Advanced Manufacturing of Sitagliptin Intermediates via Optimized Activation-Condensation Strategies
Introduction to Next-Generation Sitagliptin Manufacturing
The pharmaceutical landscape for Type II diabetes treatment continues to evolve, with Sitagliptin (Januvia) remaining a cornerstone therapy as a potent DPP-IV inhibitor. As global demand for this critical API persists, the efficiency of its supply chain becomes paramount for generic manufacturers and innovators alike. Recent intellectual property developments, specifically Chinese Patent CN114891004A, disclose a transformative preparation method for Sitagliptin intermediate compounds that addresses long-standing bottlenecks in amide bond formation. This technology shifts the paradigm from traditional, reagent-heavy coupling strategies to a streamlined activation-condensation protocol. By leveraging mixed anhydride intermediates generated in situ, the process achieves superior reaction kinetics and purity profiles. For procurement leaders and technical directors, understanding this shift is vital for securing a reliable sitagliptin intermediate supplier capable of delivering cost-effective, high-volume production without compromising on quality standards.
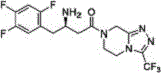
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the key amide bond in Sitagliptin has relied heavily on peptide coupling reagents such as 1-hydroxybenzotriazole (HOBt) and 1-ethyl-(3-dimethylaminopropyl)carbodiimide (EDC). While effective on a laboratory scale, these reagents present significant challenges for industrial application. The cost of HOBt and EDC is substantially higher than commodity chemicals, directly inflating the Cost of Goods Sold (COGS). Furthermore, the removal of urea byproducts and residual coupling agents requires complex workup procedures, often involving multiple crystallization steps that erode overall yield. Alternative routes utilizing chlorosulfonyl esters have been explored but introduce severe safety liabilities due to the potential genotoxicity of the sulfonyl species. These impurities necessitate rigorous analytical controls and specialized equipment to prevent cross-contamination, creating a barrier to entry for many contract development and manufacturing organizations (CDMOs).
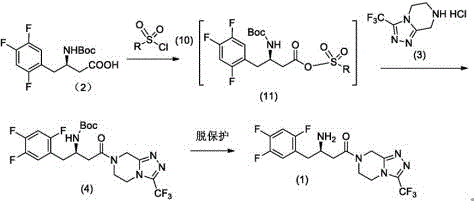
The Novel Approach
The methodology outlined in CN114891004A offers a decisive break from these constraints by employing activation reagents such as ethyl chloroformate or oxalyl chloride. This strategy generates a highly reactive mixed anhydride intermediate under mild conditions, typically between -25°C and -20°C, which then undergoes rapid nucleophilic attack by the triazolopyrazine amine component. This approach not only circumvents the use of expensive coupling additives but also avoids the formation of difficult-to-remove urea derivatives. The result is a cleaner reaction profile that facilitates easier downstream processing. By replacing hazardous chlorosulfonyl reagents with safer alternatives, the process inherently lowers the toxicological risk profile of the manufacturing line. This technological leap ensures that cost reduction in API manufacturing is achieved not just through cheaper inputs, but through a fundamentally more robust and scalable chemical architecture.
Mechanistic Insights into Mixed Anhydride Activation
At the heart of this innovation lies the precise control of the activation step. The reaction begins with the treatment of the beta-amino acid precursor (or keto acid derivative) with a base, such as N,N-diisopropylethylamine, followed by the dropwise addition of ethyl chloroformate. This generates a mixed carbonic-carboxylic anhydride, a species that possesses excellent electrophilicity towards amines but remains stable enough at low temperatures to prevent racemization or side reactions. The subsequent addition of the heterocyclic amine triggers the condensation, forming the critical amide bond with high stereochemical integrity. The patent data highlights that maintaining the temperature window of -25°C to -20°C is crucial for maximizing the molar yield, which has been demonstrated to reach 90.9% in optimized examples. This level of control minimizes the formation of N-acylurea byproducts or symmetrical anhydrides, which are common pitfalls in less controlled activation protocols.
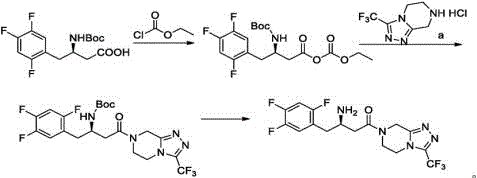
Furthermore, the mechanism allows for flexible deprotection strategies depending on the protecting group employed on the amino moiety. Whether utilizing a Boc (tert-butyloxycarbonyl) group or a benzyl group, the final cleavage can be achieved efficiently using acidic conditions. For instance, the use of hydrogen chloride in isopropanol effectively removes the Boc group, followed by salt formation with phosphoric acid to yield the final phosphate monohydrate. This modularity ensures that the process can be adapted to various starting materials while maintaining high purity specifications. The ability to drive the reaction to completion with minimal excess reagents also contributes to a greener manufacturing footprint, aligning with modern environmental compliance standards required by top-tier pharmaceutical buyers.
How to Synthesize Sitagliptin Intermediate Efficiently
Implementing this synthesis route requires careful attention to thermal management and reagent stoichiometry to replicate the high yields reported in the patent literature. The process generally involves three critical stages: the activation of the acid component, the condensation with the amine, and the final deprotection and salt formation. Operators must ensure that the activation step is performed under inert atmosphere to prevent moisture ingress, which could hydrolyze the mixed anhydride. Detailed standard operating procedures (SOPs) regarding the rate of addition for the activating agent and the maintenance of cryogenic temperatures are essential for success. For a comprehensive breakdown of the specific experimental parameters, solvent choices, and workup techniques, please refer to the standardized guide below.
- Activate the beta-amino acid or keto acid precursor using ethyl chloroformate or oxalyl chloride at low temperatures (-25°C to -20°C) to form a reactive mixed anhydride intermediate.
- Perform the condensation reaction by adding the triazolopyrazine amine component to the activated solution, maintaining strict temperature control to ensure high conversion rates.
- Execute the final deprotection step using acidic conditions (e.g., HCl in isopropanol) followed by salt formation with phosphoric acid to yield the final Sitagliptin phosphate monohydrate.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain directors and procurement managers, the adoption of this novel synthesis route translates into tangible strategic benefits beyond simple chemistry. The shift away from proprietary or high-cost coupling reagents towards commodity chemicals like ethyl chloroformate drastically simplifies the sourcing landscape. This reduces dependency on single-source suppliers for specialized reagents, thereby enhancing supply chain reliability and mitigating the risk of production stoppages due to raw material shortages. Additionally, the simplified purification process means shorter cycle times and reduced solvent consumption, which directly impacts the operational expenditure of the manufacturing facility. These factors combined create a more resilient supply chain capable of meeting the rigorous demands of the global diabetes medication market.
- Cost Reduction in Manufacturing: The elimination of expensive coupling agents like HOBt and EDC results in significant raw material savings. Furthermore, the high reaction yield (>90%) minimizes the loss of valuable intermediates, ensuring that every kilogram of input material is converted efficiently into product. This efficiency extends to waste disposal costs, as the absence of complex urea byproducts reduces the volume of hazardous waste requiring treatment, leading to substantial cost savings in environmental compliance.
- Enhanced Supply Chain Reliability: By utilizing widely available activation reagents, manufacturers can secure long-term contracts with multiple chemical vendors, ensuring continuity of supply even during market fluctuations. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, reducing the frequency of batch failures. This stability is crucial for maintaining consistent inventory levels and meeting Just-In-Time delivery schedules required by major pharmaceutical clients.
- Scalability and Environmental Compliance: The process is designed with industrial scale-up in mind, avoiding the use of potentially explosive or highly toxic reagents that complicate large-scale operations. The lower E-factor (environmental factor) resulting from higher yields and simpler workups aligns with corporate sustainability goals. This makes the technology attractive for companies aiming to reduce their carbon footprint while expanding production capacity to meet growing global demand for generic Sitagliptin.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer and for commercial teams negotiating supply agreements. We encourage stakeholders to review these points to gain a clearer picture of the operational advantages offered by this method.
Q: What are the primary advantages of this synthesis route over traditional HOBt/EDC methods?
A: The novel route eliminates the need for expensive coupling reagents like HOBt and EDC, which are costly and difficult to remove completely. Instead, it utilizes commodity chemicals like ethyl chloroformate, significantly reducing raw material costs and simplifying the purification process for industrial scale-up.
Q: How does this method address safety concerns related to genotoxic impurities?
A: Unlike prior art methods that utilize chlorosulfonyl esters, which pose potential genotoxicity risks and require stringent control limits, this invention employs safer activation reagents. This reduces the regulatory burden regarding mutagenic impurity testing and enhances the overall safety profile of the manufacturing process.
Q: What yields can be expected from this activation-condensation protocol?
A: Experimental data from the patent indicates exceptionally high efficiency, with molar yields reaching up to 90.9% for the condensation step and over 96% for subsequent transformations. This high efficiency minimizes waste generation and maximizes throughput in commercial production environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sitagliptin Intermediate Supplier
As the pharmaceutical industry moves towards more efficient and sustainable manufacturing practices, partnering with a CDMO that possesses deep expertise in process optimization is critical. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, offering extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is adept at implementing the activation-condensation strategies described in recent patents, ensuring that your supply of high-purity Sitagliptin intermediates is both secure and cost-competitive. With our rigorous QC labs and commitment to stringent purity specifications, we guarantee that every batch meets the exacting standards required for regulatory submission and commercial distribution.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific production needs. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this superior manufacturing method. We are ready to provide specific COA data and route feasibility assessments to support your decision-making process, ensuring a seamless transition to a more efficient and reliable supply chain partner.
