Revolutionizing Florfenicol Production: Enzymatic Dynamic Kinetic Resolution for Commercial Scale
Introduction to Advanced Florfenicol Synthesis
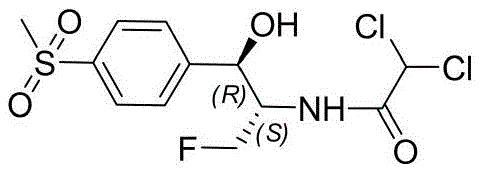
The global demand for effective veterinary antibiotics continues to drive innovation in the synthesis of florfenicol, a critical broad-spectrum antimicrobial agent. Patent CN111500652A introduces a transformative biocatalytic approach that addresses long-standing inefficiencies in the manufacturing of this essential veterinary drug. Unlike traditional methods that rely on cumbersome chemical resolutions and heavy metal catalysts, this novel technology leverages the power of engineered ketoreductases to achieve high stereoselectivity through dynamic kinetic resolution (DKR). This shift represents a paradigm change in how we approach the production of chiral intermediates for animal health pharmaceuticals, offering a pathway that is not only chemically elegant but also commercially superior.
The core of this innovation lies in the conversion of aziridine ketone precursors directly into the desired chiral alcohol intermediates with exceptional optical purity. By integrating enzymatic catalysis with downstream chemical modifications, the process simplifies the synthetic route significantly. For manufacturers seeking a reliable veterinary drug intermediate supplier, understanding the mechanistic advantages of this patent is crucial, as it lays the groundwork for a more sustainable and cost-effective supply chain. The ability to bypass traditional resolution steps means that producers can potentially double their theoretical yield from the same amount of starting material, a factor that fundamentally alters the economic landscape of florfenicol production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
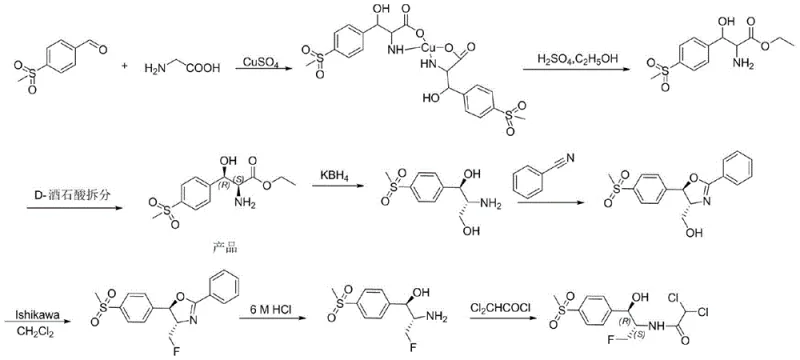
Historically, the industrial preparation of florfenicol has been plagued by significant inefficiencies and environmental burdens. The classical route typically begins with p-methylsulfonylbenzaldehyde and glycine, proceeding through a series of steps including condensation, esterification, and, most critically, chiral resolution to isolate the D-ethyl ester intermediate. This resolution step is inherently wasteful, discarding approximately 50% of the raw material as the unwanted enantiomer, which drastically inflates production costs and reduces overall atom economy. Furthermore, the process relies heavily on copper sulfate for complexation, generating substantial volumes of heavy metal-containing wastewater that require expensive and complex treatment protocols to meet environmental regulations.
Beyond the resolution issues, the subsequent chemical transformations involve harsh reagents and difficult conditions. The reduction of the D-ethyl ester often utilizes boron-based reagents, leading to boron-salt wastewater that is notoriously difficult to treat. Additionally, the fluorination step traditionally employs the Ishikawa reagent, which suffers from low fluorine atom utilization rates, high costs, and severe corrosiveness to reactor equipment. These cumulative factors result in a manufacturing process with a high E-factor (environmental factor), characterized by excessive waste generation and safety hazards, making it increasingly untenable in a regulatory environment that demands greener chemistry.
The Novel Approach
In stark contrast, the method disclosed in CN111500652A offers a streamlined alternative that circumvents these historical bottlenecks. By utilizing a specific ketoreductase to catalyze the reduction of aziridine ketones, the process achieves dynamic kinetic resolution, theoretically allowing for 100% conversion of the racemic starting material into the single desired enantiomer. This eliminates the 50% yield loss associated with classical resolution and removes the need for copper sulfate complexation entirely. The reaction proceeds under mild physiological conditions (around 35°C and pH 7.5) in aqueous buffers, significantly reducing energy consumption and eliminating the generation of heavy metal waste streams.
Furthermore, the versatility of this enzymatic approach allows for the use of various protecting groups (such as acetyl, benzyl, or dichloroacetyl) on the aziridine nitrogen, providing flexibility in downstream processing. In the most optimized embodiment where the dichloroacetyl group is pre-installed, the synthesis is shortened dramatically, requiring fewer isolation steps and reducing solvent usage. This novel route not only enhances the purity profile of the intermediate, achieving >99% ee and >94% de, but also aligns with modern principles of green chemistry, offering a compelling value proposition for cost reduction in antibiotic manufacturing while ensuring a cleaner environmental footprint.
Mechanistic Insights into Ketoreductase-Catalyzed Dynamic Kinetic Resolution
The heart of this technological advancement is the enzymatic dynamic kinetic resolution (DKR) mechanism, which elegantly solves the problem of chirality control. In a standard kinetic resolution, an enzyme selectively reacts with only one enantiomer of a racemic mixture, limiting the maximum yield to 50%. However, in this DKR system, the ketoreductase works in tandem with the inherent racemization capability of the aziridine ketone substrate under the reaction conditions. As the enzyme selectively reduces the preferred enantiomer to the chiral alcohol, the remaining unreacted enantiomer rapidly racemizes in situ, replenishing the pool of the reactive substrate. This continuous cycle allows for the theoretical conversion of the entire racemic starting material into the single desired chiral product.
The stereoselectivity is governed by the precise active site architecture of the engineered ketoreductases (such as YH2068, YH2077, or YH2045), which discriminate between the pro-chiral faces of the ketone with extreme fidelity. This results in the formation of the (R)-hydroxyl configuration with >99% enantiomeric excess (ee). Simultaneously, the configuration at the aziridine ring is controlled to ensure the correct diastereomeric ratio (de > 94%). This high level of stereocontrol is critical because any deviation can lead to diastereomeric impurities that are difficult to remove later and may affect the biological efficacy of the final veterinary drug. The use of a cofactor regeneration system (e.g., glucose/glucose dehydrogenase or isopropanol/alcohol dehydrogenase) ensures that the expensive NADH/NADPH cofactor is recycled efficiently, making the process economically viable for large-scale operations.
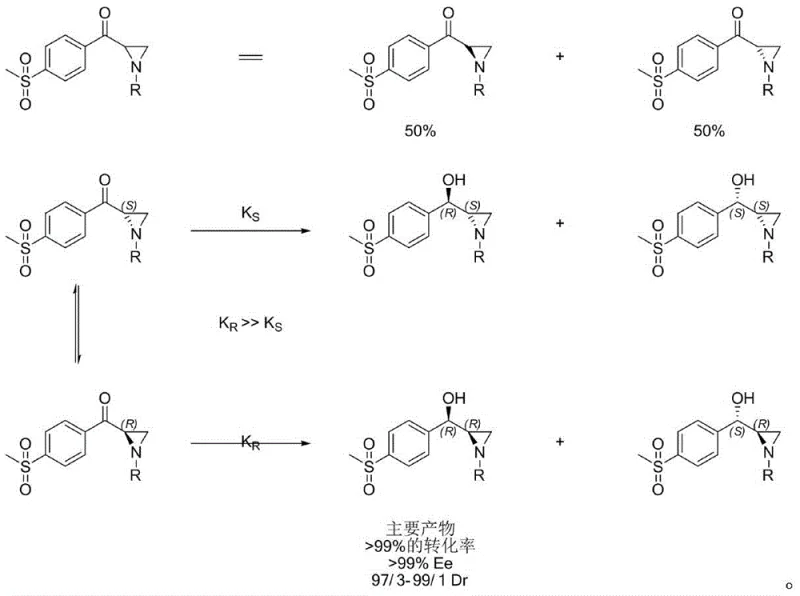
From an impurity control perspective, this biocatalytic route offers distinct advantages over chemical reduction. Chemical reducing agents like sodium borohydride or metal hydrides often lack the finesse to distinguish between subtle steric differences, leading to mixtures of diastereomers that require extensive chromatographic purification. In contrast, the enzyme acts as a molecular filter, rejecting the formation of the wrong stereoisomer at the transition state level. This "built-in" purification capability means that the crude product from the enzymatic step is of sufficiently high quality to proceed directly to the next stage (fluorination) without intermediate purification, further simplifying the process flow and reducing solvent waste. The resulting intermediate possesses a clean impurity profile, facilitating easier regulatory approval and consistent batch-to-batch quality.
How to Synthesize Florfenicol Intermediates Efficiently
Implementing this synthesis requires a coordinated sequence of biocatalytic and chemical steps designed to maximize throughput and purity. The process begins with the preparation of the aziridine ketone substrate, which is then subjected to the enzymatic reduction in a buffered aqueous system containing the specific ketoreductase and a cofactor regeneration system. Following the biotransformation, the chiral alcohol intermediate undergoes ring-opening fluorination using triethylamine hydrofluoride, a step that installs the critical fluorine atom while opening the aziridine ring. Depending on the protecting group used, a final deprotection and acylation step yields the target florfenicol molecule.
- Perform enzymatic dynamic kinetic resolution on aziridine ketone substrates using specific ketoreductases (e.g., YH2068, YH2077) with cofactor regeneration to obtain chiral alcohols with >99% ee.
- Execute ring-opening fluorination of the chiral aziridine alcohol intermediate using triethylamine hydrofluoride (Et3N·3HF) under reflux conditions to install the fluorine atom.
- Complete the synthesis through optional deprotection and dichloroacetylation steps to yield the final florfenicol API or its immediate amino precursor.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this enzymatic technology translates into tangible strategic benefits that extend beyond simple chemistry. The primary advantage is the drastic simplification of the supply chain for raw materials. By eliminating the need for chiral resolving agents and heavy metal salts like copper sulfate, manufacturers can reduce their dependency on volatile commodity markets for these specific reagents. Moreover, the removal of the 50% waste stream inherent in classical resolution means that the effective consumption of the starting aziridine ketone is halved for the same output of product. This fundamental improvement in atom economy directly correlates to a substantial cost savings in raw material procurement, allowing for more competitive pricing in the final API market.
- Cost Reduction in Manufacturing: The elimination of the chiral resolution step effectively doubles the yield from the starting material, which is a massive driver for cost efficiency. Additionally, the process operates at ambient pressure and moderate temperatures (35°C), removing the need for expensive high-pressure hydrogenation reactors or cryogenic cooling systems required by other asymmetric synthesis methods. The avoidance of corrosive reagents like the Ishikawa reagent in favor of milder fluorination conditions also extends the lifespan of production equipment, reducing capital expenditure on maintenance and reactor replacement. These factors combine to lower the overall cost of goods sold (COGS) significantly.
- Enhanced Supply Chain Reliability: Biocatalytic processes are generally more robust and easier to scale than complex multi-step chemical syntheses involving sensitive organometallic catalysts. The enzymes used (e.g., YH2068) are produced via fermentation, a scalable and renewable source that is less susceptible to the geopolitical supply risks associated with mined rare earth metals or specialized chemical ligands. The aqueous nature of the reaction medium also simplifies logistics, as it reduces the volume of flammable organic solvents that need to be transported and stored on-site. This reliability ensures a steady flow of high-purity veterinary drug intermediates to downstream formulation partners, minimizing the risk of production stoppages.
- Scalability and Environmental Compliance: As environmental regulations tighten globally, the ability to demonstrate a "green" manufacturing process is a significant commercial asset. This method generates no heavy metal wastewater and significantly reduces the load of organic solvents, simplifying effluent treatment and lowering compliance costs. The process is inherently safer, operating without high pressures or pyrophoric reagents, which reduces insurance premiums and operational risks. This environmental and safety profile makes the technology highly scalable, capable of meeting the demands of commercial scale-up of complex chiral intermediates from pilot plant to multi-ton production without encountering the bottlenecks typical of traditional chemical routes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this enzymatic synthesis route. These insights are derived directly from the experimental data and process descriptions within the patent literature, providing a clear picture of the technology's capabilities and limitations for potential adopters.
Q: How does the enzymatic DKR method improve yield compared to traditional chemical resolution?
A: Traditional chemical resolution typically wastes 50% of the unwanted enantiomer. The enzymatic dynamic kinetic resolution (DKR) described in CN111500652A allows for the conversion of the racemic substrate into a single chiral product theoretically up to 100% yield, significantly improving atom economy and reducing raw material costs.
Q: What are the environmental benefits of this biocatalytic route?
A: This method eliminates the need for heavy metal catalysts like copper sulfate used in classical amino acid complexation, thereby removing toxic heavy metal wastewater from the effluent stream. Additionally, it operates in aqueous buffer systems at mild temperatures (35°C), reducing energy consumption and hazardous solvent usage.
Q: Can this process be scaled for industrial production of veterinary antibiotics?
A: Yes, the patent demonstrates the use of robust recombinant ketoreductases (e.g., YH2045, YH2068) that function effectively in phosphate buffer systems. The process avoids extreme conditions like high-pressure hydrogenation or cryogenic temperatures, making it highly suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Florfenicol Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the enzymatic dynamic kinetic resolution technology described in CN111500652A for the veterinary pharmaceutical industry. As a leading CDMO partner, we possess the technical expertise to adapt and optimize this biocatalytic route for your specific production needs. Our facilities are equipped with state-of-the-art fermentation capabilities for enzyme production and versatile reactor suites capable of handling both aqueous biotransformations and subsequent chemical modifications. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale innovation to full-scale manufacturing is seamless. Our stringent purity specifications and rigorous QC labs guarantee that every batch of florfenicol intermediate meets the highest global regulatory standards.
We invite you to collaborate with us to leverage this advanced synthesis method for your supply chain. By partnering with our technical team, you can access a Customized Cost-Saving Analysis tailored to your current production volumes and identify specific areas where this enzymatic route can reduce your operational expenditures. We encourage you to contact our technical procurement team today to request specific COA data for our enzymatically derived intermediates and to discuss route feasibility assessments for your upcoming projects. Let us help you secure a sustainable and cost-effective supply of high-quality veterinary antibiotic intermediates.
