical synthesis.
The structural complexity of this molecule, particularly the chiral piperidine moiety, has historically posed significant challenges for synthetic chemists aiming for high purity and yield. This patent introduces a biocatalytic solution that leverages an aminotransferase derived from Arthrobacter sp. KNK168, offering a transformative approach that bypasses the limitations of conventional chemical synthesis.
The core innovation lies in the utilization of a specific recombinant enzyme, designated as AT31, which exhibits exceptional stereoselectivity and regioselectivity. Unlike traditional methods that often struggle with racemic mixtures requiring tedious separation, this enzymatic route directly constructs the chiral center with high fidelity. The patent details the genetic engineering of this enzyme, including specific nucleotide and amino acid sequences (SEQ ID NO: 1 and SEQ ID NO: 2), as well as various mutants (SEQ ID NO: 3-11) optimized for performance. For R&D directors and process chemists, this represents a pivotal shift from harsh chemical conditions to mild, environmentally friendly biocatalysis, ensuring that the final product meets the rigorous quality standards required for oncology therapeutics while simplifying the overall manufacturing workflow.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Niraparib chiral intermediates has relied on multi-step chemical processes that are fraught with inefficiencies and high costs. The first-generation synthetic routes, such as those published by Jones P et al., typically involve Suzuki coupling reactions between p-nitroiodobenzene and 3-pyridineboronic acid, followed by hydrogenation and chiral resolution using tartaric acid. While functional, these methods often suffer from moderate yields and the generation of substantial chemical waste. Furthermore, the reliance on heavy metal catalysts like palladium necessitates extensive purification steps to remove trace metals, which is a critical concern for regulatory compliance in pharmaceutical manufacturing.
Second-generation improvements attempted to address catalyst stability by switching to PdCl2(dppf) and altering halogenated starting materials, yet they still depended heavily on chromatographic separation for chiral resolution. The use of preparative-scale chiral chromatography is a major bottleneck in industrial production; it is not only capital-intensive due to the cost of chiral stationary phases but also operationally slow, limiting throughput. Additionally, these chemical routes often require the use of toxic organic solvents and harsh reaction conditions, which pose safety risks and environmental liabilities. For supply chain managers, these factors translate into longer lead times, higher volatility in raw material costs, and increased complexity in waste management, making the conventional chemical synthesis less attractive for large-scale commercialization.
The Novel Approach
In stark contrast, the novel approach detailed in patent CN116179503A introduces a chemo-enzymatic strategy that fundamentally reimagines the construction of the chiral piperidine ring. By employing the recombinant AT31 transaminase, the process achieves asymmetric amination of a ketone precursor, specifically 4-(4-bromophenyl)-5-oxobutanoic acid isopropyl ester, to yield the desired (S)-5-(4-bromophenyl)piperidin-2-one. This biological transformation occurs under mild conditions, typically between 20°C and 50°C, and utilizes isopropylamine as a benign amine donor. The most significant advantage of this method is the complete elimination of chiral chromatography. The enzyme's inherent stereoselectivity ensures that the product is formed with an enantiomeric excess (ee) of greater than 99%, rendering downstream purification significantly simpler and more cost-effective.
This biocatalytic route also streamlines the upstream synthesis of the ketone substrate. The patent outlines an improved chemical synthesis for the precursor involving a Friedel-Crafts acylation followed by an esterification and a Corey-Chaykovsky reaction to install the necessary carbon framework. By optimizing reagents, such as using DMAP and DCC for esterification and NaH for the ylide generation, the yield of the ketone precursor is markedly improved before it even reaches the enzymatic step. For procurement teams, this integrated approach means fewer unit operations, reduced solvent consumption, and a drastic reduction in the overall cost of goods sold (COGS). The transition from a resolution-based process to a dynamic kinetic resolution or direct asymmetric synthesis model represents a paradigm shift in manufacturing efficiency for this high-value pharmaceutical intermediate.
Mechanistic Insights into AT31-Catalyzed Transamination
The heart of this technological advancement is the recombinant aminotransferase derived from Arthrobacter sp. KNK168. Transaminases, or aminotransferases, are pyridoxal-5'-phosphate (PLP) dependent enzymes that catalyze the transfer of an amino group from an amine donor to a carbonyl acceptor. In this specific application, the AT31 enzyme facilitates the conversion of the prochiral ketone substrate into the chiral amine product. The mechanism involves the formation of a Schiff base intermediate between the PLP cofactor and the amine donor (isopropylamine), generating pyridoxamine phosphate (PMP) and acetone as a byproduct. The PMP then transfers the amino group to the ketone substrate, regenerating PLP and releasing the chiral amine product. The patent highlights that the specific amino acid sequence of AT31 (SEQ ID NO: 2) provides a unique active site geometry that favors the formation of the (S)-enantiomer with exceptional precision.
Furthermore, the patent describes several mutants of the AT31 enzyme (SEQ ID NO: 3-11) where specific amino acid residues have been altered to enhance catalytic activity or stability. For instance, mutations at positions 85, 193, 317, and 380 are disclosed to potentially improve the enzyme's performance under process conditions. From an impurity control perspective, this enzymatic specificity is paramount. Chemical amination often leads to over-alkylation or the formation of regio-isomers, which are difficult to separate. However, the enzyme's active site acts as a molecular sieve, strictly accepting only the specific ketone substrate and directing the amination to the correct position. This intrinsic selectivity minimizes the formation of side products, resulting in a cleaner reaction profile that simplifies downstream processing and ensures the final API meets stringent impurity specifications required by global health authorities.
How to Synthesize (S)-5-(4-bromophenyl)piperidin-2-one Efficiently
The synthesis of this critical chiral intermediate involves a convergent strategy that combines optimized chemical steps with a final biocatalytic transformation. The process begins with the preparation of the ketone precursor through a series of robust chemical reactions, including acylation and esterification, which set the stage for the enzymatic step. The subsequent biocatalytic phase requires the precise preparation of the recombinant enzyme, ensuring high expression levels in the host organism, followed by the careful control of reaction parameters such as pH, temperature, and co-factor concentration. The detailed standardized synthesis steps below outline the specific protocols for enzyme expression, substrate preparation, and the final transamination reaction as described in the patent examples.
- Preparation of the ketone substrate 4-(4-bromophenyl)-5-oxobutanoic acid isopropyl ester via Friedel-Crafts acylation and subsequent ylide reaction.
- Expression of recombinant AT31 transaminase in E. coli BL21(DE3) using the pET28a vector system followed by cell harvesting and lyophilization.
- Biocatalytic transamination of the ketone substrate using the recombinant enzyme, PLP cofactor, and isopropylamine as the amine donor at mild temperatures.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this biocatalytic technology offers profound strategic advantages beyond mere technical novelty. The primary driver for cost reduction lies in the elimination of chiral chromatography. In traditional manufacturing, chiral separation columns are expensive consumables that require frequent replacement and generate significant solvent waste. By achieving high enantiomeric purity directly through enzymatic catalysis, the process removes this entire cost center. Additionally, the mild reaction conditions reduce energy consumption associated with heating and cooling, while the use of water-based buffers in the enzymatic step lowers the demand for hazardous organic solvents. These factors collectively contribute to a substantially lower cost of manufacturing, allowing for more competitive pricing in the global market for Niraparib intermediates.
Supply chain reliability is another critical benefit enhanced by this technology. Conventional routes relying on precious metal catalysts like palladium are susceptible to supply volatility and price fluctuations in the commodities market. The biocatalytic route utilizes recombinant enzymes produced via fermentation, a scalable and renewable resource that is not subject to the same geopolitical or mining constraints as rare earth metals. Moreover, the simplified workflow reduces the number of processing steps, which inherently shortens the manufacturing lead time. Fewer steps mean fewer opportunities for batch failures or delays, ensuring a more consistent and reliable supply of high-purity intermediates. This stability is crucial for pharmaceutical companies managing tight production schedules for oncology drugs where patient demand is continuous and critical.
Scalability and environmental compliance are increasingly becoming deciding factors in vendor selection. The enzymatic process described is inherently greener, operating at near-neutral pH and ambient temperatures, which aligns with modern sustainability goals and regulatory pressures to reduce carbon footprints. The avoidance of toxic solvents and heavy metals simplifies waste treatment and disposal, reducing the environmental liability for manufacturers. From a scale-up perspective, fermentation and biocatalysis are well-established technologies in the fine chemical industry, capable of being scaled from laboratory benchtop to multi-ton production with predictable outcomes. This ease of scale-up ensures that suppliers can rapidly respond to surges in market demand without the need for massive capital investment in new specialized equipment, thereby securing the supply chain against future disruptions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this recombinant aminotransferase technology. These insights are derived directly from the experimental data and comparative analysis presented in patent CN116179503A, providing clarity on how this method outperforms legacy synthesis routes. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this biocatalytic pathway into their existing supply chains.
Q: How does the new enzymatic route improve upon traditional chemical synthesis for Niraparib intermediates?
A: Traditional routes rely on Suzuki coupling followed by inefficient chiral resolution via tartaric acid or preparative chromatography. The new enzymatic route utilizes a recombinant AT31 transaminase to achieve direct asymmetric amination with >99% ee, completely eliminating the need for expensive and low-yield chiral separation columns.
Q: What are the specific performance metrics of the AT31 transaminase described in the patent?
A: The patent data indicates that the recombinant enzyme achieves a conversion rate of approximately 87% with an enantiomeric excess (ee) value exceeding 99%. This high stereoselectivity ensures that the resulting chiral intermediate meets stringent pharmaceutical purity requirements without further purification steps.
Q: Is this biocatalytic process suitable for large-scale industrial manufacturing?
A: Yes, the process is designed for scalability. It operates under mild reaction conditions (20-50°C) and avoids toxic organic solvents and heavy metal catalysts often found in previous generations. The elimination of chromatographic purification significantly reduces processing time and waste, making it highly favorable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Niraparib Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of biocatalysis in the production of complex pharmaceutical intermediates like those for Niraparib. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes are successfully translated into robust industrial realities. Our commitment to quality is unwavering, supported by stringent purity specifications and rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch. We understand that in the oncology sector, there is no margin for error, and our infrastructure is designed to deliver the consistency and reliability that global pharmaceutical partners demand.
We invite you to collaborate with us to leverage this advanced enzymatic technology for your Niraparib supply chain. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how switching to this biocatalytic route can optimize your budget. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. By partnering with NINGBO INNO PHARMCHEM, you gain access to cutting-edge synthesis capabilities that combine scientific excellence with commercial pragmatism, securing your position in the competitive landscape of PARP inhibitor manufacturing.
