Scalable Production of 2-Amino-4-Acetamidoanisole via Catalytic Hydrogenation for Dye Intermediates
Scalable Production of 2-Amino-4-Acetamidoanisole via Catalytic Hydrogenation for Dye Intermediates
The global demand for high-performance disperse dyes, particularly deep blue varieties, necessitates a robust and reliable supply chain for critical coupling components like 2-amino-4-acetamidoanisole. As detailed in patent CN100368385C, a transformative manufacturing methodology has been established that shifts away from polluting traditional reduction techniques toward a sophisticated catalytic hydrogenation approach. This technological leap addresses the longstanding challenges of yield optimization and environmental compliance in the fine chemicals sector. By leveraging methanol as a dual-purpose solvent for both reaction and crystallization, the process ensures exceptional product consistency while minimizing waste generation. For R&D directors and procurement specialists seeking a reliable dye intermediate supplier, understanding the mechanistic advantages of this route is essential for securing long-term supply stability. The following analysis dissects the technical nuances that make this process a benchmark for modern industrial synthesis.
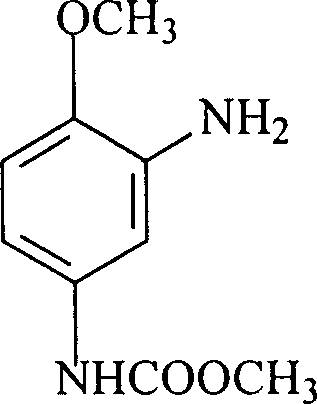
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of 2-amino-4-acetamidoanisole relied on a cumbersome three-step sequence starting from p-aminoanisole, involving acylation, nitration, and finally reduction using iron powder or sodium sulfide. This legacy pathway is fraught with inefficiencies, primarily characterized by a cumulative yield that stagnates around merely 76%, which is economically suboptimal for large-scale operations. Furthermore, the reliance on iron powder reduction generates massive quantities of hazardous solid waste, specifically iron mud, alongside acidic wastewater and phenolic effluents that are costly and difficult to treat. The use of sulfide reducing agents introduces additional toxicity concerns, creating a significant burden on environmental management systems and increasing the overall cost of compliance. These factors collectively render the traditional method unsustainable in the context of modern green chemistry standards and stringent regulatory frameworks.
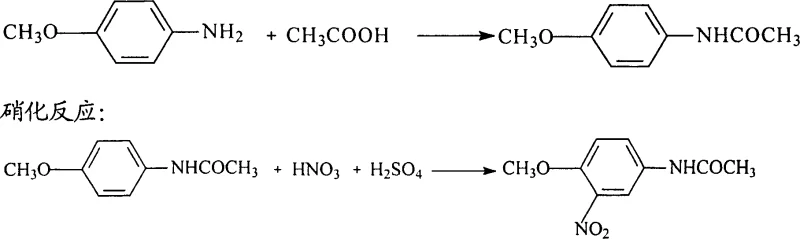
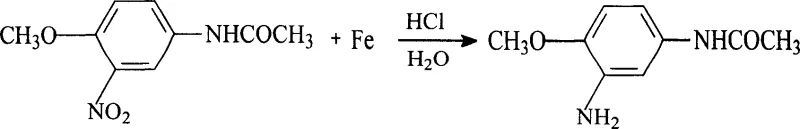
The Novel Approach
In stark contrast, the innovative process described in the patent utilizes 2,4-dinitroanisole as the starting material, subjecting it directly to catalytic hydrogenation in a methanol medium. This strategic pivot eliminates the need for hazardous reducing agents like iron or sulfides, thereby fundamentally altering the waste profile of the manufacturing operation. The subsequent acylation is performed in situ or in the same solvent system, which streamlines the workflow and reduces the number of isolation steps required. By integrating the reduction and acylation phases more closely, the process minimizes material loss during transfer and purification stages. This approach not only enhances the overall throughput but also aligns with the principles of atom economy, making it a superior choice for cost reduction in dye intermediate manufacturing where margin compression is a constant pressure.
Mechanistic Insights into Catalytic Hydrogenation and Selective Acylation
The core of this advanced synthesis lies in the precise control of the catalytic hydrogenation step, typically employing Raney-Ni or Pd-C catalysts under elevated temperatures ranging from 120°C to 200°C and pressures between 2.4 MPa and 10.0 MPa. Under these conditions, the nitro groups on the 2,4-dinitroanisole ring are selectively reduced to amino groups without compromising the integrity of the methoxy substituent or causing excessive hydrogenolysis. The choice of methanol as the solvent is critical, as it effectively lowers the viscosity of the reaction mixture, facilitating better dispersion of the catalyst particles and improving heat transfer dynamics within the reactor. This ensures uniform reaction kinetics and prevents localized hot spots that could lead to runaway reactions or the formation of unwanted byproducts. The result is a high conversion rate of the nitro precursor to the diamino intermediate, setting the stage for a highly efficient subsequent acylation.
Following the reduction, the process incorporates a highly selective acylation step where acetic anhydride is introduced in the presence of acid-binding agents such as sodium carbonate or magnesium oxide. A key mechanistic advantage here is the suppression of bis-acylation, a common side reaction that can plague aniline derivatives; the patented method successfully keeps bis-acylated impurities below 1%. This level of selectivity is achieved through careful temperature control during the addition of acetic anhydride, often maintaining the reaction between 0°C and 60°C. The resulting crude product is then subjected to a refined crystallization process using methanol, exploiting the specific solubility characteristics of 2-amino-4-acetamidoanisole to achieve a final purity exceeding 98%. This rigorous control over the impurity profile is vital for downstream dye applications where color strength and hue consistency are paramount.
How to Synthesize 2-Amino-4-Acetamidoanisole Efficiently
Implementing this synthesis route requires a coordinated sequence of unit operations designed to maximize safety and efficiency. The process begins with the loading of 2,4-dinitroanisole and the chosen catalyst into a high-pressure hydrogenation reactor, followed by the introduction of hydrogen gas under controlled thermal conditions. Once the reduction is complete, the mixture undergoes solid-liquid separation to recover the valuable catalyst for reuse, a step that is crucial for economic viability. The filtrate then proceeds to the acylation reactor where stoichiometric amounts of acetic anhydride and base are added to form the target amide bond. Finally, the product is isolated through cooling crystallization and filtration, yielding a high-quality solid ready for drying and packaging. For detailed operational parameters and safety protocols, please refer to the standardized guide below.
- Perform catalytic hydrogenation of 2,4-dinitroanisole in methanol using Raney-Ni or Pd-C catalyst at 120-200°C and 2.4-10.0 MPa.
- Separate the catalyst via filtration and recover it for recycling, then proceed to acylation with acetic anhydride.
- Crystallize the crude product from methanol solution to obtain high-purity 2-amino-4-acetamidoanisole (>98%).
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this catalytic hydrogenation technology offers profound advantages that extend beyond mere technical superiority. For procurement managers, the elimination of iron powder and sulfide reagents translates directly into a simplified raw material basket, reducing exposure to volatile commodity markets associated with metal powders. The ability to recycle the methanol solvent across both the reaction and crystallization stages significantly lowers the consumption of bulk solvents, driving down variable production costs. Moreover, the high recovery rate of the catalyst, reported to reach up to 99.5% with efficient filtration systems, ensures that expensive precious metals or nickel are not lost to the waste stream. These efficiencies cumulatively contribute to a more competitive pricing structure for the final intermediate, allowing buyers to negotiate better terms without sacrificing quality.
- Cost Reduction in Manufacturing: The transition to a clean catalytic process removes the substantial costs associated with treating hazardous iron sludge and sulfide wastewater, which are major expense centers in traditional dye intermediate plants. By avoiding these waste treatment burdens, manufacturers can allocate resources toward process optimization and capacity expansion rather than environmental remediation. Additionally, the high single-pass conversion rates and minimal byproduct formation mean that less raw material is wasted, further enhancing the overall cost-effectiveness of the production line. This structural cost advantage provides a buffer against market fluctuations and ensures stable pricing for long-term contracts.
- Enhanced Supply Chain Reliability: The robustness of the catalytic hydrogenation method ensures consistent batch-to-batch quality, which is critical for dye manufacturers who cannot afford variations in coupling component performance. The high purity of the product (>98%) reduces the need for extensive rework or blending at the customer's site, streamlining the entire value chain. Furthermore, the scalability of the equipment, which can range from batch stirred tanks to continuous fixed-bed reactors, allows suppliers to rapidly ramp up production in response to surges in demand. This flexibility mitigates the risk of supply shortages and ensures that downstream dye production schedules are met without interruption.
- Scalability and Environmental Compliance: As global regulations on industrial emissions tighten, the "clean" nature of this process becomes a strategic asset for supply chain continuity. The absence of heavy metal waste and the reduction in organic effluent load make it easier for facilities to maintain their operating licenses and avoid shutdowns due to non-compliance. The process is inherently safer, operating with manageable exotherms and avoiding the handling of pyrophoric iron powders on a massive scale. This safety profile reduces insurance premiums and operational risks, making the supply chain more resilient to disruptions caused by safety incidents or regulatory crackdowns.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 2-amino-4-acetamidoanisole using this advanced catalytic route. These insights are derived directly from the patent specifications and practical implementation data, providing clarity on purity standards, waste management, and catalyst lifecycle. Understanding these details helps stakeholders make informed decisions about integrating this intermediate into their supply chains. For further technical discussions or custom feasibility studies, our team is available to provide deeper analysis.
Q: What is the purity level achievable with this new hydrogenation process?
A: The patented process utilizing catalytic hydrogenation followed by crystallization refinement achieves a product purity of over 98%, with bis-acylated byproducts controlled to less than 1%.
Q: How does this method improve environmental compliance compared to traditional iron powder reduction?
A: Unlike traditional methods that generate significant iron sludge and sulfide wastewater, this catalytic route eliminates heavy metal waste and allows for solvent recycling, resulting in a cleaner production profile.
Q: Can the catalyst be reused effectively in this synthesis?
A: Yes, the process employs efficient blade filtration systems that allow for catalyst recovery rates reaching up to 99.5%, significantly reducing raw material consumption and operational costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Amino-4-Acetamidoanisole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to cleaner, more efficient chemical processes is not just a regulatory requirement but a strategic imperative for growth. Our expertise in scaling diverse pathways from 100 kgs to 100 MT/annual commercial production ensures that we can meet the rigorous demands of the global dye industry. We are committed to delivering 2-amino-4-acetamidoanisole with stringent purity specifications, supported by our rigorous QC labs that verify every batch against the highest international standards. Our facility is equipped to handle the high-pressure hydrogenation and precise crystallization steps required by this patent, guaranteeing a supply of high-purity dye intermediates that empower your formulation teams to create superior products.
We invite you to collaborate with us to optimize your sourcing strategy and leverage the cost benefits of this advanced manufacturing technology. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation can drive value for your organization.
