Revolutionizing Chiral Amine Synthesis via Engineered Amine Dehydrogenase for Industrial Scale-Up
The landscape of chiral amine synthesis is undergoing a profound transformation driven by the urgent need for sustainable, high-efficiency manufacturing processes in the pharmaceutical and fine chemical sectors. Patent CN110577941B, titled "Amine dehydrogenase and application thereof," represents a critical technological breakthrough in this domain, disclosing a novel class of engineered amine dehydrogenases derived from Bacillus nanhaiensis phenylalanine dehydrogenase. This intellectual property details specific site-directed mutations—such as G131L, N262V, Y285L, and M333D—that fundamentally alter the enzyme's active site architecture, enabling it to catalyze the asymmetric reductive amination of a broad spectrum of ketones with exceptional stereoselectivity. For R&D directors and process chemists, this patent offers a robust alternative to traditional chemical hydrogenation, providing a pathway to produce optically pure amines with yields often exceeding 90% and enantiomeric excess values that meet stringent regulatory standards for active pharmaceutical ingredients (APIs). The significance of this technology extends beyond mere academic interest; it provides a tangible solution for the industrial-scale production of chiral building blocks that are essential for the synthesis of complex drug molecules, thereby addressing the growing demand for reliable chiral amine suppliers who can deliver high-purity intermediates consistently.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of chiral amines has relied heavily on chemical methodologies such as high-pressure catalytic hydrogenation of imines or the resolution of racemic mixtures, both of which suffer from inherent inefficiencies and environmental drawbacks that modern supply chains can no longer tolerate. Traditional chemical hydrogenation often requires the use of expensive transition metal catalysts like rhodium or ruthenium, which not only drive up raw material costs but also introduce the risk of heavy metal contamination in the final product, necessitating costly and time-consuming purification steps to meet ppm-level specifications. Furthermore, these chemical processes frequently operate under extreme conditions involving high temperatures and pressures, which pose significant safety risks and require specialized, capital-intensive reactor infrastructure that limits flexibility in multi-purpose manufacturing facilities. Another critical limitation is the poor atom economy and stereoselectivity associated with many chemical routes, where the formation of unwanted enantiomers leads to a theoretical maximum yield of only 50% in resolution processes, resulting in substantial material waste and increased disposal costs that negatively impact the overall cost reduction in pharmaceutical intermediate manufacturing. These factors collectively create a bottleneck for procurement managers seeking to optimize supply chain reliability and reduce the carbon footprint of their production networks.
The Novel Approach
In stark contrast to these legacy methods, the novel biocatalytic approach disclosed in the patent leverages the power of protein engineering to create highly specific biocatalysts that operate under mild, ambient conditions with unparalleled precision. By introducing specific mutations at positions 131, 262, 285, and 333 of the amino acid dehydrogenase sequence, the inventors have successfully expanded the substrate scope of the enzyme to include various aliphatic and aryl ketones that were previously inaccessible to the wild-type enzyme. This engineered amine dehydrogenase facilitates the direct conversion of ketones into chiral amines using ammonia or simple amine donors in the presence of a cofactor like NADH, effectively bypassing the need for protecting groups and harsh reducing agents. The operational simplicity of this method is a major advantage, as reactions can be conducted in aqueous buffer systems at temperatures ranging from 10°C to 70°C and pH levels between 8 and 12, significantly lowering energy consumption and equipment maintenance requirements. Moreover, the high stereoselectivity inherent to the enzymatic mechanism ensures that the desired enantiomer is produced exclusively, eliminating the need for downstream resolution steps and dramatically improving the overall process mass intensity, which is a key metric for sustainable chemical manufacturing.

Mechanistic Insights into Amine Dehydrogenase-Catalyzed Reductive Amination
The core of this technological advancement lies in the sophisticated mechanistic pathway of asymmetric reductive amination, where the engineered amine dehydrogenase acts as a molecular machine to orchestrate the transfer of a hydride ion from the reduced nicotinamide adenine dinucleotide (NADH) cofactor to the prochiral ketone substrate. As illustrated in the reaction scheme, the process begins with the formation of an unstable imine intermediate through the condensation of the ketone carbonyl group with the ammonia donor, a step that is rapidly followed by the stereospecific reduction of the C=N double bond within the enzyme's chiral active site. The specific mutations introduced in the patent, such as the substitution of Glycine with Leucine at position 131, are believed to reshape the hydrophobic pocket of the enzyme, thereby enhancing the binding affinity for bulky aliphatic ketones and enforcing a rigid orientation that favors the formation of the R-configured amine product. This precise control over the transition state geometry is what allows the biocatalyst to achieve enantiomeric excess values often greater than 94%, a level of purity that is difficult to replicate with small-molecule catalysts without extensive ligand optimization. Furthermore, the regeneration of the NAD+ cofactor back to NADH is seamlessly integrated into the reaction cycle, often coupled with a secondary substrate like isopropanol or formate, ensuring that only catalytic amounts of the expensive cofactor are required to drive the reaction to completion.
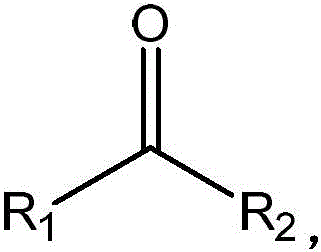
Understanding the substrate scope defined by the generic structure R1-C(=O)-R2 is crucial for applying this technology to diverse synthetic challenges, as the patent explicitly demonstrates activity against a wide range of substituents including C2-10 alkyl groups, aryl rings, and hydroxyl functionalities. The structural flexibility of the mutated enzyme allows it to accommodate variations in steric bulk and electronic properties at the R1 and R2 positions, making it a versatile tool for synthesizing structurally diverse chiral amines used in drug discovery and development. For instance, the successful conversion of 5-methyl-2-hexanone to the corresponding chiral amine with high yield and selectivity highlights the enzyme's capability to handle branched aliphatic chains, which are common motifs in bioactive molecules. This broad substrate tolerance reduces the need for custom enzyme development for every new target molecule, thereby accelerating the timeline from laboratory bench to commercial production and providing a scalable solution for the commercial scale-up of complex pharmaceutical intermediates. The ability to predictably modify the enzyme's performance through rational design further empowers process chemists to tailor the biocatalyst for specific kinetic requirements or solvent compatibilities.
How to Synthesize Chiral Amines Efficiently
The implementation of this biocatalytic route involves a series of well-defined steps starting from the construction of the recombinant expression strain to the final isolation of the chiral amine product, offering a clear roadmap for process adoption. The patent outlines a robust protocol involving PCR-based site-directed mutagenesis to introduce the desired amino acid substitutions, followed by transformation into E. coli host cells for high-level expression of the His-tagged enzyme. Once the biocatalyst is prepared, either as a crude cell lysate or a purified protein, it is employed in a reaction system containing the ketone substrate, an ammonia source such as ammonium chloride, and the necessary cofactor system buffered at an optimal pH. Detailed standardized synthesis steps see the guide below.
- Construct recombinant expression strains by introducing specific point mutations (e.g., G131L, N262V) into the phenylalanine dehydrogenase gene sequence via PCR.
- Cultivate the transformed E. coli strains in LB medium with IPTG induction to express the His-tagged amine dehydrogenase enzyme.
- Perform asymmetric reductive amination by mixing the crude or purified enzyme with ketone substrates, ammonia donors, and NADH cofactor under controlled pH and temperature.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this engineered amine dehydrogenase technology translates into tangible strategic benefits that extend far beyond simple reaction yields, fundamentally altering the cost structure and risk profile of chiral amine production. The elimination of precious metal catalysts removes a significant variable cost component and mitigates the supply risk associated with fluctuating prices of rare earth metals, while simultaneously simplifying the regulatory approval process by avoiding heavy metal residuals in the final API. The mild reaction conditions allow for the use of standard stainless steel reactors rather than specialized high-pressure vessels, reducing capital expenditure requirements and enabling existing manufacturing facilities to be repurposed for biocatalytic processes with minimal retrofitting investment. Furthermore, the aqueous nature of the reaction medium significantly reduces the volume of organic solvents required, leading to lower waste disposal costs and a smaller environmental footprint, which is increasingly becoming a decisive factor in vendor selection for multinational corporations committed to sustainability goals.
- Cost Reduction in Manufacturing: The biocatalytic process inherently drives down manufacturing costs by consolidating multiple synthetic steps into a single enzymatic transformation, thereby reducing labor hours, utility consumption, and raw material usage associated with multi-step chemical syntheses. By achieving high conversion rates and stereoselectivity in one pot, the need for costly chromatographic separations to remove unwanted enantiomers is largely eliminated, which represents a massive saving in both time and consumables for large-scale production runs. Additionally, the stability of the engineered enzyme under operational conditions allows for potential reuse or immobilization, further amortizing the cost of the biocatalyst over multiple batches and enhancing the overall economic viability of the process for high-volume commodity chemicals.
- Enhanced Supply Chain Reliability: Relying on fermentation-derived enzymes rather than petrochemical-based reagents diversifies the supply base and reduces dependency on volatile fossil fuel markets, ensuring a more stable and predictable supply of critical intermediates. The scalability of microbial fermentation means that production capacity can be rapidly ramped up to meet surges in demand without the long lead times typically associated with constructing new chemical synthesis plants, providing a agile response mechanism to market fluctuations. This biological manufacturing platform also offers greater geographic flexibility, as fermentation facilities can be established closer to key markets, reducing logistics costs and lead times for high-purity pharmaceutical intermediates delivered to global customers.
- Scalability and Environmental Compliance: The process aligns perfectly with green chemistry principles by utilizing renewable feedstocks and generating benign byproducts like water, which simplifies wastewater treatment and ensures compliance with increasingly stringent environmental regulations worldwide. The absence of toxic solvents and hazardous reagents minimizes the risk of workplace accidents and exposure, creating a safer working environment and reducing liability insurance costs for the manufacturing entity. From a lifecycle assessment perspective, the lower energy intensity and reduced waste generation of this enzymatic route contribute to a lower carbon footprint, helping corporate clients meet their Scope 3 emissions targets and enhancing the brand value of the final pharmaceutical products.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this amine dehydrogenase technology, drawing directly from the experimental data and beneficial effects described in the patent documentation. These insights are intended to clarify the operational parameters and potential applications for stakeholders evaluating this technology for integration into their existing manufacturing portfolios. Understanding these nuances is essential for making informed decisions about process development and vendor partnerships in the competitive landscape of chiral synthesis.
Q: What are the key advantages of using mutated amine dehydrogenase over wild-type enzymes?
A: The mutated amine dehydrogenase described in patent CN110577941B exhibits significantly improved catalytic activity and stereoselectivity towards non-natural aliphatic and aryl ketones compared to the wild-type phenylalanine dehydrogenase, which typically lacks activity for these substrates.
Q: How does this biocatalytic method impact environmental compliance in amine manufacturing?
A: By utilizing aqueous buffer systems and avoiding heavy metal catalysts or high-pressure hydrogenation, this enzymatic route drastically reduces hazardous waste generation and eliminates the need for complex heavy metal removal steps, aligning with green chemistry principles.
Q: Can this technology be scaled for commercial production of pharmaceutical intermediates?
A: Yes, the process utilizes standard fermentation and biocatalysis equipment with mild operating conditions (10-70°C, atmospheric pressure), making it highly suitable for scale-up from laboratory grams to multi-ton commercial production without significant infrastructure changes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Amine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the engineered amine dehydrogenase technology described in CN110577941B and are fully equipped to leverage these advancements for our global clientele. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory proof-of-concept to industrial reality is seamless and efficient. Our state-of-the-art facilities are designed to handle complex biocatalytic processes with stringent purity specifications, supported by rigorous QC labs that employ advanced analytical techniques to guarantee the identity, potency, and optical purity of every batch we produce. We understand that consistency is key in the pharmaceutical supply chain, and our robust quality management systems are aligned with international regulatory standards to provide you with peace of mind and uninterrupted supply continuity.
We invite you to collaborate with us to explore how this cutting-edge biocatalytic route can optimize your specific synthesis challenges and drive value across your organization. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your target molecule, demonstrating exactly how switching to this enzymatic method can improve your margins and sustainability metrics. Please contact our technical procurement team today to request specific COA data for similar chiral amine intermediates and to discuss detailed route feasibility assessments that will pave the way for your next successful product launch.
