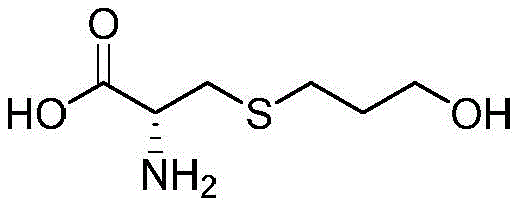
Revolutionizing Respiratory Drug Intermediates: Scalable Synthesis of High-Purity Fudosteine
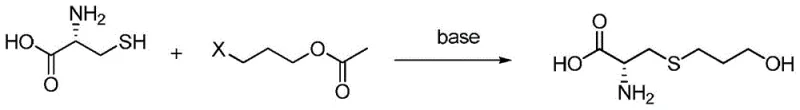
The pharmaceutical landscape for respiratory therapeutics continues to evolve, driven by the demand for mucolytic agents with superior safety and efficacy profiles. Central to this domain is Fudosteine, a potent mucoactive agent widely utilized in the treatment of chronic respiratory conditions such as bronchial asthma, bronchiectasis, and chronic bronchitis. The structural integrity and purity of this active pharmaceutical ingredient (API) are paramount, as even trace impurities can impact therapeutic outcomes and regulatory approval. A significant technological breakthrough in this sector is documented in patent CN108586298B, which discloses a novel preparation method for high-purity fudosteine. This patent outlines a sophisticated synthetic strategy that leverages 3-halopropyl acetate or propionate esters as key alkylating agents, reacting them with L-cysteine in an aqueous alkaline medium. This approach represents a paradigm shift from traditional methodologies, offering a pathway that is not only chemically elegant but also industrially robust. By addressing the critical pain points of previous synthesis routes—namely harsh reaction conditions, difficult purification, and high impurity loads—this technology provides a compelling value proposition for global supply chains seeking reliable pharmaceutical intermediate suppliers. The ability to consistently achieve unknown impurity levels below 0.1% while maintaining high molar yields positions this method as a cornerstone for modern API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of fudosteine has been plagued by significant technical hurdles that hinder efficient commercial scale-up of complex amino acid derivatives. Early methodologies often relied on the reaction of L-cysteine with allyl alcohol, a process that necessitates the use of aggressive catalysts such as sodium persulfate or exposure to intense light irradiation to drive the radical addition. These conditions introduce severe safety risks and make process control exceptionally difficult on a large scale, often resulting in products contaminated with residual salts and难以 remove byproducts. Alternative routes utilizing 3-halo-1-propanol have also been explored; however, the inherent low reactivity of the hydroxyl group in these substrates leads to incomplete conversions and prolonged reaction times. Furthermore, methods employing oxetane rings suffer from similar kinetic limitations, requiring extended periods to reach completion and often yielding products that fail to meet stringent purity specifications without exhaustive downstream processing. These legacy processes collectively contribute to inflated manufacturing costs, inconsistent batch quality, and supply chain vulnerabilities, creating an urgent need for innovation in cost reduction in API manufacturing.
The Novel Approach
The methodology described in patent CN108586298B fundamentally reimagines the synthetic trajectory by introducing activated ester derivatives as the electrophilic partners. By utilizing 3-halopropyl acetate or propionate, the process capitalizes on the enhanced leaving group ability and electronic activation of the halogenated carbon, facilitating a rapid and high-yielding nucleophilic substitution with the thiol group of L-cysteine. This reaction proceeds smoothly in an aqueous alkaline solution, where the base serves the dual purpose of deprotonating the cysteine thiol to enhance nucleophilicity and subsequently hydrolyzing the ester moiety to reveal the terminal hydroxyl group. This "one-pot" condensation and hydrolysis strategy eliminates the need for isolation of intermediate species, drastically simplifying the operational workflow. The result is a crude product of exceptional quality that requires minimal purification to achieve pharmaceutical grade standards. This streamlined approach not only accelerates production timelines but also significantly reduces the consumption of solvents and reagents, aligning perfectly with green chemistry principles and the economic imperatives of modern chemical production.
Mechanistic Insights into Base-Promoted Alkylation and Hydrolysis
The core of this synthetic innovation lies in the precise orchestration of nucleophilic substitution kinetics within an aqueous environment. The mechanism initiates with the deprotonation of the sulfhydryl (-SH) group of L-cysteine by the alkaline medium, generating a highly reactive thiolate anion. This anion acts as a potent soft nucleophile, attacking the electrophilic carbon atom bonded to the halogen in the 3-halopropyl acetate substrate. The presence of the adjacent ester group, while primarily serving as a protected alcohol, subtly influences the electronic environment of the alkyl chain, potentially stabilizing the transition state through inductive effects. As the substitution proceeds, the halide ion is expelled, forming the thioether linkage that characterizes the fudosteine backbone. Crucially, the reaction conditions are maintained such that the ester functionality remains intact during the initial alkylation but is susceptible to saponification under the prevailing basic conditions. This ensures that the final product possesses the free hydroxyl group required for biological activity without necessitating a separate deprotection step. The interplay between reaction temperature, typically ranging from -20°C to 100°C depending on the specific halide and base employed, and the concentration of the alkaline species allows for fine-tuning of the reaction rate to minimize side reactions such as disulfide formation or elimination pathways.
Impurity control is intrinsically built into the mechanistic design of this process. In conventional syntheses, the formation of disulfide dimers (cystine) is a pervasive issue caused by the oxidation of free thiols. However, the rapid consumption of the thiolate anion by the excess alkylating agent in this novel method kinetically outcompetes the oxidation pathway, thereby suppressing dimer formation. Furthermore, the use of specific extraction protocols involving non-polar solvents like n-heptane or cyclohexane effectively removes unreacted organic halides and lipophilic byproducts prior to the final crystallization. The subsequent pH adjustment to the isoelectric point of fudosteine (pH 5-6) ensures maximum precipitation of the target molecule while keeping charged impurities in the aqueous supernatant. Finally, the recrystallization step utilizing water-soluble organic solvents such as ethanol or acetone exploits the differential solubility profiles of fudosteine versus its structural analogs. This multi-stage purification logic ensures that the final API meets the rigorous requirement of having unknown impurities below 0.1%, a benchmark that is critical for regulatory compliance in major markets.
How to Synthesize Fudosteine Efficiently
Implementing this high-efficiency synthesis requires careful attention to stoichiometry and thermal management to maximize yield and purity. The process begins with the preparation of a homogeneous alkaline solution, where L-cysteine is dissolved in water containing a base such as sodium hydroxide, potassium carbonate, or organic amines like triethylamine. The choice of base can influence the solubility of the starting materials and the rate of the subsequent hydrolysis. Once the cysteine is fully solubilized, the temperature is carefully controlled, often cooled to sub-ambient conditions initially to manage the exotherm upon addition of the alkylating agent. The 3-halopropyl acetate is then introduced dropwise, either neat or dissolved in a water-miscible co-solvent like ethanol, to maintain a consistent reaction concentration. Monitoring the reaction progress via HPLC is essential to determine the precise endpoint, ensuring complete conversion before proceeding to workup. Following the reaction, the mixture undergoes extraction to remove organic impurities, followed by acidification to precipitate the crude product. The detailed standardized synthesis steps, including specific solvent ratios, temperature ramps, and crystallization parameters, are outlined in the guide below.
- Dissolve L-cysteine in an aqueous alkaline solution containing bases like sodium hydroxide or triethylamine, maintaining temperatures between -20°C and 100°C.
- Dropwise add 3-halopropyl acetate or propionate to the reaction mixture, monitoring progress via HPLC until completion of the condensation and hydrolysis.
- Adjust the pH of the aqueous phase to 5-6 using acid, concentrate the solution, and purify the crude solid through recrystallization with water-soluble organic solvents.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers transformative benefits that extend far beyond simple chemical yield. The shift from hazardous or sluggish reagents to readily available halo-ester derivatives fundamentally alters the cost structure of fudosteine production. By eliminating the need for specialized equipment required for high-pressure or photochemical reactions, capital expenditure is significantly reduced, and operational flexibility is enhanced. The robustness of the aqueous-based system minimizes the reliance on large volumes of expensive anhydrous organic solvents, leading to substantial cost savings in raw material procurement and waste disposal. Furthermore, the high selectivity of the reaction reduces the burden on downstream purification units, allowing for faster batch turnover and increased throughput without compromising quality. These factors collectively contribute to a more resilient supply chain capable of meeting fluctuating market demands for respiratory therapeutics.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the utilization of commodity-grade starting materials that are abundant in the global chemical market. Unlike methods requiring custom-synthesized precursors or precious metal catalysts, the 3-halopropyl acetates are produced at scale, ensuring stable pricing and availability. The one-pot nature of the reaction consolidates multiple unit operations into a single vessel, reducing labor costs, energy consumption for heating and cooling cycles, and equipment occupancy time. Additionally, the high molar yields reported in the patent examples indicate minimal material loss, directly translating to a lower cost of goods sold (COGS). The elimination of complex chromatographic purification steps in favor of simple crystallization further drives down processing costs, making this route highly competitive for generic API production.
- Enhanced Supply Chain Reliability: Supply continuity is a critical metric for pharmaceutical manufacturers, and this synthesis method enhances reliability by diversifying the source of key inputs. The reagents involved, such as L-cysteine and various halo-acetates, are manufactured by multiple suppliers worldwide, mitigating the risk of single-source bottlenecks. The mild reaction conditions reduce the likelihood of unplanned shutdowns due to safety incidents or equipment failures associated with high-energy processes. Moreover, the simplified workup procedure shortens the overall manufacturing cycle time, enabling producers to respond more agilely to purchase orders. This agility is crucial for reducing lead time for high-purity respiratory drug intermediates, ensuring that downstream formulation teams receive their materials exactly when needed to maintain production schedules.
- Scalability and Environmental Compliance: As regulatory scrutiny on environmental impact intensifies, the green credentials of this synthesis route provide a distinct strategic advantage. The use of water as the primary reaction medium aligns with sustainability goals by reducing the volatile organic compound (VOC) emissions associated with solvent-intensive processes. The waste streams generated are predominantly aqueous and contain biodegradable organic salts, which are easier and cheaper to treat compared to heavy metal-contaminated waste from catalytic methods. The process is inherently scalable, having been demonstrated to perform consistently from gram to kilogram scales in the patent examples, suggesting a smooth path to multi-ton commercial production. This scalability ensures that as demand for fudosteine grows, manufacturers can expand capacity without encountering the technical barriers often faced when transitioning from lab-scale to plant-scale operations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced fudosteine synthesis technology. These insights are derived directly from the experimental data and claims presented in patent CN108586298B, providing a factual basis for decision-making. Understanding the nuances of reaction conditions, impurity profiles, and scalability is essential for R&D teams evaluating technology transfer and for procurement professionals assessing supplier capabilities. The answers below clarify how this method overcomes historical challenges and delivers a superior product profile suitable for the stringent requirements of the global pharmaceutical industry.
Q: What are the primary advantages of using 3-halopropyl acetate over allyl alcohol for fudosteine synthesis?
A: Unlike allyl alcohol methods which require dangerous conditions like strong light irradiation or persulfate catalysts, the 3-halopropyl acetate route operates under mild thermal conditions with significantly improved safety profiles and easier process control.
Q: How does this novel method achieve impurity levels below 0.1%?
A: The process utilizes a highly selective nucleophilic substitution followed by a robust recrystallization protocol using specific water-soluble organic solvents, which effectively excludes structurally related byproducts and residual salts.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the method avoids expensive transition metal catalysts and hazardous reagents, utilizing readily available commodity chemicals and standard aqueous workup procedures that are inherently scalable for multi-ton manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fudosteine Supplier
The technical potential of the synthesis route described in CN108586298B is immense, offering a clear pathway to producing high-quality fudosteine at a competitive cost. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this chemistry to life. Our facility is equipped with state-of-the-art reactors capable of handling the precise temperature controls and aqueous-organic biphasic systems demanded by this process. We adhere to stringent purity specifications and operate rigorous QC labs to ensure that every batch of fudosteine meets the <0.1% unknown impurity threshold, guaranteeing safety and efficacy for your final drug product. Our commitment to quality assurance extends from raw material sourcing to final packaging, ensuring full traceability and compliance with international GMP standards.
We invite you to leverage our expertise to optimize your supply chain for respiratory therapeutics. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data from our pilot runs and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term business goals. Let us be your partner in delivering high-purity fudosteine to the patients who need it most.
