Revolutionizing 2-Amino-4-Acetamidoanisole Production: Solvent-Free Hydrogenation for Commercial Scale
The chemical industry is constantly evolving towards safer and more sustainable manufacturing processes, and patent CN108373424B represents a significant breakthrough in the synthesis of 2-amino-4-acetamidoanisole. This critical intermediate, widely utilized in the production of disperse dyes such as Disperse Blue 79 and 291, has traditionally been manufactured using hazardous organic solvents. The disclosed innovation introduces a novel solvent-free hydrogenation technique where the molten product itself serves as the reaction medium. This paradigm shift not only addresses critical safety concerns associated with flammable solvents but also streamlines the post-reaction separation process. By eliminating the need for solvent recovery and distillation, the method offers a compelling value proposition for manufacturers seeking to optimize their cost reduction in dye intermediate manufacturing while maintaining rigorous quality standards. The technical robustness of this approach positions it as a preferred route for commercial scale-up of complex dye intermediates.
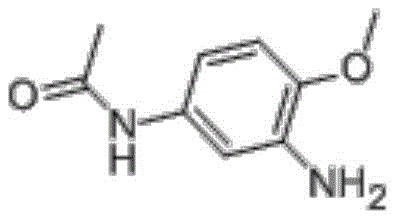
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the reduction of 2-nitro-4-acetamidoanisole to its amino counterpart has relied heavily on catalytic hydrogenation in the presence of volatile organic solvents such as methanol or ethanol. While effective, these conventional methods introduce substantial operational risks due to the flammability and explosivity of the solvents, particularly under the elevated pressures required for hydrogenation. Furthermore, the post-reaction workflow is energy-intensive, necessitating complex distillation columns to recover and recycle the solvent, which significantly drives up utility costs. The presence of residual solvent in the crude product can also complicate downstream purification, potentially affecting the color light and strength of the final dye application. These factors collectively create bottlenecks in reducing lead time for high-purity dye intermediates and increase the environmental footprint of the manufacturing facility through solvent emissions and wastewater treatment requirements.
The Novel Approach
In stark contrast, the technology described in CN108373424B utilizes a melt-phase reaction system where the product, 2-amino-4-acetamidoanisole, acts as its own solvent. This ingenious design completely removes the dependency on external organic solvents, thereby inherently mitigating the risks of fire and explosion associated with volatile liquids. The process operates at temperatures between 110°C and 180°C, keeping the reaction mixture in a molten state that facilitates efficient mass transfer and hydrogen diffusion without the dilution effect of a solvent. Following the reaction, the catalyst is separated via hot filtration, and the product is obtained directly through cooling and crystallization. This simplification of the workflow translates to significant cost savings by removing solvent procurement and recovery steps, making it an ideal solution for a reliable dye intermediate supplier aiming to enhance operational efficiency and safety profiles.
Mechanistic Insights into Raney Nickel-Catalyzed Hydrogenation
The core of this synthesis lies in the catalytic hydrogenation of the nitro group to an amino group using a Raney Nickel catalyst within a molten matrix. The reaction mechanism involves the adsorption of hydrogen gas and the nitro substrate onto the active sites of the skeletal nickel surface. In the absence of a solvent, the concentration of the reactant is maximized, which can enhance the reaction kinetics provided that the viscosity of the melt allows for adequate stirring and gas-liquid-solid contact. The use of Raney Nickel is particularly advantageous due to its high activity and selectivity for nitro reduction, ensuring that the acetamido group remains intact without hydrolysis, which is a common side reaction in acidic or basic aqueous environments. This selectivity is crucial for maintaining the structural integrity required for subsequent diazotization and coupling reactions in dye synthesis.
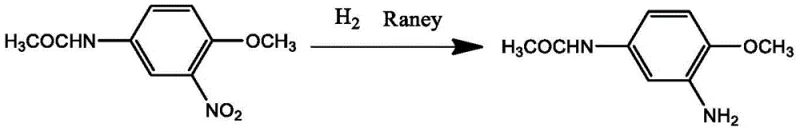
Impurity control in this solvent-free system is inherently superior due to the absence of solvent-derived byproducts. In traditional methods, solvent impurities or their degradation products can often co-crystallize with the product, requiring additional recrystallization steps. Here, the primary impurities are limited to unreacted starting material or over-reduction products, which are minimized by precise control of hydrogen pressure (0.1-10 MPa) and temperature. The hot filtration step effectively removes the solid catalyst, preventing metal contamination in the final product, which is a critical specification for high-purity dye intermediates used in sensitive textile applications. The resulting product exhibits excellent color light and high-temperature dispersion stability, as evidenced by comparative data showing improved performance over solvent-based counterparts.
How to Synthesize 2-Amino-4-Acetamidoanisole Efficiently
Implementing this solvent-free hydrogenation process requires precise control over thermal parameters and catalyst loading to ensure consistent batch quality. The procedure begins by charging molten 2-amino-4-acetamidoanisole into a pressure reactor, followed by the addition of the nitro precursor and the Raney Nickel catalyst. The system is then pressurized with hydrogen and heated to the optimal reaction window, typically between 120°C and 160°C, to maintain the melt state while driving the reduction to completion. Detailed standard operating procedures regarding catalyst activation, pressure ramping, and hot filtration techniques are essential for successful technology transfer. For a comprehensive guide on the specific operational parameters and safety protocols, please refer to the standardized synthesis steps outlined below.
- Charge molten 2-amino-4-acetamidoanisole and 2-nitro-4-acetamidoanisole into a reactor with Raney Nickel catalyst.
- Conduct catalytic hydrogenation at 110-180°C and 0.1-10 MPa pressure until reaction completion.
- Perform hot filtration to recover catalyst, then cool the filtrate to crystallize the final high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this solvent-free technology offers profound advantages in terms of cost structure and supply chain resilience. By eliminating the need for large volumes of organic solvents, manufacturers can drastically reduce raw material costs and the logistical burden associated with solvent storage and handling. The simplified process flow also means shorter cycle times and higher throughput, allowing suppliers to respond more敏捷 ly to market demand fluctuations. This efficiency gain is critical for maintaining a reliable dye intermediate supplier status in a competitive global market where consistency and speed are paramount. Furthermore, the reduced energy consumption from avoiding solvent distillation contributes to a lower carbon footprint, aligning with the increasing environmental compliance requirements of multinational corporations.
- Cost Reduction in Manufacturing: The elimination of organic solvents removes a significant variable cost component from the production budget. Without the need for solvent recovery systems, capital expenditure on equipment is also reduced, and operational expenses related to energy for distillation are drastically simplified. The ability to recycle the catalyst further enhances the economic viability of the process, leading to substantial cost savings over the lifecycle of the production campaign. These efficiencies allow for more competitive pricing structures without compromising on the quality of the high-purity dye intermediates delivered to the market.
- Enhanced Supply Chain Reliability: Removing flammable solvents from the process significantly lowers the safety risk profile of the manufacturing facility, reducing the likelihood of production stoppages due to safety incidents. The simplified workflow also minimizes the number of unit operations, thereby reducing the potential points of failure in the production line. This robustness ensures a more consistent supply of materials, which is essential for downstream dye manufacturers who rely on just-in-time inventory models. The process inherently supports commercial scale-up of complex dye intermediates by providing a safer and more manageable production environment.
- Scalability and Environmental Compliance: The solvent-free nature of this reaction makes it inherently more environmentally friendly, as it generates less hazardous waste and wastewater. This aligns well with strict environmental regulations in major chemical manufacturing hubs, ensuring long-term operational continuity. The process is easily scalable from pilot to commercial volumes because the heat transfer and mixing dynamics are more straightforward without the bulk of a solvent. This scalability ensures that supply can be ramped up quickly to meet surging demand, reducing lead time for high-purity dye intermediates and securing the supply chain against disruptions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 2-amino-4-acetamidoanisole using this advanced hydrogenation method. These insights are derived directly from the patent data and practical industrial experience, aiming to clarify the benefits for R&D and procurement stakeholders. Understanding these details is crucial for evaluating the feasibility of integrating this material into your supply chain.
Q: How does the solvent-free method improve safety compared to traditional methanol reduction?
A: The novel method eliminates the use of flammable organic solvents like methanol, significantly reducing the risk of fire and explosion under high-pressure hydrogenation conditions.
Q: What is the impact on product purity and dye performance?
A: Data indicates that the solvent-free process yields products with purity exceeding 99%, resulting in disperse dyes with superior color strength and high-temperature dispersion stability.
Q: Is the catalyst reusable in this new process?
A: Yes, the solid catalyst (Raney Nickel) can be separated via hot filtration, washed, and directly recycled for subsequent batches, enhancing process economics.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Amino-4-Acetamidoanisole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your final products. Our technical team has extensively analyzed the solvent-free hydrogenation pathway described in CN108373424B and possesses the expertise to implement this technology at an industrial scale. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab to plant is seamless and efficient. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 2-amino-4-acetamidoanisole meets the exacting standards required for premium disperse dye manufacturing.
We invite you to collaborate with us to optimize your supply chain and leverage the cost benefits of this innovative process. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact our technical procurement team to request specific COA data and route feasibility assessments that demonstrate how we can support your production goals with reliability and precision.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
