Revolutionizing Sitagliptin Intermediate Production with Advanced AcATA Mutant Biocatalysis
The pharmaceutical industry is constantly seeking more efficient and sustainable pathways for the production of critical active pharmaceutical ingredients (APIs) and their precursors. A significant breakthrough in this domain is documented in patent CN111549008B, which discloses a novel amine transaminase AcATA mutant and its specific application in the preparation of sitagliptin intermediates. This technology addresses the longstanding challenges associated with the stereoselective synthesis of chiral amines, which are essential building blocks for dipeptidyl peptidase-4 (DPP-4) inhibitors used in treating type II diabetes. The patent details a protein engineering approach where specific amino acid residues, particularly at position 122, are mutated to drastically enhance catalytic efficiency. By leveraging molecular docking and homologous modeling, researchers have identified that mutating methionine to histidine, valine, or phenylalanine at this critical site results in an enzyme variant with activity levels surpassing 460 U/g. This represents a substantial leap forward compared to wild-type enzymes, offering a robust solution for industrial-scale biocatalysis that aligns with modern green chemistry principles and stringent regulatory requirements for pharmaceutical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the chemical synthesis of sitagliptin and its intermediates has relied heavily on transition metal-catalyzed asymmetric hydrogenation or chiral auxiliary-induced reactions, which present significant operational and economic drawbacks for large-scale manufacturing. Traditional routes often necessitate the use of expensive noble metal catalysts such as ruthenium phosphides or platinum, which not only inflate raw material costs but also introduce complex downstream processing requirements to remove trace metal impurities to meet pharmacopeial standards. Furthermore, these chemical methods frequently involve harsh reaction conditions, including high pressures and temperatures, and may utilize toxic reagents like cyanides or diazotization agents that pose severe environmental and safety risks. The multi-step nature of these conventional syntheses often leads to cumulative yield losses, requiring extensive purification steps such as recrystallization to achieve the necessary optical purity, thereby extending production lead times and increasing the overall carbon footprint of the manufacturing process. These limitations create a bottleneck for reliable sitagliptin intermediate suppliers who must balance cost efficiency with the rigorous quality control demanded by global health authorities.
The Novel Approach
In stark contrast to these legacy chemical processes, the biocatalytic strategy outlined in the patent utilizes a highly engineered AcATA mutant to perform a direct asymmetric transamination, streamlining the synthesis into a more efficient and environmentally benign workflow. This novel approach capitalizes on the inherent stereoselectivity of omega-transaminases to construct the chiral center with exceptional precision, achieving enantiomeric excess (ee) values greater than 99% without the need for cumbersome resolution steps. The use of isopropylamine as an inexpensive amino donor drives the equilibrium towards the desired product, while the engineered enzyme's enhanced stability allows the reaction to proceed effectively in the presence of organic cosolvents required to solubilize the hydrophobic ketone substrate. By shifting from heavy metal catalysis to a biological system, manufacturers can eliminate the risk of metal contamination entirely, simplifying the purification train and significantly reducing the environmental burden associated with waste disposal. This paradigm shift not only enhances the sustainability profile of the production facility but also offers a more predictable and scalable route for cost reduction in pharmaceutical intermediates manufacturing, ensuring a stable supply of high-quality materials for the global market.
Mechanistic Insights into AcATA Mutant-Catalyzed Transamination
The core of this technological advancement lies in the precise structural modification of the amine transaminase AcATA derived from Arthrobacter cumminsii, where rational design techniques were employed to optimize the enzyme's active site for the specific steric and electronic demands of the sitagliptin precursor ketone. Through homology modeling based on the crystal structure of AcATA (PDB-ID: 3WWJ) and subsequent substrate docking simulations using Autodock software, researchers identified position 122 as a critical determinant of catalytic activity. The wild-type methionine residue at this position creates a steric or electronic environment that limits substrate binding or turnover; however, mutating this residue to histidine (M122H), valine (M122V), or phenylalanine (M122F) alters the local microenvironment to facilitate more efficient interaction with the bulky 2,4,5-trifluorophenyl group of the substrate. The M122H mutant, in particular, exhibits an enzyme activity of up to 610 U/g, which is approximately 5.5 times that of the wild type, demonstrating how a single point mutation can dramatically reshape the kinetic profile of the biocatalyst. This enhanced activity translates directly to process intensification, allowing for higher substrate loading and faster reaction rates, which are critical parameters for achieving commercial viability in industrial biotechnology applications.
The reaction mechanism involves the transfer of an amino group from isopropylamine to the prochiral ketone substrate, 1-piperidine-4-(2,4,5-trifluorophenyl)-1,3-dibutanone, mediated by the pyridoxal phosphate (PLP) cofactor bound within the enzyme's active site. As illustrated in the reaction scheme below, the enzyme facilitates the formation of the chiral amine product, (R)-3-amino-1-piperidine-4-(2,4,5-trifluorophenyl)-1-butanone, while converting isopropylamine into acetone as a byproduct. The removal of acetone, often driven by its volatility or through equilibrium shifts, helps push the reaction towards completion, achieving conversion rates as high as 90% within just 24 hours under optimized conditions. This high level of conversion is maintained even at relatively high substrate concentrations (e.g., 50 g/L), indicating that the mutant enzyme possesses robust tolerance to substrate inhibition and organic solvents like DMSO. Such mechanistic robustness ensures that the process remains consistent and reliable, minimizing the formation of unwanted byproducts and simplifying the downstream isolation of the pure chiral intermediate.
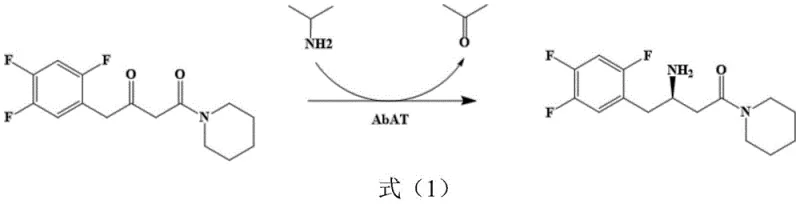
How to Synthesize Sitagliptin Intermediate Efficiently
Implementing this advanced biocatalytic route requires careful attention to reaction parameters to fully leverage the capabilities of the AcATA mutant, ensuring that the theoretical benefits observed in the laboratory are realized in production environments. The process begins with the preparation of the biocatalyst, typically involving the cultivation of recombinant Escherichia coli expressing the mutant enzyme, followed by cell harvesting and optional purification depending on the specific process design requirements. The reaction is conducted in a buffered aqueous system, typically using triethanolamine buffer adjusted to a pH between 8.0 and 9.0, which provides the optimal protonation state for the enzymatic machinery. To address the limited solubility of the fluorinated ketone substrate in water, a cosolvent such as dimethyl sulfoxide (DMSO) or methyl tert-butyl ether (MTBE) is introduced, usually at a volume concentration of around 20%, which significantly enhances mass transfer without denaturing the enzyme. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction system using the AcATA mutant (e.g., M122H) as the biocatalyst, with isopropylamine as the amino donor and pyridoxal phosphate as the coenzyme in a buffered solution.
- Maintain the reaction temperature between 45-50°C and adjust the pH to 8.0-9.0, ensuring the addition of a cosolvent like DMSO to improve substrate solubility.
- Monitor the conversion rate via HPLC over 24 hours, aiming for greater than 90% conversion, followed by extraction and purification to isolate the high-purity chiral amine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this AcATA mutant technology presents a compelling value proposition that extends beyond mere technical feasibility to tangible economic and operational benefits. The transition from chemical synthesis to this enzymatic process fundamentally alters the cost structure of sitagliptin intermediate production by removing the dependency on volatile and expensive precious metal markets. Since the process does not utilize ruthenium or platinum catalysts, the risk of cost fluctuations associated with these commodities is eliminated, leading to more stable and predictable pricing models for long-term supply contracts. Furthermore, the high conversion rates and stereoselectivity reduce the need for extensive chromatographic purification or multiple recrystallization steps, which are often the most resource-intensive parts of pharmaceutical manufacturing. This simplification of the downstream processing train results in substantial cost savings by reducing solvent consumption, energy usage, and labor hours, thereby improving the overall margin profile for the manufacturer and allowing for more competitive pricing strategies in the global marketplace.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and chiral auxiliaries significantly lowers the direct material costs associated with the synthesis, while the high enzyme activity reduces the amount of biocatalyst required per kilogram of product. Additionally, the ability to operate at moderate temperatures and atmospheric pressure reduces energy consumption compared to high-pressure hydrogenation processes, contributing to a leaner and more cost-effective manufacturing operation that maximizes return on investment for production assets.
- Enhanced Supply Chain Reliability: By utilizing a biocatalytic route that relies on renewable biological resources rather than finite petrochemical-derived reagents or scarce metals, the supply chain becomes more resilient to geopolitical disruptions and raw material shortages. The robustness of the engineered enzyme allows for consistent batch-to-batch performance, minimizing the risk of production delays caused by failed reactions or off-specification products, which is critical for maintaining the continuity of supply for downstream API manufacturers who operate on tight just-in-time schedules.
- Scalability and Environmental Compliance: The process is inherently scalable, as demonstrated by the successful translation of laboratory results to conditions suitable for industrial fermentation and biotransformation, ensuring that supply volumes can be ramped up to meet market demand without compromising quality. Moreover, the green nature of the process, characterized by the absence of toxic heavy metals and the use of aqueous systems, simplifies regulatory compliance and waste management, reducing the environmental liability and permitting hurdles often associated with traditional chemical synthesis facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this AcATA mutant technology, providing clarity for stakeholders evaluating its potential integration into their supply chains. These insights are derived directly from the experimental data and technical specifications disclosed in the patent literature, ensuring that the information provided is accurate and grounded in scientific evidence. Understanding these details is crucial for making informed decisions about process adoption and supplier qualification.
Q: What represents the key improvement of the AcATA mutant over the wild type?
A: The AcATA mutant, specifically at position 122 (e.g., M122H), demonstrates enzyme activity exceeding 460 U/g, which is more than four times higher than the wild type, enabling significantly faster reaction kinetics and higher conversion rates up to 90% within 24 hours.
Q: How does this biocatalytic route address environmental concerns in sitagliptin synthesis?
A: Unlike traditional chemical synthesis which often relies on expensive and toxic heavy metal catalysts like ruthenium or platinum, this enzymatic method operates under mild aqueous conditions, eliminating heavy metal residues and reducing hazardous waste generation.
Q: What are the optimal reaction conditions for maximizing yield with this mutant?
A: Optimal performance is achieved at temperatures between 45-50°C with a pH of 8.0-9.0, using dimethyl sulfoxide (DMSO) or methyl tert-butyl ether (MTBE) as cosolvents to enhance substrate solubility while maintaining enzyme stability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sitagliptin Intermediate Supplier
As the global demand for high-quality diabetes medications continues to rise, securing a dependable source of key intermediates like the sitagliptin precursor is paramount for pharmaceutical companies aiming to maintain market share and operational efficiency. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging advanced biocatalytic technologies such as the AcATA mutant described in CN111549008B to deliver superior products that meet the rigorous standards of international regulatory bodies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can support your needs whether you are in the clinical trial phase or full-scale commercial manufacturing. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of sitagliptin intermediate we supply is free from harmful impurities and ready for immediate use in your API synthesis workflows.
We invite you to explore how our innovative manufacturing capabilities can drive value for your organization by reducing costs and enhancing supply security. Our technical sales team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality expectations, demonstrating exactly how switching to our biocatalytic route can improve your bottom line. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing you to validate our capabilities and establish a partnership built on transparency, quality, and mutual success in the competitive pharmaceutical landscape.
