Advanced Immobilized Transaminase Mutants for Efficient Sitagliptin and Intermediate Manufacturing
The global pharmaceutical landscape continues to demand more efficient and sustainable pathways for the production of critical antidiabetic medications, particularly DPP-4 inhibitors like Sitagliptin. As the market for type II diabetes treatments expands, the pressure on supply chains to deliver high-purity active pharmaceutical ingredients (APIs) and their intermediates at scale has never been greater. In this context, the technological breakthroughs disclosed in Chinese Patent CN113061594A represent a significant leap forward in biocatalytic process engineering. This patent introduces novel transaminase mutants and their immobilized forms, specifically designed to overcome the longstanding limitations of enzyme stability and solvent compatibility in the synthesis of Sitagliptin and its key chiral intermediates. By leveraging specific amino acid mutations and advanced immobilization techniques on epoxy resins, this technology enables a robust, scalable, and cost-effective manufacturing route that addresses the core pain points of traditional chemical synthesis and earlier biocatalytic attempts.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods for synthesizing Sitagliptin, such as those disclosed in US Patents 8293507 and 9587229, have relied heavily on engineered transaminases that, while effective in free form, face severe challenges upon immobilization. A critical bottleneck in these conventional processes is the dependency on water-saturated isopropyl acetate (IPAc) as the reaction solvent. The requirement to pre-saturate IPAc with water introduces significant operational complexity and variability; controlling the exact water activity is difficult, and fluctuations can lead to rapid enzyme inactivation and denaturation. Furthermore, these earlier immobilized enzymes often exhibited poor reusability, with conversion rates dropping precipitously after just a few batches, rendering them economically unviable for continuous large-scale production. The instability of these enzymes in 100% organic solvents necessitated complex solvent systems that increased both the environmental footprint and the overall production costs, creating a barrier to entry for reliable sitagliptin intermediate suppliers seeking to optimize their manufacturing lines.
The Novel Approach
The innovation presented in Patent CN113061594A fundamentally shifts the paradigm by introducing transaminase mutants, specifically SEQ ID NO.3 (Enz.1-M122Q-P223T) and SEQ ID NO.7 (Enz.2-M122F), which retain high enzymatic activity even after immobilization. Crucially, this technology replaces the problematic IPAc system with a much simpler and more robust aqueous isopropanol solution, typically containing 2-20% water by volume. This solvent switch eliminates the need for tedious water saturation procedures, drastically simplifying the operational workflow. The immobilized enzymes demonstrate exceptional stability, maintaining conversion rates above 85% over ten consecutive batches, a stark contrast to the rapid degradation seen in prior art. This enhanced durability, combined with excellent stereoselectivity (ee value > 99.9%), ensures a consistent supply of high-quality chiral intermediates, making it an ideal solution for cost reduction in pharmaceutical intermediates manufacturing.
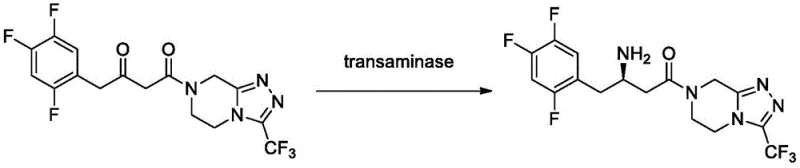
Mechanistically, the success of this approach lies in the precise engineering of the enzyme's active site and its interface with the solid support. The specific mutations, such as M122Q and P223T in the Enz.1 backbone, are strategically located to enhance the enzyme's affinity for bulky ketoamide substrates like 4-oxo-4-[3-(trifluoromethyl)-5,6-dihydro-[1,2,4]triazolo[4,3-a]pyrazin-7(8H)-yl]-1-(2,4,5-trifluorophenyl)butan-2-one. When covalently bonded to epoxy resins like SEPABEADS EC HFA, the enzyme's structural integrity is preserved even in the presence of organic cosolvents. The aqueous isopropanol system provides an optimal microenvironment that balances substrate solubility with enzyme hydration, preventing the denaturation that typically plagues biocatalysis in organic media. This delicate balance allows for high substrate loading concentrations, ranging from 20g/L to 200g/L, facilitating high-throughput processing without compromising the enzyme's catalytic efficiency or stereochemical control.
How to Synthesize Sitagliptin Intermediates Efficiently
The implementation of this biocatalytic route involves a streamlined sequence of steps designed for industrial scalability. It begins with the expression of the specific transaminase mutants in a host organism like E. coli BL21, followed by purification and subsequent immobilization onto the selected epoxy resin. The core of the process is the asymmetric transamination reaction, where the immobilized enzyme catalyzes the conversion of the prochiral ketone to the chiral amine using isopropylamine as the amino donor. The use of the aqueous isopropanol solvent system simplifies the reaction setup, removing the need for specialized solvent conditioning equipment. For a comprehensive understanding of the specific operational parameters, including temperature controls, pH adjustments, and downstream processing, the detailed standardized synthesis steps are provided in the guide below.
- Preparation of the transaminase mutant enzyme solution via gene synthesis and expression in E. coli BL21, followed by cell disruption and purification.
- Immobilization of the purified transaminase mutant onto epoxy resin (e.g., SEPABEADS EC HFA) via covalent bonding to enhance stability in organic solvents.
- Catalytic reaction of the ketoamide substrate with the immobilized enzyme in an aqueous isopropanol system containing isopropylamine as the amino donor.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this immobilized transaminase technology offers compelling strategic advantages that extend beyond mere technical performance. The shift from unstable, single-use enzyme systems to a robust, reusable biocatalyst directly translates into significant cost optimization across the entire production lifecycle. By eliminating the need for complex solvent pre-treatment and reducing the frequency of enzyme replacement, manufacturers can achieve a leaner, more predictable cost structure. This reliability is crucial for maintaining competitive pricing in the global API market, where margin pressures are constant. Furthermore, the simplified process flow reduces the risk of batch failures due to solvent variability, ensuring a more consistent supply of critical intermediates to downstream formulation teams.
- Cost Reduction in Manufacturing: The transition to a reusable immobilized enzyme system drastically reduces the consumption of fresh biocatalyst per kilogram of product. Since the enzyme can be recycled for at least ten batches with minimal loss in activity, the amortized cost of the biocatalyst is significantly lowered compared to free enzyme or less stable immobilized variants. Additionally, the elimination of the water-saturation step for the solvent reduces labor and energy costs associated with solvent preparation and quality control testing. The ability to operate at higher substrate concentrations further enhances volumetric productivity, allowing existing reactor infrastructure to produce more material in less time, thereby driving down the unit cost of production.
- Enhanced Supply Chain Reliability: Stability is the cornerstone of a reliable supply chain. The demonstrated ability of these immobilized mutants to maintain high conversion rates over multiple batches ensures that production schedules can be met without unexpected interruptions caused by enzyme deactivation. This predictability allows for better inventory planning and reduces the need for safety stock buffers. Moreover, the use of commercially available and inexpensive solvents like isopropanol, as opposed to specialized or conditioned solvents, mitigates the risk of supply disruptions for raw materials. This robustness makes the process highly resilient to external market fluctuations, securing the continuity of supply for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this process offers a greener alternative to traditional chemical synthesis which often relies on heavy metal catalysts and harsh conditions. The biocatalytic nature of the reaction ensures high atom economy and reduces the generation of hazardous waste. The simplified solvent system also facilitates easier solvent recovery and recycling, aligning with increasingly stringent environmental regulations. The scalability of the immobilization process on standard epoxy resins means that the technology can be seamlessly transferred from pilot scale to multi-ton commercial production without the need for bespoke equipment, accelerating time-to-market for new generic formulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this transaminase technology. These insights are derived directly from the experimental data and specifications outlined in the patent documentation, providing a clear picture of the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this biocatalytic route into their existing manufacturing portfolios.
Q: What are the key advantages of the new immobilized transaminase mutants over prior art?
A: The novel mutants (SEQ ID NO.3 and SEQ ID NO.7) exhibit significantly higher enzymatic activity and stability when immobilized. Unlike previous methods requiring complex water-saturated IPAc solvents, this invention utilizes a simpler aqueous isopropanol system, improving operational ease and reusability.
Q: Which resin types are compatible with these transaminase mutants?
A: The patent specifies compatibility with various epoxy resins, including SEPABEADS EC HFA, ReliZyme HFA403, ReliZyme EP113, and ReliZyme EP403, with SEPABEADS EC HFA showing particularly favorable results for stability and conversion rates.
Q: How does the solvent system impact the production cost?
A: By switching from water-saturated isopropyl acetate (IPAc) to an aqueous isopropanol solution, the process eliminates the need for precise water saturation control. This simplification reduces solvent preparation time and complexity, leading to substantial cost savings in large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sitagliptin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced biocatalysis in modern pharmaceutical manufacturing. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative technologies like the immobilized transaminase mutants described in CN113061594A can be successfully translated into industrial reality. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of Sitagliptin intermediate meets the highest global standards. We are committed to bridging the gap between laboratory innovation and commercial viability, offering our clients a seamless path from process development to full-scale supply.
We invite you to explore how this cutting-edge technology can optimize your supply chain and reduce your manufacturing costs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production volumes and quality requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in biocatalysis can drive value for your organization.
