Advanced Purification Technology for High-Purity Fudosteine: Scaling Commercial Production with Superior Quality Control
The pharmaceutical industry's relentless pursuit of higher purity standards for active pharmaceutical ingredients (APIs) and intermediates has driven significant innovation in downstream processing technologies. Patent CN102180820B represents a pivotal advancement in the manufacturing of Fudosteine, a potent mucolytic agent widely used in the treatment of chronic respiratory diseases. This patent discloses a novel purification method that transcends the limitations of traditional recrystallization techniques, utilizing a specific water-methanol solvent system to consistently achieve HPLC purity levels exceeding 99.5%. For R&D directors and process chemists, this development is critical, as it addresses the stringent regulatory requirements for impurity profiles in modern drug registration dossiers. By optimizing solubility parameters and crystallization kinetics, this technology offers a robust pathway to producing high-quality Fudosteine that meets the rigorous demands of global pharmacopoeias.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of Fudosteine has relied heavily on water-ethanol recrystallization or simple water-based precipitation methods, which often struggle to push purity levels beyond the 99.0% threshold. As detailed in prior art such as US5047428 and CN200910167947, conventional water-ethanol systems typically yield products with HPLC purity ranging from 98.5% to 98.6%, while water-only recrystallization might reach 99.0% but often at the cost of yield or crystal habit. Furthermore, alternative methods involving acetone precipitation have demonstrated even lower efficacy, with purity levels stagnating around 97.7%. These limitations pose significant challenges for procurement managers and quality assurance teams, as failing to meet the >99.5% benchmark often necessitates additional, costly purification steps like preparative chromatography, thereby inflating production costs and extending lead times for high-purity pharmaceutical intermediates.
The Novel Approach
The innovative methodology outlined in patent CN102180820B fundamentally shifts the purification paradigm by introducing a controlled water-methanol solvent system that leverages distinct solubility characteristics to maximize impurity rejection. Unlike the broader solubility curves observed in ethanol mixtures, the specific interaction between Fudosteine, water, and methanol allows for a more precise crystallization window, typically between -25°C and 15°C. This approach not only facilitates the removal of colored impurities through integrated activated carbon treatment but also ensures that the crystal lattice forms with minimal inclusion of mother liquor contaminants. Experimental data confirms that this novel route consistently delivers purity levels of 99.5% to 99.7%, effectively eliminating the need for complex secondary purification stages and providing a streamlined, cost-effective solution for cost reduction in API manufacturing.
Mechanistic Insights into Solvent-Mediated Recrystallization
The success of this purification strategy lies in the precise manipulation of intermolecular forces and solvation dynamics inherent to the Fudosteine molecule. Fudosteine, chemically known as 3-hydroxypropyl sulfo-L-Ala, possesses a zwitterionic structure with multiple hydrogen-bonding donors and acceptors, including amine, carboxyl, hydroxyl, and thioether groups.
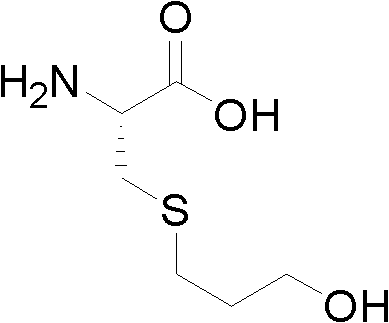
These functional groups create a complex solubility profile where the molecule is highly soluble in hot water but exhibits sharply reduced solubility upon the addition of methanol, a less polar anti-solvent. By carefully tuning the volume ratio of water to methanol—ranging from 4 to 6000 parts depending on the specific embodiment—the process engineers can control the supersaturation level, which is the driving force for nucleation and crystal growth. This controlled supersaturation prevents the rapid, chaotic precipitation that often traps impurities within the crystal matrix, instead favoring the slow, ordered growth of high-purity crystals. Additionally, the inclusion of activated carbon prior to crystallization targets organic impurities and colored by-products via adsorption, ensuring that the final solid form is not only chemically pure but also visually compliant with aesthetic standards required for pharmaceutical-grade materials.
From an impurity control perspective, this mechanism is particularly effective against structurally related by-products formed during the upstream synthesis of Fudosteine from L-cysteine and 3-bromo-1-propanol. The differential solubility ensures that unreacted starting materials and side-reaction products remain in the mother liquor rather than co-crystallizing with the target compound. The temperature gradient, moving from dissolution at up to 100°C down to crystallization at sub-zero temperatures, further exploits the thermodynamic differences between the product and its impurities. This thermodynamic selectivity is crucial for maintaining a clean impurity spectrum, which simplifies the analytical validation process and reduces the risk of batch rejection during quality control testing, thereby enhancing overall supply chain reliability.
How to Synthesize Fudosteine Efficiently
The implementation of this purification protocol requires strict adherence to the specified solvent ratios and temperature controls to replicate the high-purity results observed in the patent examples. The process begins with the dissolution of the crude material in a defined volume of water, followed by a critical decolorization step using activated carbon, which must be filtered off before the anti-solvent is introduced. The subsequent addition of methanol must be managed to avoid local over-concentration, which could lead to oiling out or amorphous precipitation rather than crystalline formation. For a comprehensive understanding of the operational parameters, the detailed standardized synthesis steps are provided in the guide below.
- Dissolve Fudosteine crude product in water (40-600 volumes) at temperatures between 10°C and 100°C.
- Add activated carbon (0.005-0.05 times the weight of water) for adsorption and decolorization, then filter the solution.
- Add methanol (4-6000 parts by weight) to the filtrate and induce crystallization at temperatures between -25°C and 15°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this water-methanol purification technology translates directly into tangible operational efficiencies and risk mitigation strategies. By replacing less efficient solvent systems with this optimized protocol, manufacturers can significantly reduce the number of processing cycles required to meet specification, thereby lowering utility consumption and labor costs associated with extended production runs. The reliance on commodity solvents like water and methanol, which are globally available and cost-effective, eliminates the supply risks associated with specialized or regulated reagents, ensuring a stable and continuous flow of raw materials for commercial scale-up of complex pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The primary economic driver of this technology is the elimination of the need for expensive and time-consuming chromatographic purification steps that are often required when recrystallization fails to achieve >99.5% purity. By achieving high purity in a single crystallization step, the process drastically simplifies the workflow, reducing solvent recovery loads and minimizing waste disposal costs. Furthermore, the high yield associated with this method ensures that valuable raw materials are not lost to excessive mother liquor retention, maximizing the overall mass balance and contributing to substantial cost savings in the final price per kilogram of the active ingredient.
- Enhanced Supply Chain Reliability: The robustness of the water-methanol system enhances supply chain resilience by utilizing solvents that are not subject to the same volatile market fluctuations or regulatory restrictions as halogenated or aromatic solvents. This stability allows for long-term contracting and inventory planning without the fear of sudden raw material shortages. Additionally, the mild reaction conditions and straightforward filtration steps reduce the likelihood of equipment fouling or process deviations, leading to more predictable batch cycles and shorter lead times for high-purity pharmaceutical intermediates, which is critical for meeting just-in-time delivery schedules.
- Scalability and Environmental Compliance: From an environmental and safety perspective, this process is inherently greener, utilizing methanol which is easier to recover and recycle compared to higher boiling point alcohols, thus reducing the facility's overall carbon footprint. The simplicity of the unit operations—dissolution, filtration, and cooling—makes the technology highly scalable from pilot plant quantities to multi-ton commercial production without requiring significant re-engineering of existing infrastructure. This ease of scale-up ensures that supply can be rapidly ramped up to meet market demand spikes, while the reduced solvent usage aligns with increasingly stringent environmental regulations regarding volatile organic compound (VOC) emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this purification technology, derived directly from the experimental data and beneficial effects described in the patent documentation. These insights are intended to clarify the operational feasibility and quality advantages for potential partners evaluating this supply chain opportunity.
Q: Why is the water-methanol system superior to water-ethanol for Fudosteine purification?
A: The water-methanol system provides a sharper solubility differential compared to water-ethanol, allowing for more effective rejection of impurities during the crystallization phase, consistently achieving HPLC purity above 99.5% versus the 98.5-99.0% typical of older methods.
Q: What are the critical control parameters for this purification process?
A: Critical parameters include the precise ratio of water to methanol, the temperature range for dissolution (10-100°C) versus crystallization (-25 to 15°C), and the amount of activated carbon used for decolorization to ensure low related substances.
Q: Is this purification method suitable for large-scale industrial production?
A: Yes, the process relies on standard unit operations such as dissolution, filtration, and cooling crystallization using common solvents (water and methanol), making it highly scalable and safe for multi-ton commercial manufacturing without requiring complex equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fudosteine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory-scale innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high-purity standards achieved in the lab are maintained throughout mass manufacturing. We operate with stringent purity specifications and utilize rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify that every batch of Fudosteine meets the >99.5% HPLC purity benchmark required by top-tier pharmaceutical clients.
We invite global partners to collaborate with us to leverage this advanced purification technology for their respiratory drug portfolios. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis to quantify the economic benefits of switching to this superior process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of quality, reliability, and scientific excellence.
