Advanced Biocatalytic Synthesis of (S)-1-(3-Chlorophenyl)-1,3-Propanediol for Commercial Scale-Up
Introduction to Next-Generation Chiral Diol Synthesis
The pharmaceutical industry constantly seeks more efficient and sustainable routes for critical chiral intermediates, particularly those used in antiviral and anticancer drug development. A pivotal advancement in this domain is detailed in Patent CN115537405A, which discloses a novel ketoreductase mutant specifically engineered for the preparation of (S)-1-(3-chlorophenyl)-1,3-propanediol. This molecule serves as a vital building block for synthesizing cyclic phosphate prodrugs, including derivatives of Tenofovir and Lenvatinib, as well as compounds for treating Hepatitis C. The structural integrity and chiral purity of this diol are paramount for the efficacy of the final active pharmaceutical ingredients (APIs).
The innovation lies in overcoming the limitations of traditional chemical synthesis through semi-rational protein engineering. By modifying the amino acid sequence of a wild-type ketoreductase derived from Novosphingobium aromaticivorans, researchers have achieved a biocatalyst capable of operating under mild conditions with exceptional stereoselectivity. This breakthrough addresses the growing demand for a reliable pharmaceutical intermediate supplier who can deliver high-quality chiral synthons without the environmental burden of heavy metal catalysts.
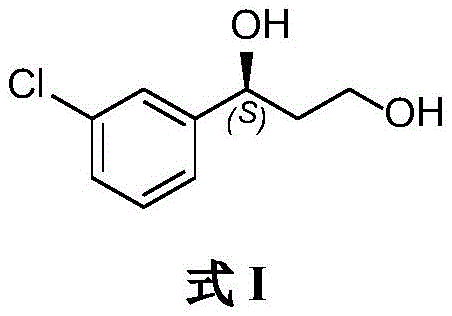
Understanding the molecular architecture is crucial for R&D teams evaluating process feasibility. The target molecule features a chlorophenyl ring attached to a chiral carbon bearing a hydroxyl group, followed by an ethylene glycol chain. The precise control over the (S)-configuration at the benzylic position is the primary challenge that this new biocatalytic method solves with remarkable efficiency, setting a new standard for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
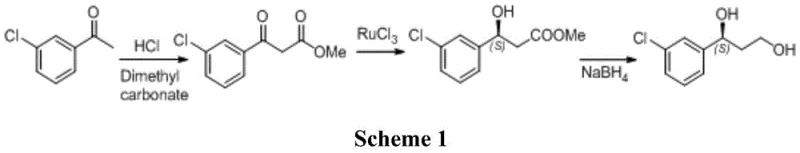
Historically, the synthesis of (S)-1-(3-chlorophenyl)-1,3-propanediol has relied on chemically intensive routes that pose significant safety and environmental challenges. One prominent method, described in patent WO2015154716 and illustrated in Scheme 1, utilizes 3-chloroacetophenone and dimethyl carbonate. While effective, this route depends heavily on Ruthenium Trichloride (RuCl3), a corrosive and toxic transition metal catalyst. The presence of such heavy metals necessitates rigorous and costly purification steps to meet strict regulatory limits for residual metals in pharmaceutical products, thereby complicating the supply chain and increasing production costs.
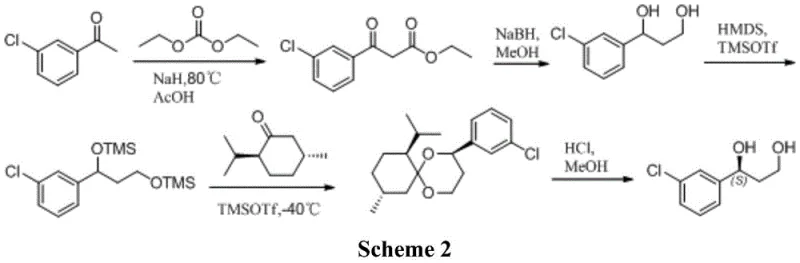
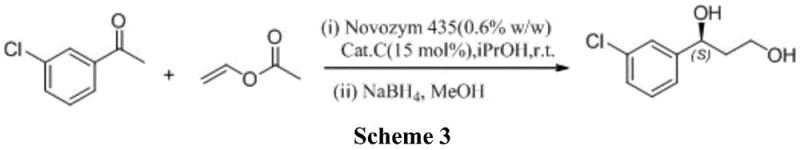
Alternative chemical strategies, such as the one reported in the Journal of Medicinal Chemistry (Scheme 2), involve even more convoluted sequences. This approach requires the protection of hydroxyl groups using hexamethyldisilazane (HMDS) and trimethylsilyl trifluoromethanesulfonate (TMSOTf), followed by a ketalization reaction with menthone to separate diastereomers. The necessity for silica gel column chromatography to isolate the desired isomer renders this method impractical for industrial scale-up due to low throughput and excessive solvent consumption. Furthermore, earlier enzymatic attempts using lipases (Scheme 3) suffered from prolonged reaction times (up to 96 hours) and mediocre enantiomeric excess (87% ee), failing to meet the stringent quality requirements of modern drug manufacturing.
The Novel Approach
In stark contrast to these legacy methods, the process disclosed in Patent CN115537405A offers a streamlined, green chemistry solution. As depicted in Scheme 5, the new route begins with the condensation of 3-chlorobenzoic acid and potassium monomethyl malonate to form a beta-keto ester intermediate. The critical step is the asymmetric reduction of this keto-ester using the engineered ketoreductase (KRED). This biocatalytic transformation occurs in an aqueous buffer system at a mild temperature of 40°C, completely eliminating the need for hazardous organic solvents or toxic metal catalysts during the chirality-setting step.
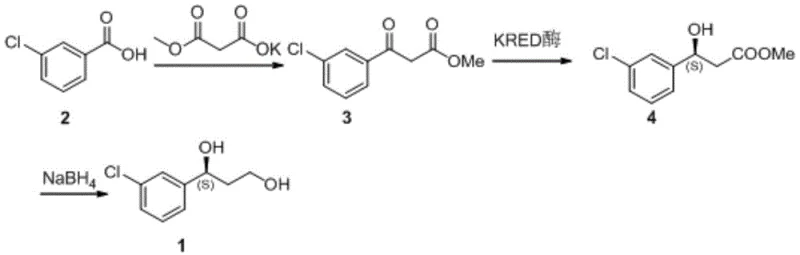
The final step involves a straightforward chemical reduction of the ester moiety using sodium borohydride to yield the target diol. This hybrid chemo-enzymatic strategy combines the precision of biocatalysis with the robustness of chemical reduction. For procurement managers, this translates to cost reduction in API manufacturing by simplifying the workflow, reducing waste disposal costs associated with heavy metals, and shortening the overall production timeline. The ability to achieve high conversion rates without complex separation techniques makes this route exceptionally attractive for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Ketoreductase-Catalyzed Asymmetric Reduction
The core of this technological leap is the specific engineering of the ketoreductase enzyme. The inventors utilized semi-rational design to modify the wild-type enzyme from Novosphingobium aromaticivorans. Through saturation mutation at key hotspot residues—specifically P41, S58, G141, I221, and G254—they developed the Mut1 variant (P41G/S58V/G141A/I221V/G254H). These mutations alter the steric environment of the enzyme's active site, creating a tighter fit for the bulky 3-chlorophenyl substrate while excluding the formation of the (R)-enantiomer. This precise molecular recognition is what drives the enantiomeric excess (ee) to an impressive 99.6%, far surpassing the 87% ee of previous lipase-based methods.
From an impurity control perspective, the mechanism ensures that side reactions are minimized. The high specificity of the Mut1 enzyme means that fewer by-products are generated during the reduction of the ketone group. In traditional chemical reductions using borohydrides or aluminum hydrides without chiral ligands, racemic mixtures are common, requiring difficult resolution steps later. Here, the chirality is established early in the synthesis with near-perfect fidelity. This reduces the burden on downstream purification units and ensures a cleaner impurity profile for the final API, a critical factor for R&D directors validating process robustness.
How to Synthesize (S)-1-(3-Chlorophenyl)-1,3-Propanediol Efficiently
Implementing this biocatalytic route requires careful attention to enzyme formulation and reaction parameters to maximize yield and turnover. The process is designed to be scalable, utilizing readily available starting materials and standard fermentation or lyophilized enzyme preparations. The following guide outlines the critical operational phases derived from the patent examples, focusing on the preparation of the key chiral intermediate.
- Preparation of the keto-ester substrate (Compound 3) from 3-chlorobenzoic acid and potassium monomethyl malonate.
- Asymmetric reduction of Compound 3 to Compound 4 using the engineered ketoreductase mutant (Mut1) at 40°C with NADP cofactor regeneration.
- Final chemical reduction of the ester group in Compound 4 using sodium borohydride to yield the target diol.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders, the shift from metal-catalyzed or multi-step protection chemistry to this enzymatic route represents a significant strategic advantage. The elimination of Ruthenium and other transition metals removes a major bottleneck in raw material sourcing and waste management. Heavy metal catalysts often face supply volatility and require specialized handling protocols; replacing them with a biocatalyst stabilizes the supply chain and reduces regulatory compliance risks associated with metal residues in drug substances.
- Cost Reduction in Manufacturing: The new process drastically simplifies the synthetic sequence by removing the need for expensive silyl protecting groups and chromatographic separations. By avoiding reagents like TMSOTf and HMDS, which are costly and generate significant acidic waste, the overall material cost is substantially lowered. Furthermore, the high substrate loading capacity (up to 200g/L) mentioned in the patent implies that smaller reactor volumes can produce the same amount of product, leading to better capital efficiency and reduced utility costs per kilogram of output.
- Enhanced Supply Chain Reliability: Biocatalytic processes are generally more robust against fluctuations in raw material quality compared to sensitive organometallic reactions. The use of a stable enzyme mutant ensures consistent batch-to-batch quality, reducing the risk of production delays caused by failed reactions or off-spec material. This reliability is essential for maintaining continuous supply to downstream API manufacturers, especially for critical medications like antivirals where supply interruptions can have severe public health consequences.
- Scalability and Environmental Compliance: Operating at mild temperatures (40°C) and neutral pH significantly reduces energy consumption compared to high-temperature chemical refluxes. Additionally, the aqueous nature of the biocatalytic step minimizes the use of volatile organic compounds (VOCs), aligning with increasingly strict global environmental regulations. This 'green' profile not only facilitates easier permitting for new manufacturing facilities but also enhances the sustainability credentials of the final pharmaceutical product, a growing priority for major pharma buyers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel ketoreductase technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear picture of the process capabilities.
Q: What are the advantages of the new ketoreductase mutant over traditional chemical synthesis?
A: The new mutant eliminates the need for corrosive metal catalysts like Ruthenium Trichloride and avoids complex protection-deprotection steps, resulting in milder conditions and higher chiral purity (>99% ee).
Q: What is the substrate loading capacity of this biocatalytic process?
A: The optimized process allows for a high substrate concentration of up to 200g/L, which significantly improves volumetric productivity compared to previous enzymatic methods.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method features mild reaction temperatures (40°C), high conversion rates (>99%), and simplified downstream processing, making it highly viable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-1-(3-Chlorophenyl)-1,3-Propanediol Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the biocatalytic route described in Patent CN115537405A for the production of high-value chiral diols. As a leading CDMO partner, we possess the technical expertise to adapt and optimize such advanced enzymatic pathways for industrial application. Our facilities are equipped with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volume demands of global pharmaceutical clients. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of (S)-1-(3-chlorophenyl)-1,3-propanediol meets the highest standards of chiral purity and chemical identity.
We invite procurement teams and R&D directors to collaborate with us to leverage this cutting-edge technology for your specific drug development programs. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your project's unique requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring a seamless transition from laboratory innovation to commercial reality.
