Scalable Diaryl Ether Synthesis Using Amino Acid Ligands for Commercial Pharmaceutical Manufacturing
Scalable Diaryl Ether Synthesis Using Amino Acid Ligands for Commercial Pharmaceutical Manufacturing
The development of efficient and cost-effective synthetic routes for diaryl ethers is a critical priority for the global pharmaceutical and fine chemical industries, particularly given the prevalence of this structural motif in active pharmaceutical ingredients (APIs) and agrochemical intermediates. Patent CN1203040C introduces a transformative methodology that addresses long-standing challenges in Ullmann-type coupling reactions by utilizing amino acids as additives in conjunction with copper salt catalysts. This innovation allows for the synthesis of diaryl ethers under remarkably mild conditions, circumventing the need for the extreme temperatures and stoichiometric amounts of copper reagents that characterized historical protocols. By leveraging inexpensive and air-stable ligands such as N,N-dimethylglycine, this process not only enhances reaction yields but also simplifies downstream purification, making it an exceptionally attractive option for industrial scale-up. The technical breakthrough described in this patent provides a robust foundation for manufacturers seeking to optimize their supply chains for complex aromatic ethers while maintaining rigorous quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of diaryl ethers via the Ullmann reaction has been plagued by significant operational drawbacks that hinder its application in modern, high-throughput manufacturing environments. Traditional protocols typically necessitate harsh reaction conditions, often requiring temperatures well above 150°C and the use of stoichiometric or even excess quantities of copper reagents to drive the coupling to completion. These severe conditions frequently lead to the decomposition of sensitive functional groups, limiting the substrate scope and resulting in complex impurity profiles that are difficult and costly to remove. Furthermore, while palladium-catalyzed alternatives have emerged to mitigate some temperature issues, they introduce substantial economic burdens due to the high cost of palladium metals and the stringent requirements for removing trace heavy metal residues from final drug substances. The reliance on expensive ligands and inert atmosphere handling in many modern catalytic systems further exacerbates production costs and complicates logistics for large-scale operations.
The Novel Approach
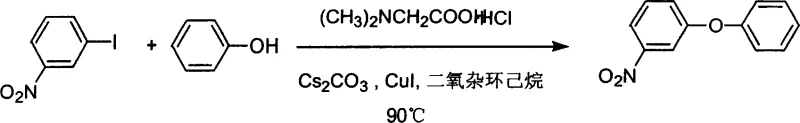
In stark contrast to these conventional limitations, the methodology disclosed in CN1203040C employs a catalytic system based on readily available copper salts and simple amino acid derivatives to achieve high-efficiency coupling under mild thermal conditions. The use of N-substituted alpha-amino acids or beta-amino acids, such as N,N-dimethylglycine, acts as a powerful bidentate ligand that stabilizes the copper center and facilitates the oxidative addition and reductive elimination steps essential for the catalytic cycle. This approach allows reactions to proceed effectively at temperatures around 90°C, which is approximately 30°C lower than many comparable literature methods, thereby preserving the integrity of thermally labile substituents. As demonstrated in the patent examples, this system exhibits remarkable versatility, successfully coupling aryl iodides and bromides with various phenols to produce diaryl ethers in high yields, often exceeding 90%, without the need for exotic or prohibitively expensive reagents.
Mechanistic Insights into Amino Acid-Promoted Copper Catalysis
The efficacy of this synthetic route lies in the unique coordination chemistry between the copper catalyst and the amino acid ligand, which creates a highly active catalytic species capable of overcoming the kinetic barriers of C-O bond formation. The amino acid ligand, typically featuring a nitrogen donor and a carboxylate or similar coordinating group, chelates the copper ion to form a stable complex that resists aggregation into inactive copper clusters, a common deactivation pathway in Ullmann reactions. This stabilization allows the use of catalytic loadings as low as 1 mol% to 20 mol% relative to the aryl halide, significantly reducing the metal burden in the reaction mixture. The mechanism likely proceeds through a Cu(I)/Cu(III) catalytic cycle where the ligand facilitates the oxidative addition of the aryl halide to the copper center, followed by coordination of the phenoxide anion generated by the base. The subsequent reductive elimination releases the diaryl ether product and regenerates the active Cu(I) species, ready for another turnover.

From an impurity control perspective, the mildness of this reaction conditions is paramount for ensuring high product purity, a critical metric for R&D directors overseeing API intermediate production. Lower reaction temperatures minimize side reactions such as homocoupling of the aryl halide or degradation of sensitive functional groups like nitro or cyano moieties, which are often present in bioactive molecules. The patent data indicates that even with electron-deficient substrates like m-nitroiodobenzene, the reaction proceeds smoothly to afford the desired ether in 98% yield, suggesting that the catalyst system is robust against electronic deactivation. Furthermore, the use of simple inorganic bases like cesium carbonate or potassium carbonate in polar aprotic solvents such as dioxane ensures that the reaction medium remains homogeneous and manageable, facilitating straightforward workup procedures that involve simple aqueous extraction and silica gel chromatography to isolate the pure product.
How to Synthesize Diaryl Ethers Efficiently
Implementing this amino acid-promoted copper catalysis protocol requires careful attention to reagent ratios and reaction parameters to maximize yield and reproducibility on a commercial scale. The general procedure involves charging a reaction vessel with the aryl halide substrate, the phenolic coupling partner, and a slight excess of base to ensure complete deprotonation of the phenol. The catalyst system is then introduced, typically consisting of copper(I) iodide and the amino acid ligand in a specific molar ratio, often ranging from 1:1 to 10:1 ligand-to-copper, dissolved in a suitable solvent like 1,4-dioxane. The mixture is heated under an inert atmosphere, although the reagents themselves are noted to be air-stable, which simplifies handling requirements compared to sensitive organometallic catalysts. For detailed operational parameters and specific stoichiometric recommendations derived directly from the patent examples, please refer to the standardized synthesis guide below.
- Combine aryl halide, phenol, cesium carbonate base, copper iodide catalyst, and N,N-dimethylglycine ligand in dioxane solvent.
- Heat the reaction mixture to 90°C under nitrogen protection for 16 to 24 hours to ensure complete conversion.
- Cool the reaction, extract with ethyl acetate, wash with brine, dry over sodium sulfate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this amino acid-ligand copper catalysis represents a strategic opportunity to drive significant cost reductions and enhance supply chain resilience for diaryl ether intermediates. The primary economic driver is the substitution of expensive palladium catalysts with commodity-grade copper salts and inexpensive amino acid derivatives, which are produced on a massive scale for other industries and thus benefit from stable pricing and abundant availability. This shift eliminates the volatility associated with precious metal markets and removes the need for specialized, costly scavenging resins often required to meet strict residual metal specifications in pharmaceutical products. Additionally, the ability to run reactions at lower temperatures translates directly into reduced energy consumption for heating and cooling cycles, contributing to lower overall utility costs per kilogram of product manufactured.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts and the use of cheap, commercially available ligands drastically lowers the raw material cost profile for diaryl ether synthesis. By avoiding the need for stoichiometric copper reagents and expensive phosphine ligands often used in palladium chemistry, manufacturers can achieve substantial savings in bill of materials. Furthermore, the high yields reported in the patent examples minimize waste generation and maximize the throughput of existing reactor capacity, effectively lowering the cost of goods sold without compromising on quality. The simplified purification process also reduces solvent usage and labor hours associated with complex workups, adding another layer of operational efficiency.
- Enhanced Supply Chain Reliability: Sourcing reliability is significantly improved as the key reagents—copper iodide, amino acids, and inorganic bases—are standard industrial chemicals with multiple global suppliers, reducing the risk of single-source bottlenecks. Unlike specialized ligands that may have long lead times or limited production capacity, these commoditized inputs ensure that production schedules can be maintained consistently even during periods of market fluctuation. The robustness of the reaction to air and moisture, as indicated by the stability of the catalyst components, further reduces the risk of batch failures due to environmental exposure during transport or storage, ensuring a steady flow of high-quality intermediates to downstream customers.
- Scalability and Environmental Compliance: The mild reaction conditions and high atom economy of this catalytic system make it inherently scalable from laboratory benchtop to multi-ton commercial production without the need for specialized high-pressure or high-temperature equipment. The reduced thermal load lowers the safety risks associated with exothermic runaway reactions, facilitating easier regulatory approval for new manufacturing processes. Moreover, the use of less toxic copper compared to other heavy metals and the potential for solvent recycling aligns well with increasingly stringent environmental regulations and corporate sustainability goals, positioning manufacturers favorably for green chemistry certifications and eco-conscious client partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this amino acid-mediated Ullmann coupling technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the process capabilities and limitations for potential adopters. Understanding these nuances is essential for R&D teams evaluating route selection and for procurement professionals assessing the feasibility of sourcing intermediates produced via this method.
Q: What are the advantages of using amino acid ligands in Ullmann coupling?
A: Amino acid ligands like N,N-dimethylglycine are inexpensive, air-stable, and allow the reaction to proceed at significantly lower temperatures (around 90°C) compared to traditional methods, improving safety and yield.
Q: Can this method tolerate electron-withdrawing groups?
A: Yes, the patent demonstrates excellent tolerance for functional groups such as nitro, cyano, and fluoro substituents, achieving yields up to 98% even with sensitive substrates.
Q: Is palladium required for this ether synthesis?
A: No, this method utilizes catalytic amounts of copper salts (e.g., CuI) instead of expensive palladium catalysts, significantly reducing raw material costs for large-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diaryl Ether Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust and scalable synthetic routes in the fast-paced pharmaceutical landscape, and we have integrated advanced copper-catalyzed methodologies like the one described in CN1203040C into our process development portfolio. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of diaryl ether intermediate meets the exacting standards required for API synthesis, providing our partners with the confidence they need to advance their drug candidates through clinical trials and into the market.
We invite you to collaborate with us to leverage these technological advancements for your specific project needs, whether you are looking to optimize an existing route or develop a new process from scratch. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your target molecule, demonstrating how this mild copper-catalyzed approach can reduce your overall manufacturing expenses. Please contact us today to request specific COA data and route feasibility assessments, and let us help you secure a reliable, cost-effective supply of high-quality diaryl ether intermediates for your global operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
