Advanced Biocatalytic Synthesis of Dexketoprofen: A Commercial Scale-Up Perspective
Advanced Biocatalytic Synthesis of Dexketoprofen: A Commercial Scale-Up Perspective
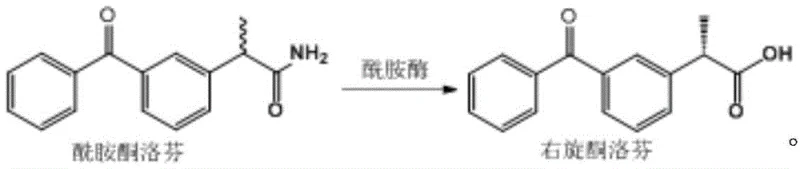
The pharmaceutical industry is currently witnessing a paradigm shift towards greener, more efficient synthetic routes for non-steroidal anti-inflammatory drugs (NSAIDs), particularly for chiral molecules like dexketoprofen. A pivotal development in this domain is documented in patent CN111363736A, which discloses a highly specific enzymatic method for preparing dexketoprofen. This technology leverages a novel amidase derived from Klebsiella species to catalyze the deamination of amide ketoprofen, offering a robust alternative to traditional chemical synthesis. For R&D directors and procurement strategists, this patent represents a significant opportunity to optimize the manufacturing of high-purity pharmaceutical intermediates. The core innovation lies in the enzyme's exceptional stereoselectivity, which addresses long-standing challenges regarding optical purity and environmental impact in the production of this critical analgesic agent.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the commercial production of (S)-(+)-ketoprofen has relied heavily on traditional chemical synthesis or chiral resolution techniques, both of which present substantial drawbacks for modern sustainable manufacturing. Conventional chemical routes often involve multi-step sequences requiring harsh reagents, such as Sharpless epoxidation followed by enantioselective hydrogenolysis and oxidation using toxic ruthenium tetroxide (RuO4) and sodium periodate. These processes not only introduce significant safety hazards due to the flammability and toxicity of reagents but also generate complex waste streams that are costly to treat. Furthermore, chiral resolution methods, while effective, typically suffer from a theoretical maximum yield of 50% for the desired enantiomer, leading to inefficient atom economy and the accumulation of unwanted levorotatory byproducts that must be disposed of or racemized, adding further complexity and cost to the supply chain.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a biocatalytic strategy that fundamentally simplifies the synthetic landscape. By employing an amidase derived from Klebsiella, the process achieves a direct deamination of amide ketoprofen under mild aqueous conditions. This biological route bypasses the need for precious metal catalysts and hazardous oxidants entirely, replacing them with a renewable enzymatic system that operates at ambient temperatures and neutral pH. The inventors have identified that this specific amidase possesses a unique active site configuration capable of distinguishing between enantiomers with extraordinary precision. This specificity allows for the production of dexketoprofen with an ee value reaching 100%, effectively eliminating the need for extensive downstream purification steps that are characteristic of less selective chemical or enzymatic methods.
Mechanistic Insights into Klebsiella Amidase Catalyzed Hydrolysis
The success of this synthetic route hinges on the precise molecular recognition capabilities of the Klebsiella-derived amidase (SEQ ID NO: 1). Unlike generic esterases which often exhibit broad substrate tolerance but poor enantioselectivity, this specific amidase interacts with the amide bond of the ketoprofen precursor in a highly stereo-specific manner. The enzyme's active site is geometrically constrained to accommodate only the specific spatial arrangement of the (S)-enantiomer precursor, facilitating the hydrolysis of the amide bond to release the free carboxylic acid while leaving the unwanted enantiomer untouched or reacting with it at a negligible rate. This kinetic resolution mechanism ensures that the resulting product stream is enriched almost exclusively with the pharmacologically active dextrorotatory form, minimizing the formation of diastereomeric impurities that could complicate regulatory approval.
Furthermore, the stability and expression efficiency of this enzyme contribute significantly to the process's viability. The patent details the successful expression of the amidase in E. coli hosts, achieving high cell densities (OD600 of 30-35) in fermentation tanks. This high expression level translates to a high catalyst loading per unit volume of reaction mixture, which is critical for driving the reaction kinetics forward despite the inherent equilibrium limitations of hydrolysis reactions. The ability to use the enzyme in various forms, including wet cells, spray-dried powder, or freeze-dried powder, provides flexibility in process design. For instance, the dried powder forms offer enhanced shelf-life and ease of transport, allowing for decoupled production of the biocatalyst and the final API intermediate, a logistical advantage that strengthens supply chain resilience against fermentation batch failures.
How to Synthesize Dexketoprofen Efficiently
The synthesis of dexketoprofen via this enzymatic pathway is designed for operational simplicity and scalability, making it an attractive option for contract development and manufacturing organizations (CDMOs). The process begins with the preparation of the biocatalyst through controlled fermentation, followed by a straightforward biphasic reaction system where the substrate is introduced. The reaction conditions are mild, requiring only a phosphate buffer and moderate heating, which reduces energy consumption compared to high-temperature chemical processes. For a comprehensive understanding of the specific operational parameters, including exact substrate-to-catalyst ratios and induction protocols, please refer to the standardized synthesis guide below.
- Prepare the biocatalyst by fermenting E. coli transformed with the Klebsiella amidase gene (SEQ ID NO: 1) to obtain either wet cells or dried enzyme powder.
- Suspend the amidase catalyst in a phosphate buffer (pH 7.5) and add the substrate, amide ketoprofen, dissolved in an organic co-solvent like toluene.
- Maintain the reaction at 30°C for approximately 10 to 108 hours depending on the catalyst form, then terminate with acid and extract the product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this Klebsiella amidase technology offers compelling advantages that directly address the pain points of procurement managers and supply chain heads. The transition from chemical to enzymatic synthesis eliminates the dependency on volatile and expensive noble metal catalysts, thereby insulating the production cost from fluctuations in the commodities market. Additionally, the simplified downstream processing required to achieve high purity reduces the consumption of solvents and chromatography media, leading to substantial cost savings in materials and waste disposal. The robustness of the fermentation process ensures a consistent and reliable supply of the biocatalyst, mitigating the risks associated with batch-to-batch variability that often plague complex chemical syntheses.
- Cost Reduction in Manufacturing: The elimination of toxic heavy metal catalysts such as ruthenium removes the necessity for expensive metal scavenging and removal steps, which are often bottlenecks in API manufacturing. This simplification of the purification train significantly lowers the overall cost of goods sold (COGS) by reducing both material inputs and processing time. Furthermore, the high enantioselectivity minimizes the loss of valuable starting materials to unwanted byproducts, improving the overall atom economy and reducing the raw material burden on the procurement budget.
- Enhanced Supply Chain Reliability: The ability to produce the enzyme via fermentation in standard E. coli hosts ensures that the biocatalyst can be manufactured at scale with high reproducibility. Unlike natural extraction methods which are subject to seasonal or biological variations, this recombinant approach guarantees a steady supply of the critical processing aid. The stability of the enzyme in dried powder forms also facilitates easier logistics and inventory management, allowing manufacturers to stockpile the catalyst without significant degradation, thus ensuring continuity of supply even during upstream disruptions.
- Scalability and Environmental Compliance: The process operates under mild aqueous conditions, drastically reducing the generation of hazardous organic waste and volatile organic compounds (VOCs) associated with traditional organic synthesis. This aligns perfectly with increasingly stringent global environmental regulations, reducing the regulatory burden and potential fines associated with waste discharge. The scalability demonstrated in the patent, moving from shake flasks to 50L fermenters, indicates a clear path to industrial-scale production, enabling manufacturers to meet large-volume demands without compromising on green chemistry principles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this biocatalytic route. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances is essential for evaluating the feasibility of integrating this technology into existing manufacturing workflows.
Q: What is the primary advantage of using Klebsiella amidase over traditional chemical synthesis for dexketoprofen?
A: The primary advantage is the elimination of toxic heavy metal catalysts like RuO4 and the avoidance of hazardous oxidants, resulting in a significantly greener process with easier downstream purification and reduced environmental compliance costs.
Q: How does the ee value of this enzymatic method compare to chiral resolution techniques?
A: This enzymatic method achieves an ee value of up to 100%, which is superior to many traditional chiral resolution methods that often struggle with lower optical purity and require multiple recrystallization steps to achieve similar standards.
Q: Can this process be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the patent demonstrates successful fermentation in a 50L tank and the use of stable spray-dried or freeze-dried enzyme powders, indicating strong potential for commercial scale-up and long-term storage stability required for supply chain reliability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dexketoprofen Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the biocatalytic synthesis route described in CN111363736A for the production of high-value pharmaceutical intermediates. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are seamlessly translated into robust industrial processes. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced fermentation capabilities, allowing us to meet stringent purity specifications required for global regulatory markets. We are committed to delivering dexketoprofen and related intermediates with the highest levels of quality and consistency.
We invite potential partners to engage with our technical team to explore how this enzymatic technology can be optimized for your specific supply chain needs. By collaborating with us, you can leverage our expertise to conduct a Customized Cost-Saving Analysis tailored to your production volumes. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your next project is built on a foundation of scientific excellence and commercial viability.
