Revolutionizing Chiral Epoxide Synthesis with Stable Salen Catalysts for Commercial Scale-Up
The landscape of chiral synthesis is undergoing a significant transformation driven by the innovations disclosed in patent CN1463272A, which introduces a novel class of chiral salen catalysts designed to overcome the persistent challenges of racemization and catalyst deactivation. This technology represents a pivotal advancement for the production of high-value chiral epoxides and 1,2-diols, which serve as critical building blocks in the manufacture of chiral pharmaceuticals and food additives. Traditional methods have long struggled with maintaining optical purity during large-scale processing, often necessitating costly purification steps or resulting in significant material loss. The core breakthrough lies in the specific molecular architecture of the new catalyst, which incorporates a non-nucleophilic activating group that stabilizes the chiral center against degradation. By addressing the fundamental chemical instability that plagues conventional systems, this invention offers a pathway to more economical and reliable production of optically pure compounds. For industry leaders seeking a reliable pharmaceutical intermediate supplier, understanding the mechanistic advantages of this technology is essential for optimizing supply chain resilience and product quality.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of chiral epoxides via stereoselective hydrolysis has been hindered by the inherent instability of traditional chiral salen catalysts, particularly those utilizing nucleophilic counterions such as acetate or halide groups. These conventional catalysts suffer from a critical flaw where the counterion can dissociate or participate in reverse reactions with the hydrolysis products, leading to gradual racemization of the desired chiral epoxide over time. This phenomenon is especially detrimental during downstream processing steps like distillation, where prolonged exposure to heat and the catalyst residue can significantly degrade optical purity, rendering the batch unsuitable for high-grade pharmaceutical applications. Furthermore, the catalytic activity of these traditional systems tends to diminish rapidly after a single use, necessitating complex and energy-intensive activation procedures involving acetic acid treatment to restore functionality. This requirement not only increases operational costs but also introduces additional variables that can compromise batch-to-batch consistency and overall process efficiency in a commercial manufacturing environment.
The Novel Approach
In stark contrast to legacy technologies, the novel approach detailed in the patent utilizes a unique sandwich-type configuration where two molecules of chiral salen ligand coordinate with a central activating group such as boron trifluoride (BF3) or aluminum chloride (AlCl3). This specific structural arrangement effectively eliminates the presence of nucleophilic counterions that are responsible for inducing racemization in conventional systems. By employing a non-nucleophilic activating group, the new catalyst ensures that the chiral epoxide product remains stable even during extended reaction times and subsequent purification processes. The robustness of this design allows the catalyst to retain its high stereoselectivity and activity over multiple consecutive cycles without requiring any regeneration or activation steps. This paradigm shift simplifies the manufacturing workflow drastically, removing the need for intermediate treatment stages and enabling a more continuous and streamlined production process that is ideally suited for the commercial scale-up of complex pharmaceutical intermediates.
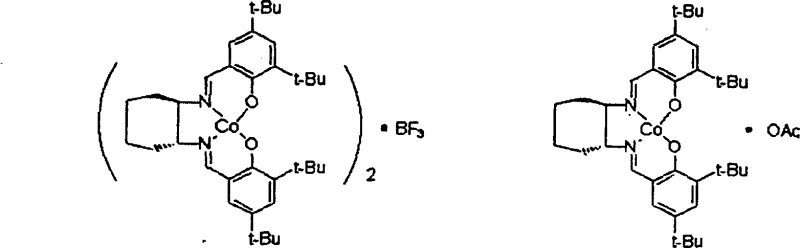
Mechanistic Insights into BF3-Activated Stereoselective Hydrolysis
The superior performance of this new catalyst system is rooted in its distinct electronic and steric properties, which fundamentally alter the reaction pathway compared to standard metallo-salen complexes. The central boron or aluminum atom acts as a Lewis acid bridge between the two salen ligand units, creating a rigid sandwich structure that protects the active metal center from deactivation. Crucially, the absence of nucleophilic groups means there is no species available to attack the chiral epoxide product and induce ring-opening or isomerization reactions that lead to racemization. This mechanistic stability is vital for maintaining the stringent optical purity specifications required by regulatory bodies for active pharmaceutical ingredients. The kinetic data suggests that the reaction proceeds through both intramolecular and intermolecular pathways, with the sandwich configuration facilitating a highly organized transition state that favors the selective hydrolysis of one enantiomer over the other. This level of control ensures that the unhydrolyzed epoxide remaining in the reaction medium retains its high optical integrity, maximizing the yield of the desired chiral building block.
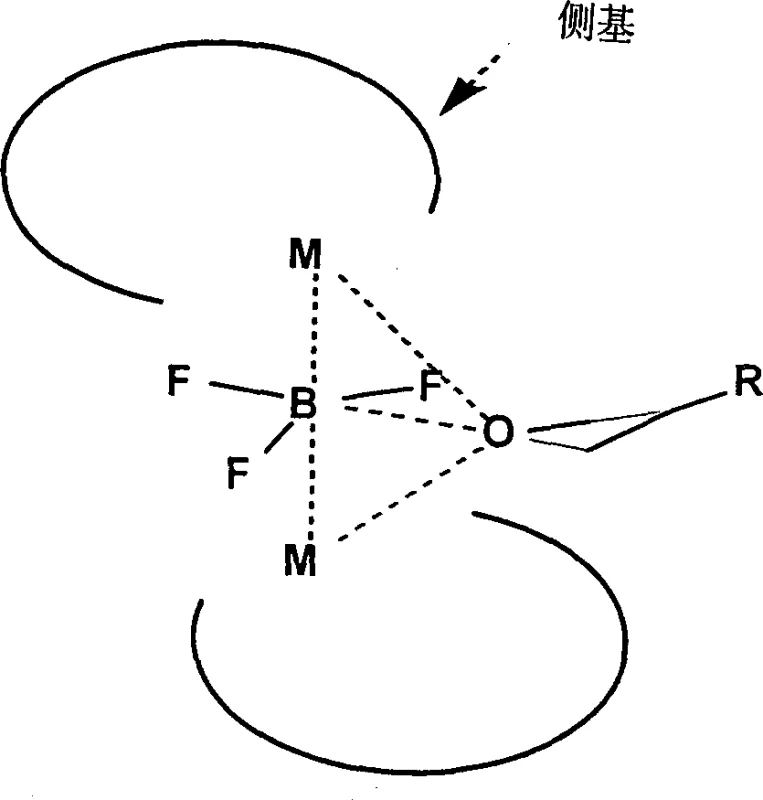
Furthermore, the impurity profile generated by this process is significantly cleaner due to the suppression of side reactions associated with counterion dissociation. In traditional systems, free acetate or halide ions can catalyze unwanted decomposition pathways, leading to complex impurity spectra that are difficult and expensive to remove. The new catalyst minimizes these side reactions, resulting in a crude product that requires less intensive purification. This reduction in downstream processing burden translates directly into improved process mass intensity and reduced solvent consumption. For R&D teams focused on process chemistry, this implies a more robust method that is less sensitive to minor fluctuations in reaction conditions. The ability to consistently achieve optical purity levels exceeding 99% ee without extensive recrystallization or chromatographic separation underscores the practical value of this mechanistic innovation for industrial applications.
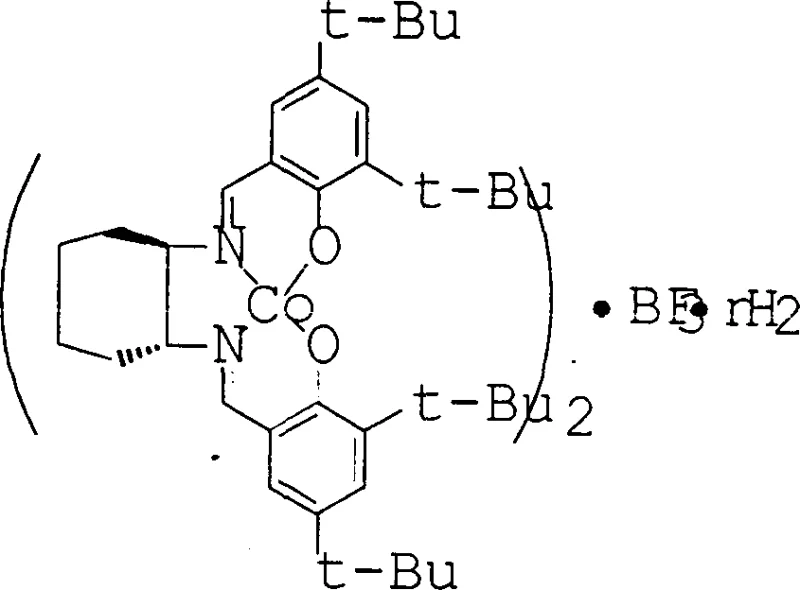
How to Synthesize Chiral Salen Catalyst Efficiently
The synthesis of this advanced catalyst is designed to be straightforward and scalable, utilizing readily available starting materials and standard laboratory equipment to facilitate easy technology transfer. The process begins with the formation of the metal-salen intermediate through the condensation of chiral diamines with salicylaldehyde derivatives followed by metallation with cobalt or manganese salts. This intermediate is then treated with the activating Lewis acid source, such as boron trifluoride dihydrate, in a suitable organic solvent to assemble the final sandwich structure. The simplicity of this two-step sequence avoids the need for exotic reagents or extreme reaction conditions, making it accessible for widespread adoption. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during implementation.
- React chiral diamine ligands with metal acetates in ethanol under reflux to form the intermediate metal-salen complex.
- Filter and wash the solid intermediate compound to remove unreacted starting materials and byproducts thoroughly.
- Treat the intermediate with a non-nucleophilic activating group like BF3 in an organic solvent to finalize the stable catalyst structure.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this catalyst technology offers substantial opportunities for cost reduction in pharmaceutical intermediates manufacturing by eliminating several cost-driving factors inherent in traditional processes. The primary economic benefit stems from the catalyst's ability to be reused continuously without activation, which removes the recurring expense of regenerating agents and the labor costs associated with additional processing steps. This durability significantly extends the effective lifespan of the catalyst inventory, reducing the frequency of replenishment orders and mitigating supply risks associated with specialized reagent availability. Moreover, the enhanced stability of the product reduces the likelihood of batch failures due to off-specification optical purity, thereby minimizing waste disposal costs and maximizing the return on raw material investments. These factors combine to create a more predictable and efficient cost structure for long-term production campaigns.
- Cost Reduction in Manufacturing: The elimination of the catalyst activation step represents a direct saving in both utility consumption and chemical usage, as there is no longer a need for acetic acid treatments or solvent exchanges to regenerate the catalyst. Additionally, the higher selectivity of the process reduces the burden on purification units, leading to lower energy consumption for distillation and less solvent waste generation. Over the lifecycle of a production campaign, these operational efficiencies accumulate to provide significant cost savings without compromising product quality. The simplified workflow also reduces the man-hours required for batch execution, allowing technical staff to focus on value-added activities rather than routine maintenance tasks.
- Enhanced Supply Chain Reliability: The robustness of the catalyst contributes to greater supply chain reliability by ensuring consistent production throughput and reducing the risk of unplanned downtime caused by catalyst failure. Since the catalyst does not require complex activation, the lead time for turning around production vessels is shortened, allowing for faster response to market demand fluctuations. This agility is crucial for maintaining service levels in the fast-paced pharmaceutical sector where delays can have cascading effects on downstream drug formulation schedules. Furthermore, the use of stable and commercially available activating groups like BF3 reduces dependency on niche supply chains, enhancing the overall resilience of the manufacturing network against external disruptions.
- Scalability and Environmental Compliance: The process is inherently scalable, as demonstrated by successful large-scale experiments producing hundreds of kilograms of product with consistent quality, indicating readiness for multi-ton annual commercial production. The reduction in solvent usage and waste generation aligns with increasingly stringent environmental regulations, helping companies meet their sustainability goals and reduce their carbon footprint. By minimizing the release of hazardous byproducts and lowering energy intensity, this technology supports a greener manufacturing profile that is increasingly valued by global partners. This environmental advantage also simplifies regulatory compliance and permitting processes for facility expansions or new production lines.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalyst technology, derived directly from the patent specifications and experimental data. These insights are intended to clarify the operational benefits and technical feasibility for potential adopters evaluating this solution for their supply chain. Understanding these details is key to making informed decisions about process optimization and vendor selection.
Q: How does this catalyst prevent racemization during purification?
A: Unlike traditional catalysts with nucleophilic counterions like acetate, this novel structure uses non-nucleophilic groups such as BF3, preventing reverse reactions that cause optical purity loss during distillation.
Q: Can the catalyst be reused without activation?
A: Yes, the patent data confirms the catalyst maintains activity over multiple cycles without requiring the energy-intensive activation steps necessary for conventional salen catalysts.
Q: What optical purity levels are achievable with this method?
A: Experimental data demonstrates consistent optical purity exceeding 99% ee even after multiple reuse cycles, meeting stringent requirements for pharmaceutical raw materials.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Salen Catalyst Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating this patented technology into commercial reality, offering our partners access to high-performance chiral salen catalysts produced under rigorous quality standards. As a dedicated CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. Our facilities are equipped with stringent purity specifications and rigorous QC labs to guarantee that every batch of catalyst meets the exacting requirements for pharmaceutical intermediate synthesis. We understand the critical nature of chirality in drug development and are committed to delivering materials that support your regulatory filings and clinical timelines with unwavering consistency.
We invite you to engage with our technical procurement team to discuss how this innovative catalyst can optimize your specific production needs and drive value across your organization. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the potential economic impact on your operations. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your target molecules. Let us collaborate to enhance your supply chain efficiency and secure a competitive advantage in the global market through advanced catalytic solutions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
