Advanced Acid-Catalyzed Synthesis of Bepotastine Intermediates for Commercial Scale-Up
Introduction to Novel Bepotastine Synthesis Technology
The pharmaceutical industry continuously seeks robust synthetic pathways for active pharmaceutical ingredients (APIs) that balance high stereochemical purity with economic viability. A significant breakthrough in this domain is documented in Chinese Patent CN102675283A, which discloses a novel method for preparing Bepotastine, a potent second-generation histamine H1 receptor antagonist used widely in the treatment of allergic rhinitis and urticaria. This patent introduces a sophisticated acid-catalyzed condensation strategy that fundamentally alters the construction of the ether linkage between the chiral benzhydryl moiety and the piperidine side chain. Unlike conventional approaches that rely on harsh basic conditions, this innovation leverages the reactivity of trichloroacetimidate intermediates under mild Lewis acid catalysis. For R&D directors and process chemists, this represents a critical evolution in synthetic design, offering a pathway that inherently mitigates the risks of racemization and elimination side reactions. By shifting the mechanistic paradigm from nucleophilic substitution under basic conditions to activated electrophilic substitution under acidic conditions, the technology ensures the preservation of the critical (S)-configuration essential for biological activity. This report analyzes the technical merits of this approach, evaluating its potential to redefine the supply chain reliability and cost structure for reliable pharmaceutical intermediates supplier networks globally.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Bepotastine and its analogues has been plagued by significant chemical challenges associated with the formation of the chiral ether bond. Traditional methodologies, such as those disclosed in earlier patents by major Japanese pharmaceutical firms, typically involve the reaction of a chiral alcohol with a halogenated piperidine derivative or vice versa under strongly basic conditions. These processes frequently employ reagents like sodium hydride (NaH) or sodium amide to deprotonate the hydroxyl group, generating a highly reactive alkoxide nucleophile. However, the presence of such strong bases creates a hostile environment for the sensitive chiral center adjacent to the pyridine and chlorophenyl rings. The high basicity promotes the abstraction of the acidic alpha-proton, leading to rapid racemization and a consequent loss of optical purity, which is detrimental to the drug's efficacy and safety profile. Furthermore, the basic conditions often trigger unwanted elimination reactions on the piperidine ring, particularly when leaving groups like bromine or chlorine are present, resulting in the formation of olefinic impurities that are difficult to remove. These side reactions not only depress the overall yield but also complicate the downstream purification process, necessitating expensive chromatographic separations or multiple recrystallizations to meet stringent regulatory standards for high-purity pharmaceutical intermediates.
The Novel Approach
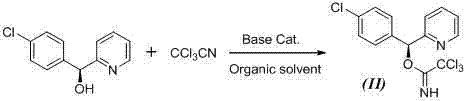
In stark contrast to these legacy methods, the technology described in CN102675283A employs a trichloroacetimidate activation strategy that operates under acidic catalysis, effectively circumventing the pitfalls of base-mediated chemistry. The core of this innovation lies in the conversion of the chiral (S)-(4-chlorophenyl)-(pyridin-2-yl)methanol into a trichloroacetimidate ester, which serves as a highly reactive electrophile. When treated with a Lewis acid catalyst such as trifluoromethanesulfonic acid (TfOH) or trimethylsilyl trifluoromethanesulfonate (TMSOTf), the imidate group is activated, facilitating a smooth nucleophilic attack by the hydroxyl group of the piperidine derivative. This mechanism proceeds without the need for strong bases, thereby eliminating the driving force for racemization and preserving the stereochemical integrity of the molecule with exceptional fidelity. The reaction conditions are remarkably mild, typically proceeding at temperatures ranging from 0°C to 30°C, which further suppresses thermal degradation and side reactions. As illustrated in the reaction scheme, the subsequent hydrolysis step converts the ester intermediate directly into the target carboxylic acid, streamlining the synthesis into a concise and efficient sequence. This approach not only enhances the chemical yield, reported to exceed 70% for the chiral series, but also significantly simplifies the impurity profile, making it an ideal candidate for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Lewis Acid-Catalyzed Etherification
The success of this synthetic route hinges on the precise electronic activation of the trichloroacetimidate leaving group by the Lewis acid catalyst. In the absence of base, the oxygen atom of the imidate coordinates with the Lewis acid (e.g., TMSOTf), increasing the electrophilicity of the benzylic carbon. This activation lowers the energy barrier for the nucleophilic substitution, allowing the relatively weak nucleophile (the piperidine alcohol) to attack efficiently. Crucially, because the reaction medium remains acidic or neutral throughout the condensation phase, the chiral benzylic proton is never exposed to a basic environment that could facilitate enolization or carbanion formation. This mechanistic feature is the key to maintaining the high enantiomeric excess (ee%) observed in the examples, where values greater than 98% are consistently achieved starting from high-purity chiral alcohols. The stability of the trichloroacetimidate intermediate itself is also noteworthy; it can be prepared separately and stored, or generated in situ, providing flexibility in process design. The preparation of this key intermediate involves the reaction of the chiral alcohol with trichloroacetonitrile in the presence of a mild base, a step that is well-controlled and does not compromise the stereochemistry if managed correctly.
Furthermore, the choice of solvent plays a pivotal role in optimizing the reaction kinetics and selectivity. The patent data indicates that halogenated solvents like dichloromethane or chloroform, often mixed with non-polar solvents like hexane, provide the optimal environment for the Lewis acid catalysis. These solvents stabilize the charged intermediates formed during the activation of the imidate while ensuring good solubility for both the polar piperidine derivative and the lipophilic aromatic components. The avoidance of protic solvents is essential to prevent premature hydrolysis of the reactive imidate species. From an impurity control perspective, the acidic conditions effectively suppress the formation of elimination byproducts that are characteristic of the competing E2 mechanisms seen in basic conditions. Instead, the reaction pathway is funneled almost exclusively towards the desired SN1-like or SN2-like substitution depending on the specific stability of the carbocation character induced by the Lewis acid. This high degree of chemoselectivity reduces the burden on downstream purification, allowing for simpler workup procedures such as direct concentration and crystallization, which are highly desirable for scaling up complex pharmaceutical intermediates.
How to Synthesize Bepotastine Efficiently
The implementation of this acid-catalyzed protocol offers a straightforward operational framework for laboratory and pilot-scale synthesis. The process begins with the activation of the chiral alcohol to form the trichloroacetimidate, followed by the coupling reaction with the piperidine alcohol component under strictly controlled acidic conditions. The detailed standardized synthesis steps see the guide below, which outlines the specific molar ratios, temperature controls, and workup procedures necessary to achieve the high yields and purity reported in the patent literature. Adhering to these parameters ensures that the benefits of the novel methodology are fully realized in a production setting.
- Preparation of the chiral trichloroacetimidate intermediate (Formula II) by reacting (S)-(4-chlorophenyl)-(pyridin-2-yl)methanol with trichloroacetonitrile under basic catalysis.
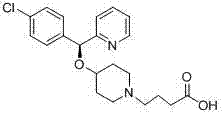
- Condensation of the imidate intermediate with 4-(4-hydroxypiperidin-1-yl)ethyl butyrate in an organic solvent using a Lewis acid catalyst such as triflic acid or TMSOTf.
- Hydrolysis of the resulting ester intermediate using aqueous alkali followed by acidification to isolate the final Bepotastine product with retained stereochemistry.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this acid-catalyzed synthesis route presents compelling economic and logistical advantages over traditional manufacturing methods. The primary value driver is the substantial improvement in process efficiency, which translates directly into lower production costs and enhanced supply security. By eliminating the need for expensive chiral resolution steps that are often required to correct racemization in older processes, manufacturers can significantly reduce raw material consumption and waste generation. The higher yields achieved through this method mean that less starting material is required to produce the same amount of final API, effectively lowering the cost of goods sold (COGS). Moreover, the use of milder reaction conditions reduces the energy demand for heating or cryogenic cooling, contributing to a more sustainable and cost-effective operation. The simplified impurity profile also means reduced solvent usage for purification and shorter cycle times, allowing for faster throughput and better responsiveness to market demand fluctuations.
- Cost Reduction in Manufacturing: The elimination of strong bases and the associated side reactions leads to a drastic simplification of the purification workflow. In traditional base-mediated routes, significant resources are often allocated to removing olefinic impurities and separating racemic byproducts, which drives up costs. By preventing these impurities from forming in the first place, the new route minimizes the need for complex chromatography or multiple recrystallizations. This streamlining of the downstream processing results in significant cost savings, making the production of high-purity pharmaceutical intermediates more economically viable. Additionally, the catalysts used, such as triflic acid or TMSOTf, are used in catalytic amounts and are compatible with standard reactor materials, avoiding the need for specialized corrosion-resistant equipment that might be required for more aggressive reagents.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route enhances supply chain continuity by reducing the risk of batch failures due to stereochemical drift. In the pharmaceutical industry, a batch that fails to meet chiral purity specifications can result in significant delays and financial losses. The inherent stability of the chiral center under the acidic conditions described in the patent ensures consistent quality across batches, reducing the likelihood of out-of-specification (OOS) results. Furthermore, the raw materials required, including trichloroacetonitrile and the piperidine alcohol derivative, are commercially available and stable, reducing the risk of supply disruptions. This reliability is crucial for maintaining uninterrupted production schedules for critical allergy medications, ensuring that downstream formulation partners receive their supplies on time.
- Scalability and Environmental Compliance: The mild operating temperatures and the absence of hazardous strong bases make this process highly scalable and safer to operate on a multi-ton scale. Strong bases like sodium hydride pose significant safety risks, including fire hazards and the generation of hydrogen gas, which require specialized handling and containment measures. By replacing these with Lewis acids, the process safety profile is markedly improved, facilitating easier scale-up from kilogram to metric ton quantities. From an environmental perspective, the reduction in waste generation and solvent usage aligns with green chemistry principles, helping manufacturers meet increasingly stringent environmental regulations. The ability to recycle solvents and the reduced load on wastewater treatment systems further contribute to the sustainability credentials of this manufacturing route.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Bepotastine synthesis technology. These answers are derived directly from the experimental data and technical disclosures found in the source patent, providing a factual basis for decision-making.
Q: How does the acid-catalyzed method prevent racemization compared to traditional base-mediated routes?
A: Traditional methods often utilize strong bases like sodium hydride (NaH), which can deprotonate the chiral center leading to racemization. The patented acid-catalyzed route activates the leaving group via protonation or Lewis acid coordination, facilitating nucleophilic attack without disturbing the chiral alpha-proton, thus preserving high enantiomeric excess (ee% > 98%).
Q: What are the primary impurities avoided by using trichloroacetimidate intermediates?
A: By avoiding strong alkaline conditions, this method significantly suppresses elimination reactions on the piperidine ring that typically generate olefinic byproducts. Additionally, the mild reaction temperatures (0°C to 30°C) minimize thermal degradation and side reactions common in harsher synthetic protocols.
Q: Is this synthesis route suitable for large-scale manufacturing of Bepotastine?
A: Yes, the process utilizes commercially available solvents like dichloromethane and THF, and catalysts such as triflic acid which are scalable. The high yields (reported up to 78% for the racemate and >70% for the chiral form) and simplified workup procedures make it highly viable for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bepotastine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthetic methodologies like the acid-catalyzed route for Bepotastine. As a leading CDMO and supplier in the fine chemical sector, we possess the technical expertise and infrastructure to translate such innovative patent technologies into commercial reality. Our team of experienced process chemists is adept at optimizing reaction parameters to maximize yield and purity, ensuring that the theoretical benefits of this route are fully captured in production. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, leveraging our state-of-the-art facilities to deliver consistent quality. Our commitment to excellence is underpinned by stringent purity specifications and rigorous QC labs that utilize advanced analytical techniques, including chiral HPLC, to verify the stereochemical integrity of every batch we produce.
We invite global pharmaceutical partners to collaborate with us to leverage this cost-effective and robust synthesis route for your Bepotastine supply needs. Whether you require custom synthesis services or bulk supply of key intermediates, our technical procurement team is ready to assist. Contact us today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to understand how our capabilities can support your supply chain goals and drive value for your organization.
