Advanced Florfenicol Manufacturing: Overcoming Ishikawa Reagent Limitations with Sulfuryl Fluoride Technology
The global demand for broad-spectrum antibiotics in the veterinary sector continues to surge, placing immense pressure on manufacturers to optimize the production of critical active pharmaceutical ingredients (APIs) like florfenicol. Patent CN111978218A, published in late 2020, introduces a transformative synthetic methodology that addresses long-standing inefficiencies in florfenicol manufacturing. This innovation pivots away from the traditionally expensive and hazardous Ishikawa reagent fluorination, utilizing instead sulfuryl fluoride ($SO_2F_2$) as a highly efficient fluorinating agent. For R&D directors and procurement strategists, this patent represents a pivotal shift towards greener chemistry, offering a pathway to significantly lower production costs while enhancing supply chain resilience. The method simplifies the operational workflow by eliminating complex purification steps between fluorination and hydrolysis, thereby streamlining the entire production lifecycle for this essential veterinary drug.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of florfenicol has relied heavily on the Ishikawa reagent (N,N-diethyl-1,1,2,3,3,3-hexafluoropropylamine) for the critical fluorination step. While effective, this legacy approach suffers from severe economic and environmental drawbacks that hinder scalable manufacturing. The Ishikawa reagent is notoriously expensive, accounting for approximately 15% of total material costs, and requires usage in large excess (1.5 times theoretical amount) to drive the reaction, resulting in a dismal fluorine atom utilization rate of only one-sixth. Furthermore, the reaction generates stoichiometric amounts of N,N-diethyl-2,3,3,3-tetrafluoropropionamide, a stable byproduct that is difficult to recycle and poses significant challenges for wastewater treatment due to its toxicity to biological treatment systems. The synthesis of the reagent itself demands harsh conditions involving ultra-low temperatures and high pressures, compounding energy consumption and safety risks for plant operators.
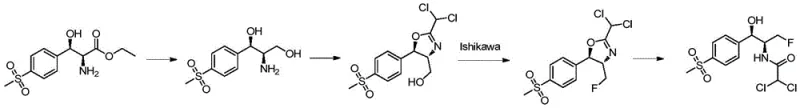
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent leverages sulfuryl fluoride, a widely available and inexpensive fumigant gas, to achieve superior fluorination efficiency. This new route dramatically improves atom economy, boosting fluorine utilization rates to approximately 50%, which fundamentally alters the cost structure of the synthesis. The process operates under much milder conditions, typically between -15°C and 30°C, removing the need for energy-intensive cryogenic setups. Crucially, the reaction mixture containing the fluorinated intermediate (Compound II) can be concentrated and directly subjected to the subsequent ring-opening hydrolysis without intermediate isolation or purification. This telescoping of steps not only reduces solvent consumption and processing time but also minimizes product loss, leading to higher overall yields and a more robust manufacturing process suitable for large-scale industrial application.
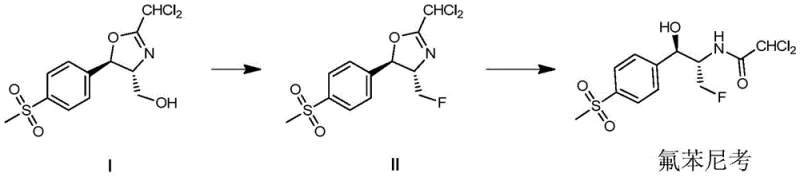
Mechanistic Insights into Sulfuryl Fluoride-Mediated Fluorination
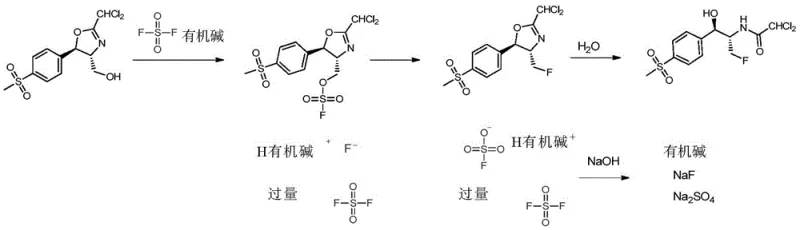
The core chemical innovation lies in the nucleophilic substitution facilitated by sulfuryl fluoride in the presence of an organic base such as triethylamine or diisopropylethylamine. In the first stage, the hydroxyl group of the precursor oxazoline (Compound I) is activated and displaced by fluoride derived from the decomposition of the sulfuryl fluoride-base complex. This reaction proceeds efficiently in inert solvents like acetonitrile or dichloromethane, with the system pressure maintained between 1 to 3 atmospheres to ensure adequate reagent concentration. The resulting fluorinated oxazoline (Compound II) retains the stereochemical integrity required for the biological activity of the final API. Unlike traditional methods that might require strict anhydrous conditions throughout, this process tolerates the subsequent transition to an aqueous environment, showcasing the stability and versatility of the intermediate formed.
Following fluorination, the mechanism shifts to a hydrolytic ring-opening in a mixed solvent system of water and lower alcohols (C1-6 alkyl alcohols). Heating the mixture to temperatures between 60°C and 100°C facilitates the cleavage of the oxazoline ring, unveiling the primary amine and secondary alcohol functionalities characteristic of florfenicol. A distinct advantage of this mechanistic pathway is the simplicity of impurity management; the byproducts generated are primarily inorganic salts and easily separable organic bases. The patent details a specific waste treatment protocol where residual organic fluorine compounds in the mother liquor are converted into inorganic fluoride ions via alkaline hydrolysis, ensuring that the final effluent meets stringent environmental discharge standards with fluorine content below 10 ppm.
How to Synthesize Florfenicol Efficiently
The synthesis protocol outlined in the patent provides a clear, reproducible framework for producing high-purity florfenicol suitable for veterinary use. The process begins with the fluorination of the serine-derived oxazoline precursor using sulfuryl fluoride gas in the presence of a tertiary amine base. Following the completion of the fluorination, indicated by HPLC analysis, the reaction mixture is concentrated to remove volatile components. The resulting concentrate is then directly dissolved in an aqueous alcohol solution and heated to effect ring-opening hydrolysis. The crude product precipitates upon cooling and is subsequently purified via recrystallization from an aqueous alcohol system to meet pharmacopoeial specifications. For detailed operational parameters and specific stoichiometric ratios, please refer to the standardized synthesis guide below.
- Conduct fluorination of Compound I with sulfuryl fluoride and an organic base in an inert solvent at -15 to 30°C to form Compound II.
- Concentrate the reaction mixture and subject it directly to ring-opening hydrolysis in an aqueous alcohol system at 60 to 100°C.
- Cool the mixture, filter the solid product, and recrystallize from an aqueous alcohol system to obtain purified florfenicol meeting pharmacopoeia standards.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this sulfuryl fluoride-based technology offers compelling strategic advantages that extend beyond mere chemical efficiency. The primary benefit is a drastic reduction in raw material costs, driven by the substitution of the high-value Ishikawa reagent with commodity-grade sulfuryl fluoride. This shift not only lowers the direct cost of goods sold (COGS) but also mitigates the risk of supply disruptions associated with specialized fluorinating agents. Additionally, the simplified process flow, which eliminates intermediate isolation steps, reduces the requirement for processing equipment and labor hours, further enhancing the economic viability of large-scale production campaigns.
- Cost Reduction in Manufacturing: The elimination of expensive fluorinating reagents and the reduction in solvent usage due to telescoped steps lead to substantial cost savings. By avoiding the need for complex purification between the fluorination and hydrolysis stages, manufacturers can significantly reduce energy consumption and waste disposal fees. The high atom economy of the sulfur fluoride reagent ensures that less raw material is wasted, optimizing the input-to-output ratio and improving overall margin potential for the final API.
- Enhanced Supply Chain Reliability: Sulfuryl fluoride is a commercially abundant chemical with a stable supply chain, unlike specialized fluorinating reagents that may be sourced from limited suppliers. This availability ensures consistent production scheduling and reduces the risk of bottlenecks caused by raw material shortages. Furthermore, the milder reaction conditions reduce the wear and tear on reactor vessels and ancillary equipment, leading to higher asset utilization rates and fewer unplanned maintenance downtimes.
- Scalability and Environmental Compliance: The process is inherently safer and more environmentally friendly, as it avoids the generation of persistent fluorinated organic waste. The ability to treat wastewater effectively using standard alkaline and lime treatments allows facilities to meet rigorous environmental discharge standards without investing in exotic waste treatment infrastructure. This compliance capability is critical for maintaining operating licenses and sustaining long-term production capacity in increasingly regulated markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this novel synthesis route. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, providing a reliable basis for feasibility assessments and technology transfer planning.
Q: Why is sulfuryl fluoride preferred over Ishikawa reagent for florfenicol synthesis?
A: Sulfuryl fluoride offers significantly higher fluorine atom utilization (up to 50% vs 16%) and eliminates the generation of toxic, non-recyclable fluorinated amide byproducts associated with Ishikawa reagents, drastically reducing waste treatment costs.
Q: How does the new process handle fluorine-containing wastewater?
A: The process employs a simple post-treatment where waste liquid is refluxed with sodium hydroxide to convert organic fluorine into inorganic fluoride ions, followed by lime water treatment to ensure discharge standards of ≤10 ppm fluorine content are met.
Q: What are the typical reaction conditions for the fluorination step?
A: The fluorination is conducted in inert solvents like acetonitrile or dichloromethane at mild temperatures ranging from -15°C to 30°C under slight pressure (1-3 atm), avoiding the extreme cryogenic conditions required by older methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Florfenicol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global veterinary pharmaceutical market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the sulfuryl fluoride fluorination process can be seamlessly translated from the laboratory to full-scale manufacturing. We are committed to delivering high-purity intermediates and APIs that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic impact of switching to this greener technology. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your production needs, ensuring a reliable and cost-effective supply of high-quality florfenicol for your operations.
