Scalable Biocatalytic Production of Chiral Bepotastine Intermediates for Global Pharma Supply Chains
The pharmaceutical industry continuously seeks robust, scalable, and environmentally sustainable pathways for synthesizing complex chiral intermediates, particularly for antihistamine agents like Bepotastine. Patent CN109957585B introduces a groundbreaking biocatalytic methodology for the preparation of (S)-(4-chlorophenyl)-2-pyridinemethanol, a critical chiral building block. This technology leverages a specifically engineered ketoreductase (KRED) to achieve near-perfect stereocontrol and conversion efficiency. By shifting away from traditional transition metal catalysis, this process addresses key pain points in modern API manufacturing, including heavy metal residue limits and high-pressure safety concerns. The patent details a highly optimized enzymatic system capable of operating at unprecedented substrate concentrations, signaling a major leap forward in industrial biotechnology applications for fine chemical synthesis.
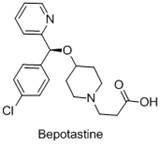
Bepotastine besilate is a potent, non-sedative histamine H1 receptor antagonist widely used for treating allergic rhinitis and urticaria. The therapeutic efficacy and safety profile of this drug are strictly dependent on the stereochemical purity of its core structure. As illustrated in the molecular architecture, the presence of the chiral ether linkage requires precise synthetic control. The demand for high-purity intermediates has driven extensive research into asymmetric synthesis, yet many existing routes suffer from low yields or require costly chiral resolution steps. This patent provides a direct, asymmetric reduction pathway that bypasses these inefficiencies, offering a streamlined route to the active pharmacophore.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral benzylic alcohols like (S)-(4-chlorophenyl)-2-pyridinemethanol has relied heavily on chemical reduction strategies involving stoichiometric chiral auxiliaries or transition metal catalysts. Traditional approaches, such as those utilizing BINAL-H or rhodium-based complexes, often necessitate harsh reaction conditions, including high-pressure hydrogenation and strictly anhydrous environments. These methods not only incur substantial capital expenditure for specialized high-pressure reactors but also introduce significant safety hazards associated with hydrogen gas handling. Furthermore, chemical catalysts frequently struggle to achieve the stringent enantiomeric excess (ee) values required by regulatory bodies like the FDA and EMA without additional, yield-eroding purification steps. The residual risk of heavy metal contamination, particularly from noble metals like rhodium, imposes rigorous and costly downstream purification requirements to meet ICH Q3D guidelines.
The Novel Approach
In stark contrast, the biocatalytic process disclosed in CN109957585B utilizes a highly specific ketoreductase derived from Novosphingobium aromaticivorans to effect the asymmetric reduction under mild, ambient conditions. This enzymatic approach operates efficiently in a mixed aqueous-organic solvent system, eliminating the need for flammable organic solvents as the primary reaction medium. The novelty lies in the enzyme's exceptional tolerance to high substrate loadings, achieving concentrations close to 330g/L, which drastically improves volumetric productivity compared to prior art enzymatic methods that typically operate below 10g/L. By coupling the reduction with an in-situ cofactor regeneration system using isopropanol, the process ensures atom economy and minimizes waste generation. This shift from chemocatalysis to biocatalysis represents a paradigm shift towards greener, safer, and more cost-effective manufacturing paradigms for complex pharmaceutical intermediates.
Mechanistic Insights into Ketoreductase-Catalyzed Asymmetric Reduction
The core of this technological advancement is the specific ketoreductase (KRED) defined by the amino acid sequence SEQ ID No. 2. This enzyme facilitates the hydride transfer from the reduced cofactor (NADPH) to the prochiral ketone substrate, 4-chlorophenyl-(pyridin-2-yl)-methanone, with exquisite stereoselectivity. The mechanism involves the formation of a transient enzyme-substrate-cofactor ternary complex where the active site geometry strictly dictates the approach of the hydride ion to the re-face or si-face of the carbonyl group. In this specific case, the enzyme architecture favors the formation of the (S)-enantiomer with an ee value exceeding 99.5%. The catalytic cycle is sustained through a coupled oxidation of isopropanol to acetone, which regenerates the oxidized cofactor (NADP+) back to its reduced form (NADPH), allowing the enzyme to turnover thousands of times without the need for stoichiometric amounts of expensive cofactors.
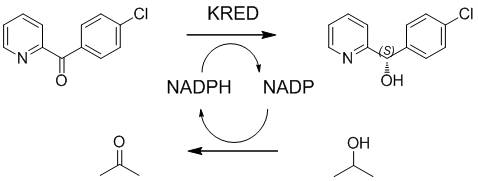
Impurity control in this biocatalytic system is inherently superior to chemical methods due to the high specificity of the enzyme. Unlike chemical reducing agents which may reduce other functional groups or cause over-reduction, the ketoreductase targets only the specific ketone moiety. The patent data indicates a substrate conversion rate of greater than 99%, leaving minimal unreacted starting material which simplifies downstream isolation. The absence of metal catalysts eliminates the formation of metal-complexed impurities, and the mild pH and temperature conditions prevent thermal degradation or racemization of the sensitive chiral product. This high fidelity in reaction specificity ensures that the crude product profile is exceptionally clean, reducing the burden on crystallization and chromatography steps during final purification.
How to Synthesize (S)-(4-chlorophenyl)-2-pyridinemethanol Efficiently
The practical implementation of this synthesis involves preparing a reaction mixture comprising the ketone substrate, the recombinant ketoreductase (either as cell lysate or purified enzyme powder), and a buffering agent. Isopropanol serves a dual role as a co-solvent to solubilize the hydrophobic substrate and as the hydrogen donor for cofactor regeneration. The reaction is typically conducted at temperatures between 22°C and 45°C with magnetic stirring to ensure homogeneity. Detailed standardized operating procedures, including specific ratios of enzyme to substrate and precise pH controls, are critical for maximizing yield and optical purity. For a comprehensive step-by-step guide on executing this synthesis at scale, please refer to the technical protocol section below.
- Prepare a reaction system containing the ketone substrate, specific ketoreductase (SEQ ID No. 2), and coenzyme NADP+ in a phosphate buffer/isopropanol mixture.
- Maintain the reaction temperature between 22-45°C and stir magnetically to ensure efficient mass transfer and enzyme activity.
- Monitor conversion via TLC or HPLC until completion (>99%), then proceed to filtration and organic extraction for product isolation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this biocatalytic technology offers transformative economic and logistical benefits. The transition from noble metal catalysis to enzymatic catalysis fundamentally alters the cost structure of the intermediate. By eliminating the need for expensive rhodium or iridium catalysts and the associated high-pressure equipment, the capital expenditure (CAPEX) and operational expenditure (OPEX) are significantly reduced. The ability to run reactions at high substrate concentrations means that smaller reactor volumes can produce the same amount of product, effectively increasing facility throughput without physical expansion. This efficiency translates directly into a more competitive pricing structure for the final API, providing a strategic advantage in cost-sensitive generic drug markets.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts removes a major cost driver and supply risk associated with fluctuating metal prices. Furthermore, the simplified work-up procedure, which avoids complex metal scavenging steps, reduces the consumption of auxiliary chemicals and processing time. The high conversion rate ensures that raw material utilization is maximized, minimizing the cost of goods sold (COGS) related to wasted starting materials. Overall, the process economics are vastly improved by the combination of high yield, mild conditions, and reduced downstream processing complexity.
- Enhanced Supply Chain Reliability: Biocatalytic processes are generally more robust and easier to scale than sensitive organometallic reactions. The use of genetically engineered microorganisms allows for consistent, renewable production of the catalyst, insulating the supply chain from geopolitical disruptions often associated with mining-derived metals. The mild reaction conditions reduce the risk of batch failures due to thermal runaways or pressure excursions, ensuring consistent on-time delivery. Additionally, the high purity of the crude product reduces the likelihood of batch rejection during quality control, further stabilizing the supply flow to downstream API manufacturers.
- Scalability and Environmental Compliance: The process operates in an aqueous environment with low toxicity solvents, aligning perfectly with increasingly stringent global environmental regulations. The reduction in hazardous waste generation and the absence of heavy metal effluents simplify wastewater treatment and lower disposal costs. This green chemistry profile not only ensures regulatory compliance but also enhances the corporate sustainability image of the manufacturing entity. The demonstrated scalability from gram to kilogram levels in the patent examples confirms the technology's readiness for multi-ton commercial production, securing long-term supply continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this biocatalytic route. Understanding these details is crucial for R&D teams evaluating process transfer and procurement teams assessing vendor capabilities. The answers are derived directly from the experimental data and claims presented in the patent documentation, ensuring accuracy and relevance for decision-making.
Q: What is the optical purity achievable with this biocatalytic process?
A: The patented process utilizing the specific ketoreductase achieves an enantiomeric excess (ee) value of not less than 99.5%, meeting strict ICH guidelines for chiral pharmaceutical intermediates without requiring further chiral purification.
Q: How does this method compare to traditional metal-catalyzed reduction?
A: Unlike traditional methods using expensive rhodium catalysts and high-pressure hydrogenation, this biocatalytic route operates under mild atmospheric conditions in an aqueous system, eliminating heavy metal contamination risks and significantly lowering equipment and safety costs.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process supports extremely high substrate concentrations up to 330g/L with conversion rates exceeding 99%, demonstrating robust scalability and high atom utilization suitable for commercial tonnage production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-(4-chlorophenyl)-2-pyridinemethanol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality chiral intermediates in the development of next-generation antihistamines. Our technical team has thoroughly analyzed the biocatalytic pathways described in CN109957585B and possesses the expertise to implement and optimize this technology for commercial production. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of materials. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee ee values exceeding 99.5%.
We invite potential partners to engage with our technical procurement team to discuss how this advanced biocatalytic route can be integrated into your supply chain. By leveraging our manufacturing capabilities, you can secure a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, ensuring that your project moves forward with the most efficient and compliant synthetic strategy available.
