Advanced Immobilized Transaminase Technology for Scalable Sitagliptin Intermediate Production
The pharmaceutical industry's relentless pursuit of efficient Type II diabetes treatments has placed sitagliptin at the forefront of metabolic disease management. As disclosed in Chinese Patent CN113061594A, a significant technological leap has been achieved in the biocatalytic synthesis of this critical API and its intermediates. This patent introduces a novel immobilized transaminase system that overcomes the historical limitations of enzyme stability in organic media. By utilizing specific transaminase mutants, such as those defined by SEQ ID NO.3 and SEQ ID NO.7, coupled with optimized epoxy resin supports, the process achieves exceptional stereoselectivity and operational robustness. For R&D directors and process chemists, this represents a paradigm shift from fragile liquid enzyme systems to durable, reusable biocatalysts that can withstand the rigors of industrial manufacturing environments while maintaining high enantiomeric excess.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the enzymatic synthesis of sitagliptin intermediates has been plagued by the instability of transaminases in high concentrations of organic solvents. Prior art, such as that described in US9587229, relied on water-saturated isopropyl acetate (IPAc) as the reaction medium. While effective for initial conversion, this approach introduced significant operational complexities. The requirement to pre-saturate the solvent with water added a cumbersome preprocessing step that was difficult to control consistently on a large scale. Furthermore, maintaining the precise water activity necessary to prevent enzyme denaturation during multi-batch operations proved challenging, often leading to rapid loss of catalytic activity and inconsistent product quality. These factors collectively increased the cost of goods and limited the practical scalability of the biocatalytic route for commercial production.
The Novel Approach
The technology outlined in CN113061594A fundamentally resolves these solvent compatibility issues by transitioning to an aqueous isopropanol system. This innovative approach utilizes a carefully screened solvent mixture containing 2-20% water, which provides an optimal environment for the immobilized enzyme's hydration shell without compromising substrate solubility. When combined with the specific transaminase mutants and epoxy resins like SEPABEADS® EC HFA, the system demonstrates remarkable stability. Unlike the prior art which struggled beyond a few batches, this new method supports repeated use of the immobilized catalyst for at least 10 consecutive reaction cycles with sustained high conversion rates. This transition not only simplifies the workflow by eliminating solvent pre-saturation but also drastically enhances the economic viability of the process through improved catalyst longevity.
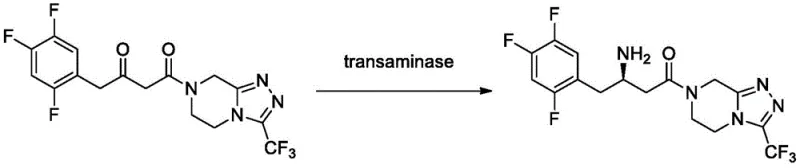
Mechanistic Insights into Transaminase-Catalyzed Asymmetric Amination
The core of this technological advancement lies in the precise protein engineering of the transaminase enzyme. The patent specifies mutations at key amino acid positions, such as M122Q and P223T, which alter the enzyme's active site geometry and surface properties. These modifications enhance the binding affinity for the bulky sitagliptin ketone substrate, specifically 4-oxo-4-[3-(trifluoromethyl)-5,6-dihydro-[1,2,4]triazolo[4,3-a]pyrazin-7(8H)-yl]-1-(2,4,5-trifluorophenyl)butan-2-one. The mechanism involves the transfer of an amino group from isopropylamine to the prochiral ketone, facilitated by the pyridoxal phosphate (PLP) cofactor. The engineered active site imposes strict steric constraints that favor the formation of the (R)-enantiomer, ensuring an ee value exceeding 99.9%, which is critical for meeting the stringent purity specifications required for pharmaceutical intermediates.
Furthermore, the immobilization strategy plays a pivotal role in preserving this mechanistic efficiency. By covalently bonding the mutant enzyme to macroporous epoxy resins, the protein structure is rigidified, preventing the conformational unfolding that typically occurs in organic solvents. This structural stabilization allows the enzyme to maintain its catalytic cycle efficiency even in the presence of high substrate concentrations up to 200 g/L. The result is a process that combines the high selectivity of biocatalysis with the robustness typically associated with heterogeneous chemical catalysis, providing a reliable pathway for the production of high-purity sitagliptin intermediates suitable for downstream phosphorylation.
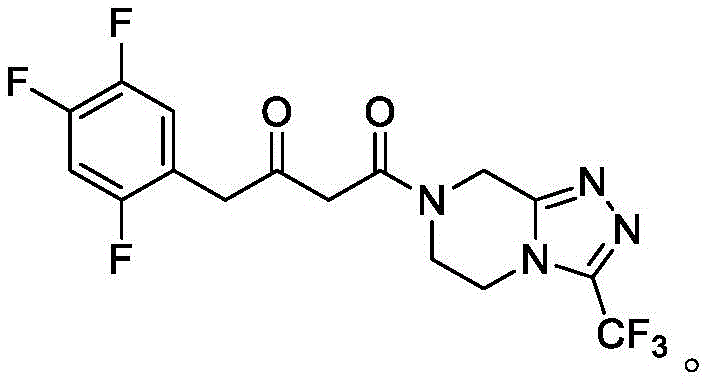
How to Synthesize Sitagliptin Intermediate Efficiently
The synthesis protocol described in the patent offers a streamlined pathway for producing chiral amines essential for sitagliptin manufacturing. The process begins with the preparation of the immobilized biocatalyst, followed by the establishment of the reaction conditions in the optimized aqueous isopropanol medium. This section outlines the critical parameters for achieving maximum yield and stereoselectivity. For laboratory and pilot-scale teams looking to implement this technology, adherence to the specific resin selection and solvent ratios is paramount to replicating the high performance reported in the patent data. The detailed standardized synthesis steps for implementing this immobilized transaminase protocol are provided in the guide below.
- Preparation of Immobilized Enzyme: Contact the transaminase mutant solution (SEQ ID NO.3 or 7) with epoxy resin (e.g., SEPABEADS EC HFA) to form covalent bonds, followed by filtration and washing.
- Reaction Setup: Suspend the immobilized enzyme in an aqueous isopropanol solution (2-20% water content) containing the ketoamide substrate, isopropylamine as amino donor, and pyridoxal phosphate cofactor.
- Catalysis and Recovery: Maintain the reaction at 30-60°C with shaking. After completion, filter to recover the immobilized enzyme for reuse and process the filtrate to isolate the chiral amine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this immobilized transaminase technology translates directly into tangible operational efficiencies and cost optimizations. The primary driver of value is the dramatic improvement in catalyst reusability. Traditional liquid enzyme processes often require fresh enzyme for every batch or suffer from rapid activity decay, leading to high recurring costs for biocatalysts. In contrast, the immobilized system described allows for the same batch of enzyme to be recovered and reused across multiple production cycles without significant loss of performance. This extended lifecycle substantially reduces the unit cost of the enzyme per kilogram of product, lowering the overall raw material expenditure for the manufacturing process.
- Cost Reduction in Manufacturing: The elimination of complex solvent preparation steps, specifically the removal of the water-saturation requirement for isopropyl acetate, streamlines the production workflow. This simplification reduces labor hours and energy consumption associated with solvent conditioning. Additionally, the ability to operate at higher substrate concentrations (up to 200 g/L) increases the volumetric productivity of the reactors, allowing manufacturers to produce more product in less time with the same equipment footprint. These factors combine to drive down the cost of goods sold (COGS) for sitagliptin intermediates, making the supply chain more resilient to price fluctuations in raw materials.
- Enhanced Supply Chain Reliability: The stability of the immobilized enzyme in the aqueous isopropanol system ensures consistent batch-to-batch quality, reducing the risk of production failures or off-spec material that can disrupt supply schedules. The robust nature of the immobilized catalyst also simplifies logistics; unlike sensitive liquid enzymes that may require cold chain storage and have short shelf lives, immobilized preparations generally offer better stability during storage and transport. This reliability allows for more flexible inventory management and reduces the safety stock requirements for critical biocatalytic components.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, the use of isopropanol, a greener solvent compared to some alternatives, aligns well with modern sustainability goals. The high conversion rates and excellent stereoselectivity minimize the formation of unwanted byproducts and reduce the burden on downstream purification processes, thereby lowering waste generation. The process is inherently scalable, as the fixed-bed or batch-wise reuse of the solid catalyst is easily adapted from pilot to commercial scale, facilitating the rapid ramp-up of production capacity to meet global demand for diabetes medications without compromising environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this immobilized transaminase technology. These answers are derived directly from the experimental data and disclosures within the patent documentation, providing clarity on the operational parameters and expected performance metrics. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this biocatalytic route into existing manufacturing lines.
Q: What are the advantages of using aqueous isopropanol over isopropyl acetate (IPAc) for this transamination?
A: The patent highlights that aqueous isopropanol eliminates the complex pre-saturation step required for IPAc, simplifies water activity control, and significantly improves the operational stability and reusability of the immobilized enzyme over multiple batches.
Q: Which specific resin types are compatible with the transaminase mutants described in CN113061594A?
A: The invention specifically identifies epoxy resins such as SEPABEADS® EC HFA, ReliZyme™ HFA403, and EP series resins as highly effective supports that maintain high enzymatic activity and stability after immobilization.
Q: How does the M122Q-P223T mutation improve the process compared to prior art enzymes?
A: This specific double mutation significantly enhances the enzyme's catalytic activity towards the bulky sitagliptin ketone substrate and improves its structural stability in organic solvent systems, allowing for higher conversion rates and extended reuse cycles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sitagliptin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced biocatalysis in the production of complex pharmaceutical intermediates like sitagliptin. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to delivering high-purity sitagliptin intermediates that meet stringent purity specifications, leveraging rigorous QC labs and state-of-the-art manufacturing facilities to guarantee consistency and quality in every batch we supply to our global partners.
We invite you to collaborate with us to optimize your supply chain for diabetes therapeutics. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in immobilized enzyme technology can drive efficiency and reliability in your sitagliptin production strategy.
