Scalable Enzymatic Production of Dexketoprofen Intermediates via Novel Rhodococcus Amidase Technology
Scalable Enzymatic Production of Dexketoprofen Intermediates via Novel Rhodococcus Amidase Technology
The pharmaceutical industry is continuously seeking more efficient and environmentally sustainable pathways for the production of chiral non-steroidal anti-inflammatory drugs (NSAIDs), and the recent disclosure in patent CN116121310A represents a significant leap forward in this domain. This patent details a groundbreaking method for synthesizing dexketoprofen, the pharmacologically active (S)-enantiomer of ketoprofen, utilizing a highly specific amidase derived from Rhodococcus erythropolis CCM2595. Unlike traditional chemical synthesis routes that often rely on toxic heavy metal catalysts and harsh reaction conditions, this biocatalytic approach leverages the exquisite stereoselectivity of enzymes to produce the desired isomer with exceptional purity. The technical breakthrough lies in the enzyme's ability to tolerate high substrate loads while maintaining a remarkably low enzyme-to-substrate ratio, addressing two of the most persistent bottlenecks in industrial biocatalysis. For R&D directors and process chemists, this technology offers a robust alternative that simplifies downstream processing and eliminates the need for complex chiral resolution steps typically associated with racemic mixtures.
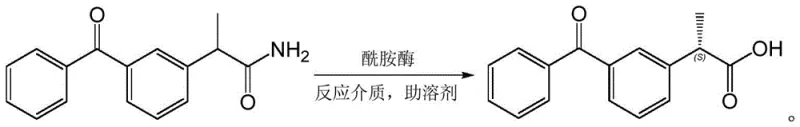
The core innovation described in the patent involves the selective hydrolysis of ketoprofen amide to yield dexketoprofen, a transformation that is visually represented in the reaction scheme above. The amidase employed in this process exhibits remarkable catalytic activity and regioselectivity, specifically targeting the amide bond to release the carboxylic acid functionality while preserving the chiral center with high fidelity. This specificity is crucial because the therapeutic efficacy of ketoprofen resides almost exclusively in the (S)-enantiomer, while the (R)-enantiomer is largely inactive and can contribute to unwanted side effects. By employing this enzymatic route, manufacturers can bypass the inefficiencies of classical resolution techniques, which theoretically discard 50% of the material as the unwanted enantiomer. Furthermore, the patent highlights that the enzyme maintains its structural integrity and catalytic function even in the presence of organic co-solvents like dimethyl sulfoxide (DMSO), which is essential for solubilizing the hydrophobic ketoprofen amide substrate in the aqueous reaction medium.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of dexketoprofen has been plagued by significant technical and economic challenges inherent to both chemical asymmetric synthesis and earlier generations of enzymatic resolution. Chemical asymmetric synthesis typically necessitates the use of expensive chiral ligands and transition metal catalysts, which not only drive up raw material costs but also introduce the risk of heavy metal contamination in the final active pharmaceutical ingredient (API). Removing these trace metals to meet regulatory limits requires additional purification steps, such as specialized adsorption or crystallization, which further erode overall yield and increase waste generation. On the other hand, earlier enzymatic methods using lipases, esterases, or less efficient amidases suffered from low substrate tolerance, often requiring dilute reaction conditions with substrate concentrations below 60g/L. These low concentrations result in massive reactor volumes for a given output, drastically reducing space-time yield and increasing energy consumption for heating, cooling, and agitation. Additionally, many prior art enzymes required high enzyme loading ratios, sometimes approaching 1:1 relative to the substrate, making the cost of the biocatalyst prohibitive for large-scale commercial application.
The Novel Approach
The methodology presented in patent CN116121310A fundamentally disrupts these limitations by introducing an amidase variant that combines high catalytic turnover with exceptional stability under process-relevant conditions. The most striking advantage of this novel approach is the ability to operate at substrate concentrations as high as 100g/L, with the system demonstrating tolerance up to 150g/L, which is more than double the capacity of many previously reported enzymatic systems. This increase in concentration directly translates to a reduction in solvent usage and reactor footprint, allowing manufacturers to produce significantly more product per batch without expanding their physical infrastructure. Moreover, the process utilizes an optimized enzyme-to-substrate mass ratio of 1/20, which is substantially lower than the ratios required by competing technologies. This efficiency means that less biological material is needed to drive the reaction to completion, lowering the cost of goods sold (COGS) and simplifying the supply chain for enzyme procurement. The combination of high concentration and low enzyme loading creates a synergistic effect that enhances the overall economic viability of the process, making it a compelling candidate for industrial adoption.
Mechanistic Insights into Rhodococcus erythropolis CCM2595 Amidase Catalysis
From a mechanistic perspective, the success of this synthesis relies on the unique structural properties of the amidase derived from Rhodococcus erythropolis CCM2595, which possesses a binding pocket exquisitely tuned to accommodate the bulky aromatic structure of ketoprofen amide. The enzyme functions through a nucleophilic attack on the carbonyl carbon of the amide group, facilitated by a catalytic triad that stabilizes the transition state and ensures the stereochemical integrity of the alpha-carbon is maintained throughout the hydrolysis. Unlike non-specific hydrolases that might promiscuously attack other functional groups or exhibit poor enantioselectivity, this specific amidase demonstrates a profound preference for the pro-chiral environment of the substrate, leading to the exclusive formation of the (S)-enantiomer. The patent data indicates that the enzyme's active site is robust enough to withstand the presence of DMSO, a polar aprotic solvent used to enhance substrate solubility, without undergoing denaturation or significant loss of activity. This solvent tolerance is a critical mechanistic feature, as it allows for a homogeneous or semi-homogeneous reaction phase that improves mass transfer kinetics compared to heterogeneous slurry reactions often seen in water-only systems.
Furthermore, the impurity profile of the resulting dexketoprofen is tightly controlled by the enzyme's inherent selectivity, which minimizes the formation of side products such as over-hydrolyzed species or racemized byproducts. The high optical purity of >99.5% ee reported in the examples suggests that the enzyme effectively discriminates between the enantiomers of the substrate or selectively hydrolyzes only the desired prodrug form without affecting the chiral center. This level of control is vital for regulatory compliance, as impurities in chiral drugs can have unpredictable pharmacological effects. The reaction conditions, specifically the maintenance of pH around 8.0 and temperature at 40°C, are optimized to balance the enzyme's catalytic rate with its stability, preventing thermal degradation while ensuring rapid conversion. Understanding these mechanistic nuances allows process engineers to fine-tune parameters such as agitation speed and feed rates to maximize the reaction efficiency, ensuring that the theoretical benefits observed in the lab scale can be reliably translated to multi-ton production scales.
How to Synthesize Dexketoprofen Efficiently
Implementing this synthesis route requires a systematic approach to biocatalyst preparation and reaction engineering to fully capitalize on the enzyme's high performance characteristics. The process begins with the fermentation of the engineered E. coli strain to produce the amidase, followed by careful formulation into a stable enzyme preparation suitable for industrial use. Once the biocatalyst is ready, the reaction is conducted in a buffered aqueous system supplemented with a co-solvent to ensure the ketoprofen amide remains in solution throughout the conversion. The following guide outlines the critical operational parameters derived from the patent examples, providing a roadmap for R&D teams to replicate and optimize this high-yield pathway in their own facilities. Adhering to these standardized steps ensures consistent product quality and maximizes the economic advantages of the low enzyme-loading protocol.
- Construct genetically engineered E. coli containing the Rhodococcus erythropolis CCM2595 amidase gene and induce expression to prepare the biocatalyst.
- Prepare the enzyme formulation (powder, liquid, or immobilized) and suspend in a reaction medium containing Tris-HCl buffer and DMSO co-solvent.
- Add ketoprofen amide substrate to achieve concentrations up to 100g/L, maintain pH at 8.0 and temperature at 40°C to achieve >99.5% ee.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this enzymatic technology offers tangible benefits that extend beyond mere technical feasibility, directly impacting the bottom line through reduced operational expenditures and enhanced supply security. The primary driver of cost reduction is the drastic decrease in enzyme consumption; by operating at an enzyme-to-substrate ratio of 1/20, the process significantly lowers the recurring cost of biocatalysts compared to methods requiring stoichiometric or near-stoichiometric amounts. This efficiency also reduces the volume of biological waste generated per kilogram of product, simplifying waste treatment protocols and lowering disposal fees. Furthermore, the ability to run reactions at high substrate concentrations means that existing manufacturing assets can be utilized more intensively, effectively increasing capacity without the need for capital-intensive expansion of reactor farms. This intensification of production allows companies to respond more agilely to market demand fluctuations, ensuring a steady supply of this critical pharmaceutical intermediate even during periods of high global demand.
- Cost Reduction in Manufacturing: The economic model of this process is heavily favored by the low enzyme loading requirement, which decouples production costs from the volatile pricing of specialized biocatalysts. By minimizing the amount of enzyme needed per batch, manufacturers can achieve a substantial reduction in variable costs, making the final dexketoprofen intermediate more price-competitive in the global market. Additionally, the elimination of heavy metal catalysts removes the need for expensive scavenging resins and rigorous metal testing, further streamlining the cost structure. The simplified downstream processing, resulting from high conversion rates and clean reaction profiles, also contributes to lower utility and labor costs per unit of output.
- Enhanced Supply Chain Reliability: The robustness of the Rhodococcus erythropolis CCM2595 amidase ensures consistent batch-to-batch performance, reducing the risk of production failures that can disrupt supply chains. The enzyme's high tolerance to substrate and product inhibition means that the process is less sensitive to minor variations in feed quality or reaction conditions, providing a safety margin that enhances operational reliability. This stability allows for longer campaign runs and reduces the frequency of reactor turnarounds, ensuring a continuous flow of material to downstream formulation units. For supply chain heads, this predictability is invaluable for maintaining inventory levels and meeting delivery commitments to pharmaceutical customers.
- Scalability and Environmental Compliance: From an environmental standpoint, this green chemistry approach aligns perfectly with increasingly stringent global regulations regarding solvent use and hazardous waste. The aqueous-based system with minimal organic co-solvent reduces the volatile organic compound (VOC) emissions associated with traditional chemical synthesis. The high space-time yield achieved through concentrated reactions means less energy is consumed for heating and cooling large volumes of solvent, contributing to a lower carbon footprint for the manufacturing site. These sustainability credentials not only facilitate regulatory approval but also enhance the brand value of the manufacturer as a responsible supplier of pharmaceutical ingredients.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating the viability of this technology for their specific applications, we have compiled a set of frequently asked questions based on the detailed experimental data provided in the patent literature. These answers address common concerns regarding scalability, purity specifications, and process robustness, offering clarity on how this enzymatic route compares to established industry standards. Understanding these technical details is essential for conducting a thorough feasibility study and for engaging in informed discussions with potential technology partners or licensors.
Q: What is the optical purity achieved with this amidase method?
A: The process consistently achieves an optical purity (ee value) greater than 99.5%, which is critical for meeting stringent pharmaceutical specifications for dexketoprofen.
Q: How does the substrate concentration compare to traditional enzymatic methods?
A: This novel amidase allows for substrate concentrations up to 150g/L, significantly higher than the 40-60g/L typical of prior art, enabling much higher space-time yields.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the low enzyme-to-substrate ratio of 1/20 and high tolerance to product inhibition make this method highly cost-effective and scalable for commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dexketoprofen Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced biocatalytic routes like the one described in patent CN116121310A for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes are seamlessly translated into robust industrial operations. Our facility is equipped with state-of-the-art fermentation and enzymatic reaction capabilities, allowing us to implement complex biocatalytic syntheses with stringent purity specifications and rigorous QC labs to guarantee product consistency. We understand that the transition from chemical to enzymatic manufacturing requires specialized expertise, and our team of process chemists and engineers is dedicated to optimizing every parameter to maximize yield and minimize cost for our clients.
We invite pharmaceutical companies and chemical distributors to explore the commercial opportunities presented by this high-efficiency dexketoprofen synthesis. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this enzymatic route for your supply chain. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your volume requirements. Whether you are looking to secure a long-term supply of high-purity dexketoprofen or need assistance in developing a proprietary manufacturing process, NINGBO INNO PHARMCHEM is committed to delivering solutions that drive value and innovation in the global pharmaceutical market.
