Revolutionizing Chiral Epoxide Production with Advanced Salen Catalyst Technology
The chemical manufacturing landscape for high-value chiral intermediates is undergoing a significant transformation driven by the innovations disclosed in patent CN1233648C. This pivotal intellectual property introduces a novel class of chiral salen catalysts designed specifically to overcome the longstanding limitations of stereoselective hydrolysis in racemic epoxides. For R&D Directors and Procurement Managers seeking a reliable chiral epoxide supplier, this technology represents a paradigm shift towards more economical and scalable production methods. The core breakthrough lies in the specific molecular structure of the catalyst, which incorporates a non-nucleophilic activating group such as boron trifluoride. This structural modification effectively eliminates the racemization issues that have historically plagued the mass production of chiral drugs and food additives. By enabling the continuous reuse of the catalyst without the need for complex reactivation processes, this invention offers a robust solution for the commercial scale-up of complex pharmaceutical intermediates. The implications for supply chain stability and cost reduction in pharmaceutical intermediate manufacturing are profound, as it simplifies the workflow and enhances the overall yield of high-optical-purity compounds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for preparing chiral epoxides and 1,2-glycols have long been hindered by the inherent instability of conventional chiral salen catalysts. Historically, catalysts containing nucleophilic counter ions such as aceticoceptors or halides have been the standard, yet they suffer from critical defects when applied to industrial-scale operations. The primary issue is the tendency of these traditional catalysts to induce racemization of the chiral product over time, particularly during the purification and distillation processes required to isolate the final compound. This racemization significantly degrades the optical purity, often rendering the product unsuitable for stringent pharmaceutical applications where enantiomeric excess is paramount. Furthermore, conventional catalysts typically experience a rapid decline in catalytic activity after a single use, necessitating a cumbersome and costly reactivation step involving acetates or other chemicals before they can be employed again. This requirement not only increases the operational complexity but also extends the production lead time for high-purity fine chemicals, creating bottlenecks in the supply chain. The cumulative effect of these limitations is a manufacturing process that is both economically inefficient and technically risky for producing sensitive chiral intermediates.
The Novel Approach
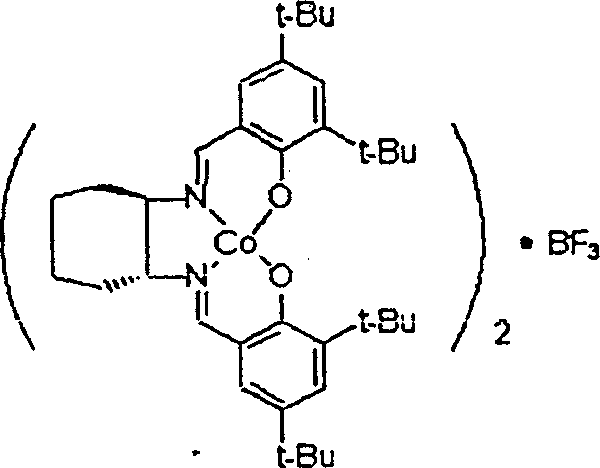
In stark contrast to the deficiencies of legacy systems, the novel approach detailed in the patent data utilizes a specifically engineered chiral salen catalyst that maintains its structural integrity and catalytic activity throughout multiple reaction cycles. By replacing the problematic nucleophilic counter ions with a non-nucleophilic group like boron trifluoride, the new catalyst effectively prevents the reverse reaction that leads to racemization. This ensures that the optical purity of the chiral epoxide or glycol remains exceptionally high, consistently exceeding the thresholds required for medicinal raw materials. The ability to reuse the catalyst continuously without any activation process is a game-changer for cost reduction in electronic chemical manufacturing and related sectors. It streamlines the production workflow by removing the downtime associated with catalyst regeneration and reduces the consumption of auxiliary chemicals. This technological advancement allows manufacturers to achieve a level of process efficiency and product consistency that was previously unattainable, making it an ideal solution for partners seeking a reliable agrochemical intermediate supplier or pharma partner.
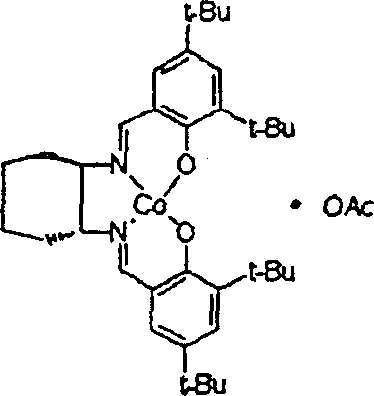
Mechanistic Insights into BF3-Modified Salen Catalyzed Hydrolysis
The superior performance of this novel catalyst system can be attributed to its unique mechanistic pathway, which fundamentally alters the interaction between the catalyst and the reaction substrates. The catalyst features a sandwich configuration where the activating group is positioned centrally, stabilizing the chiral salen part and preventing the dissociation that leads to activity loss. Unlike traditional catalysts where weak bonds between the counter ion and the central metal allow for nucleophilic attacks that cause racemization, the BF3-modified structure creates a robust barrier against such side reactions. This structural stability ensures that the stereoselective hydrolysis proceeds with high fidelity, selectively hydrolyzing one isomer of the racemic epoxide while leaving the other intact with minimal optical degradation. The kinetic data suggests that the reaction follows a path that minimizes intermolecular interference, further enhancing the selectivity and speed of the transformation. For technical teams, understanding this mechanism is crucial as it validates the feasibility of scaling this process without compromising on the stringent purity specifications required for regulatory compliance in the pharmaceutical industry.
Furthermore, the impurity control mechanism inherent in this catalytic system provides an additional layer of assurance for quality assurance teams. The prevention of racemization means that the formation of undesired optical stereoisomers is drastically reduced, simplifying the downstream purification process. In traditional methods, removing these isomers often requires complex chromatography or repeated crystallization, which drives up costs and waste. With the novel catalyst, the reaction mixture yields a product with such high optical purity that the need for extensive purification is significantly diminished. This not only improves the overall yield but also aligns with green chemistry principles by reducing solvent usage and waste generation. The robustness of the catalyst against deactivation also means that metal leaching into the product is minimized, addressing another critical quality concern for pharmaceutical intermediates. This comprehensive control over the reaction environment ensures that the final product meets the rigorous standards expected by global regulatory bodies.
How to Synthesize Chiral Salen Catalyst Efficiently
The synthesis of this advanced catalyst is designed to be straightforward and adaptable to existing manufacturing infrastructure, facilitating rapid adoption by production teams. The process involves the reaction of a specific chiral salen ligand with a metal salt followed by the introduction of the activating group in a controlled solvent environment. This method ensures that the final catalyst possesses the precise stoichiometry required for optimal performance in stereoselective hydrolysis. The detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating the results.
- Prepare the novel chiral salen catalyst by reacting the salen ligand with cobalt acetate and boron trifluoride in a suitable organic solvent.
- Mix the racemic epoxide substrate with the catalyst and water at controlled low temperatures to initiate stereoselective hydrolysis.
- Separate the unhydrolyzed chiral epoxide from the reaction mixture and recover the catalyst for direct reuse without activation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel catalyst technology translates into tangible strategic advantages that go beyond mere technical specifications. The elimination of the catalyst reactivation step represents a significant reduction in operational complexity and resource consumption. By removing the need for additional chemicals and processing time to regenerate the catalyst, manufacturers can achieve substantial cost savings in the overall production budget. This efficiency gain is particularly valuable in the context of volatile raw material markets, where minimizing auxiliary consumption can provide a competitive edge. Additionally, the ability to reuse the catalyst for multiple cycles without loss of activity enhances the predictability of production schedules. This reliability is critical for maintaining supply continuity for high-purity pharmaceutical intermediates, ensuring that downstream customers receive their orders on time without unexpected delays caused by catalyst failure or regeneration bottlenecks.
- Cost Reduction in Manufacturing: The economic benefits of this technology are driven by the fundamental simplification of the catalytic cycle. By utilizing a catalyst that does not require reactivation, the process eliminates the costs associated with purchasing activation agents and the labor required for the regeneration procedure. Furthermore, the high selectivity of the reaction reduces the burden on downstream purification units, leading to lower energy consumption and solvent usage. The extended lifespan of the catalyst means that the frequency of catalyst replacement is drastically reduced, lowering the overall material cost per kilogram of product. These factors combine to create a more lean and cost-effective manufacturing process that improves the margin profile for specialty chemical production.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the robust nature of this catalytic system. The catalyst's ability to retain activity over multiple uses reduces the dependency on frequent catalyst shipments and inventory management. This stability allows for longer production runs without interruption, ensuring a steady flow of products to the market. For global buyers, this means a more dependable source of supply that can withstand fluctuations in demand. The reduced risk of batch failure due to catalyst deactivation further secures the supply chain, minimizing the potential for costly production stoppages and ensuring that critical timelines for drug development or agrochemical production are met consistently.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this technology offers a cleaner and more sustainable pathway for chemical synthesis. The reduction in waste generation from catalyst regeneration and the minimization of by-products align with increasingly strict environmental regulations. This makes the process easier to scale up from pilot plants to commercial production without encountering significant regulatory hurdles. The simplified waste profile also reduces the costs associated with waste treatment and disposal. For companies aiming to improve their sustainability metrics, adopting this catalytic method demonstrates a commitment to green chemistry principles while simultaneously achieving commercial scale-up of complex fine chemicals with greater efficiency.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel chiral salen catalyst technology. These answers are derived directly from the patent data and experimental results to provide clarity on the operational capabilities and limitations of the system. Understanding these details is essential for making informed decisions about integrating this technology into existing production lines.
Q: How does the novel BF3-modified catalyst prevent racemization compared to traditional OAc catalysts?
A: The novel catalyst utilizes a non-nucleophilic BF3 counter-ion which prevents the reverse reaction of the hydrolysate that typically causes racemization in traditional aceticoceptor-based catalysts.
Q: Is the catalyst reusable without reactivation steps?
A: Yes, the patent data confirms that the catalyst retains its activity after reaction and can be continuously reused for multiple cycles without requiring any activation process.
Q: What optical purity levels can be achieved with this technology?
A: Experimental data indicates that optical purity exceeding 99% ee can be consistently achieved for various chiral epoxides and glycols using this method.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Salen Catalyst Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the technologies described in patent CN1233648C and are well-positioned to support your transition to this advanced manufacturing paradigm. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this catalyst are realized in practical, large-scale operations. Our facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of chiral intermediate produced meets the highest industry standards. We understand the critical nature of optical purity in pharmaceutical applications and have the technical expertise to optimize this catalytic process for your specific target molecules. Our commitment to quality and consistency makes us the ideal partner for companies looking to leverage this innovation for their supply chain.
We invite you to engage with our technical procurement team to explore how this technology can be tailored to your specific needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic impact this catalyst can have on your specific product line. We encourage potential partners to contact us for specific COA data and route feasibility assessments to validate the performance of this system against your current benchmarks. Our team is ready to provide the technical support and commercial flexibility required to make this transition smooth and profitable. Let us help you engineer a more efficient and reliable supply chain for your critical chiral intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
