Advanced Biocatalytic Production of Sitagliptin Intermediates Using Engineered Omega-Transaminase Mutants
The pharmaceutical industry is currently witnessing a paradigm shift towards greener, more efficient synthetic routes for complex chiral intermediates, particularly for blockbuster drugs like Sitagliptin. A pivotal development in this domain is documented in Chinese Patent CN110540975B, which discloses a novel omega-transaminase mutant capable of catalyzing the asymmetric synthesis of key sitagliptin precursors with unprecedented efficiency. This technology addresses the critical bottlenecks of traditional chemical synthesis, such as low stereoselectivity and harsh reaction conditions, by leveraging protein engineering to optimize the active site of the enzyme. For R&D directors and procurement strategists, understanding the implications of this patent is vital, as it offers a pathway to high-purity pharmaceutical intermediates that align with modern sustainability mandates. The mutant enzymes described, specifically those with substitutions at positions 56 or 134, demonstrate a remarkable ability to accommodate bulky substrates, thereby unlocking higher conversion rates that were previously unattainable with wild-type biocatalysts.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the manufacturing of optically pure sitagliptin intermediates has relied heavily on chemical synthesis methods involving asymmetric hydrogenation or chiral resolution, both of which present significant industrial drawbacks. Traditional ruthenium-catalyzed asymmetric hydrogenation, while effective, necessitates the use of high-pressure hydrogen gas and expensive noble metal catalysts that pose severe safety risks and complicate the removal of trace metal impurities from the final API. Furthermore, chiral resolution techniques are inherently inefficient, with a theoretical maximum yield of only 50%, requiring the disposal or recycling of the unwanted enantiomer, which drastically inflates production costs and environmental waste. Other chemical routes involving Grignard reagents or cyanide substitutions introduce highly toxic reagents into the process stream, creating substantial regulatory hurdles and necessitating complex waste treatment protocols that strain operational budgets. These legacy methods often struggle to consistently achieve the stringent enantiomeric excess (ee) values required for modern diabetes therapeutics, leading to batch variability and potential quality control failures.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes engineered omega-transaminases to perform a direct asymmetric amination of the pro-chiral ketone substrate under mild, aqueous conditions. By employing site-directed mutagenesis to modify specific amino acid residues near the enzyme's active pocket, the inventors have created biocatalysts that exhibit substantially improved affinity and turnover numbers for the bulky trifluorophenyl-containing substrate. This biocatalytic route eliminates the need for high-pressure equipment and toxic heavy metals, replacing them with renewable biological catalysts and benign amino donors like isopropylamine. The process operates at moderate temperatures between 40°C and 45°C and atmospheric pressure, significantly reducing energy consumption and capital expenditure on specialized reactor infrastructure. Moreover, the enzymatic specificity ensures that the reaction proceeds with exceptional stereoselectivity, routinely achieving enantiomeric excess values greater than 99%, which simplifies downstream purification and guarantees the high quality demanded by global regulatory agencies.
Mechanistic Insights into Omega-Transaminase Catalyzed Asymmetric Amination
The core of this technological breakthrough lies in the precise structural modification of the omega-transaminase derived from Arthrobacter cumminsii, where specific mutations alter the steric and electronic environment of the catalytic center. The patent highlights that substituting phenylalanine at position 56 with valine, histidine, or tyrosine, or replacing threonine at position 134 with glycine, creates a more spacious and flexible active site pocket. This structural relaxation is crucial for accommodating the sterically demanding 2,4,5-trifluorophenyl group of the sitagliptin precursor ketone, which typically hinders binding in the wild-type enzyme. Molecular docking studies suggest that these mutations optimize the orientation of the substrate relative to the pyridoxal phosphate (PLP) cofactor, facilitating a more efficient transfer of the amino group from the donor to the ketone acceptor. The result is a dramatic increase in specific activity, with the T134G mutant exhibiting enzyme activity levels exceeding 300 U/g, which is nearly three times higher than the wild-type counterpart.
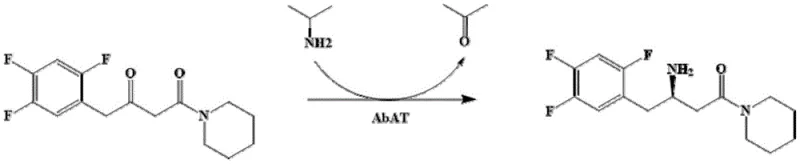
Beyond mere activity enhancement, the engineered enzyme demonstrates superior stability and tolerance to organic co-solvents, which is essential for dissolving the hydrophobic ketone substrate in the aqueous reaction medium. The mechanism involves the formation of a Schiff base intermediate between the PLP cofactor and the amino donor, followed by the transfer of the amino group to the substrate ketone to form the chiral amine product. The use of isopropylamine as the amino donor drives the equilibrium towards product formation by generating acetone as a byproduct, which can be easily removed due to its volatility. This thermodynamic driving force, combined with the kinetic improvements from the mutations, allows the reaction to reach conversion rates as high as 85% within 36 hours, a significant improvement over the 50% conversion observed with the wild-type enzyme under similar conditions. Such mechanistic efficiency translates directly into reduced reactor occupancy time and higher throughput for manufacturing facilities.
How to Synthesize Sitagliptin Intermediate Efficiently
Implementing this biocatalytic process requires a systematic approach to strain construction, fermentation, and biotransformation to ensure consistent high-yield production of the chiral amine. The protocol begins with the cloning of the mutated transaminase gene into an expression vector, followed by transformation into a robust host strain like E. coli BL21(DE3) for high-level protein expression. Once the recombinant cells are cultivated and induced, the wet biomass serves as the whole-cell biocatalyst, eliminating the need for costly enzyme purification steps while maintaining high catalytic density. The reaction is conducted in a buffered aqueous system supplemented with DMSO to enhance substrate solubility, ensuring that the mass transfer limitations do not hinder the overall reaction rate.
- Construct the recombinant vector containing the mutated omega-transaminase gene (e.g., T134G or F56V) and transform into E. coli BL21(DE3) competent cells.
- Culture the engineered bacteria in LB medium with ampicillin, induce expression with IPTG at 28°C, and harvest wet cells via centrifugation.
- Perform the biotransformation reaction using the wet cells, substrate ketone, isopropylamine as amino donor, and PLP cofactor at 40-45°C for 36-48 hours.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this enzymatic technology offers profound strategic advantages that extend beyond simple technical metrics, fundamentally altering the cost structure and risk profile of the supply chain. The elimination of expensive noble metal catalysts and high-pressure hydrogenation equipment results in a significant reduction in capital expenditure and raw material costs, making the final intermediate more price-competitive in the global market. Furthermore, the reliance on fermentation-based production allows for rapid scalability; increasing output simply requires larger fermentation tanks rather than the complex engineering modifications needed for high-pressure chemical reactors, ensuring that supply can be ramped up quickly to meet market demand fluctuations without compromising quality.
- Cost Reduction in Manufacturing: The transition from chemical catalysis to biocatalysis removes the financial burden associated with purchasing and recovering precious metal catalysts like ruthenium, which are subject to volatile market pricing. Additionally, the high conversion rates and stereoselectivity minimize the loss of raw materials and reduce the need for extensive chromatographic purification, leading to substantial savings in solvent usage and waste disposal fees. The process operates under mild conditions, which lowers energy consumption for heating and cooling, further contributing to a leaner operational cost structure that enhances overall profit margins for the final drug product.
- Enhanced Supply Chain Reliability: Biological production systems are inherently more flexible and resilient compared to rigid chemical synthesis lines, as the biocatalyst can be produced on-demand through standard fermentation processes. This reduces dependency on specialized chemical reagents that may face supply shortages or geopolitical trade restrictions, thereby securing a more stable and continuous supply of critical intermediates. The robustness of the engineered E. coli strains ensures consistent batch-to-batch performance, mitigating the risk of production delays caused by catalyst deactivation or inconsistent reaction outcomes that often plague traditional chemical methods.
- Scalability and Environmental Compliance: The aqueous nature of the biocatalytic reaction significantly reduces the generation of hazardous organic waste, simplifying compliance with increasingly stringent environmental regulations and lowering the cost of wastewater treatment. The process avoids the use of toxic cyanides and heavy metals, creating a safer working environment and reducing the liability associated with handling dangerous chemicals. This green chemistry profile not only aligns with corporate sustainability goals but also facilitates smoother regulatory approvals in markets that prioritize environmentally friendly manufacturing practices, adding intangible value to the brand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented transaminase technology in industrial settings. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making.
Q: What represents the primary advantage of the T134G mutant over the wild-type enzyme?
A: The T134G mutant exhibits significantly enhanced enzymatic activity, reaching over 300 U/g compared to approximately 110 U/g for the wild type, and achieves a substrate conversion rate of up to 85% within 36 hours, drastically reducing reaction time.
Q: How does this biocatalytic route compare to traditional ruthenium-catalyzed asymmetric hydrogenation?
A: Unlike ruthenium-catalyzed methods which require high-pressure hydrogen and expensive metal catalysts that are difficult to remove, this enzymatic process operates under mild atmospheric conditions, eliminates heavy metal contamination risks, and provides superior enantioselectivity (>99% ee).
Q: Is this process suitable for large-scale commercial manufacturing of DPP-4 inhibitor intermediates?
A: Yes, the process utilizes robust recombinant E. coli strains and standard fermentation techniques, allowing for scalable production. The use of aqueous buffer systems and readily available amino donors like isopropylamine simplifies downstream processing and waste treatment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sitagliptin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the enzymatic routes described in CN110540975B and possess the technical expertise to translate these laboratory innovations into commercial reality. As a premier CDMO partner, we have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of high-purity pharmaceutical intermediates. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced fermentation capabilities, allowing us to meet stringent purity specifications and deliver products that exceed global regulatory standards for diabetes therapeutics.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this cutting-edge biocatalytic technology for their sitagliptin supply chains. By partnering with our technical procurement team, you can obtain a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this enzymatic route for your specific production volume. We encourage you to contact us today to request specific COA data and route feasibility assessments, ensuring that your next project is built on a foundation of scientific excellence and commercial viability.
