Advanced Biocatalytic Production of Chiral Pyrrolidine Intermediates for Global Pharmaceutical Supply Chains
Advanced Biocatalytic Production of Chiral Pyrrolidine Intermediates for Global Pharmaceutical Supply Chains
The pharmaceutical industry is currently witnessing a paradigm shift towards greener, more efficient synthetic methodologies, particularly for complex oncology drug intermediates. A prime example of this innovation is found in patent CN111057729A, which discloses a novel preparation method for (R)-2-(2,5-difluorophenyl)pyrrolidine, a critical chiral building block for the synthesis of Larotretinib. Larotretinib is a potent, oral, selective inhibitor of Tropomyosin Receptor Kinase (TRK), representing a breakthrough 'basket trial' therapy for various advanced tumors. The demand for high-purity intermediates for such targeted therapies is surging, necessitating robust supply chains capable of delivering stereochemically pure materials. This patent outlines a biocatalytic route that leverages imine reductase (IRED) and reduced coenzyme NADPH to achieve asymmetric hydrogenation, offering a compelling alternative to traditional chemical synthesis.
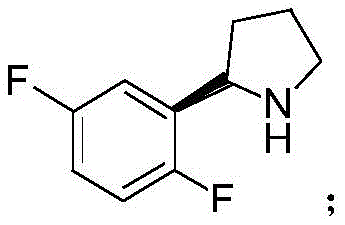
For R&D Directors and Procurement Managers, the implications of this technology are profound. The ability to produce this specific pyrrolidine derivative with an enantiomeric excess (ee) value reaching at least 94% and a conversion rate exceeding 99% addresses two major pain points: purity specifications and yield efficiency. As a reliable pharmaceutical intermediate supplier, understanding the nuances of this biocatalytic pathway allows us to better serve clients seeking cost reduction in API manufacturing while adhering to stringent regulatory standards regarding residual solvents and heavy metals. The transition from chemical to enzymatic catalysis is not merely a technical upgrade but a strategic supply chain enhancement.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral pyrrolidines like the one required for Larotretinib has relied heavily on transition metal-catalyzed asymmetric hydrogenation or non-selective chemical reduction followed by resolution. Patents such as US2016/137654 and US20170281632A1 describe routes utilizing precious metal catalysts to reduce 5-(2,5-difluorophenyl)-3,4-dihydro-2H-pyrrole. While these methods can achieve high conversion rates (95% to 100%), they often suffer from moderate stereoselectivity, with ee values typically ranging between 75% and 85%. More critically, the use of precious metals introduces significant downstream processing challenges, including the need for rigorous heavy metal clearance to meet ICH Q3D guidelines, which drastically increases production costs and waste generation.
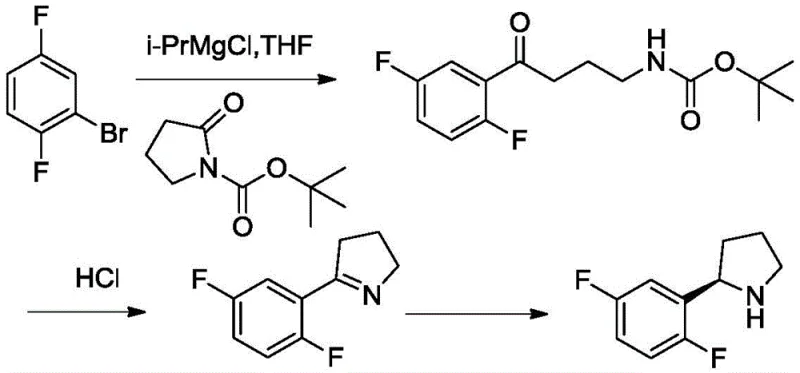
Furthermore, alternative chemical approaches reported in patents like WO2009/140128 utilize sodium borohydride (NaBH4) for reduction. As illustrated in the prior art, this method lacks stereoselectivity entirely, producing a racemic mixture that requires subsequent chiral resolution. This resolution step inherently limits the maximum theoretical yield to 50%, leading to poor atom economy and increased raw material consumption. For a procurement manager, these inefficiencies translate directly into higher unit costs and longer lead times. The reliance on hazardous reductants and the generation of stoichiometric amounts of boron waste also pose environmental compliance risks, making these conventional routes less attractive for sustainable, large-scale manufacturing.
The Novel Approach
In stark contrast, the novel approach detailed in CN111057729A employs a highly specific imine reductase (IRED) to catalyze the asymmetric reduction of the imine bond. This biocatalytic strategy operates under mild physiological conditions, typically in an aqueous buffer system at temperatures around 30°C and a neutral pH of 6.4 to 6.6. The use of engineered enzymes, specifically those with amino acid sequences corresponding to SEQ ID NO.17 or SEQ ID NO.19 (such as IRED72 derived from Paenibacillus elgii), ensures exceptional stereocontrol. The process integrates a cofactor regeneration system using glucose dehydrogenase (GDH) and glucose, which continuously recycles the expensive NADPH cofactor, making the process economically viable. This shift from harsh chemical environments to benign aqueous systems represents a fundamental improvement in process safety and environmental footprint.
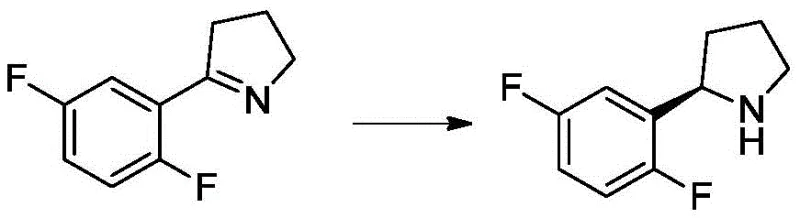
Mechanistic Insights into Imine Reductase-Catalyzed Asymmetric Hydrogenation
The core of this technological advancement lies in the precise mechanistic action of the imine reductase enzyme. Unlike broad-spectrum chemical catalysts, IREDs possess a chiral active site that strictly binds the substrate, 5-(2,5-difluorophenyl)-3,4-dihydro-2H-pyrrole, in a specific orientation. This spatial constraint forces the hydride transfer from the NADPH cofactor to occur exclusively on one face of the imine double bond, resulting in the formation of the (R)-enantiomer with high fidelity. The patent data indicates that specific variants like IRED72 are R-stereoselective, achieving ee values of up to 97.6% in optimized examples. This level of precision eliminates the need for chiral chromatography or crystallization-induced diastereomer transformation, which are often bottlenecks in traditional synthesis.
Furthermore, the integration of the GDH/NADP+ system creates a self-sustaining catalytic cycle. In this coupled reaction, the oxidation of glucose to gluconolactone by GDH provides the reducing equivalents necessary to convert NADP+ back to NADPH. This in situ regeneration means that only a catalytic amount of the expensive cofactor is required (mass ratio of 1:100 relative to substrate), rather than a stoichiometric amount. From an impurity control perspective, this is advantageous because the byproduct, gluconolactone, hydrolyzes to gluconic acid, which is water-soluble and easily removed during the aqueous workup. This contrasts sharply with metal-catalyzed reactions where metal-ligand complexes can persist as difficult-to-remove impurities, complicating the purification of high-purity chiral pyrrolidine.
How to Synthesize (R)-2-(2,5-difluorophenyl)pyrrolidine Efficiently
The implementation of this biocatalytic route requires careful optimization of reaction parameters to maximize both conversion and enantioselectivity. The patent provides a robust framework for scaling this reaction, emphasizing the importance of maintaining specific pH and temperature windows to preserve enzyme activity. The process begins with the preparation of crude enzyme lysates from engineered E. coli strains, followed by the setup of the reaction vessel with the substrate, cofactor, and glucose donor. Detailed standard operating procedures for enzyme expression, reaction monitoring via HPLC, and product isolation are critical for reproducibility. For technical teams looking to adopt this methodology, the following standardized synthesis steps outline the proven protocol derived from the patent examples.
- Preparation of crude enzyme solutions containing imine reductase (IRED72) and glucose dehydrogenase (GDH) from engineered E. coli BL21(DE3) cells via ultrasonic disruption.
- Execution of the asymmetric hydrogenation reaction in an aqueous phosphate buffer (pH 6.4-6.6) at 30°C, utilizing glucose as a hydrogen donor for NADPH cofactor regeneration.
- Downstream processing involving acid-base extraction and solvent removal to isolate the final (R)-configured product with >94% ee and >99% conversion.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and supply continuity, the transition to this enzymatic process offers tangible strategic benefits beyond mere technical elegance. The elimination of precious metal catalysts removes a significant variable cost component and mitigates the supply risk associated with fluctuating prices of metals like palladium or ruthenium. Additionally, the avoidance of hazardous reductants like sodium borohydride simplifies safety protocols and reduces the costs associated with hazardous waste disposal. These factors collectively contribute to a more resilient and cost-effective manufacturing model, positioning suppliers who adopt this technology as preferred partners for long-term API projects.
- Cost Reduction in Manufacturing: The biocatalytic route fundamentally alters the cost structure of producing this intermediate. By replacing expensive chiral metal ligands and stoichiometric chemical reductants with renewable enzymes and inexpensive glucose, the direct material costs are significantly lowered. Furthermore, the high conversion rate (>99%) and high ee value (>94%) minimize the loss of valuable starting materials and eliminate the yield penalties associated with racemic resolution. The simplified downstream processing, which avoids complex metal scavenging steps, further reduces operational expenditures, driving substantial cost savings in API manufacturing without compromising quality.
- Enhanced Supply Chain Reliability: Dependence on specialized chemical reagents often introduces fragility into the supply chain. In contrast, the reagents for this biocatalytic process—glucose, phosphate buffers, and recombinant enzymes—are widely available and can be produced at scale with high consistency. The use of robust bacterial strains (like E. coli BL21) for enzyme production ensures a stable supply of the biocatalyst. This reliability is crucial for meeting the rigorous delivery schedules of pharmaceutical clients, reducing the risk of production delays caused by raw material shortages or quality failures in complex chemical synthesis steps.
- Scalability and Environmental Compliance: Scaling chemical hydrogenation often requires high-pressure equipment and strict safety measures, which can be capital intensive. The enzymatic process described operates at atmospheric pressure and mild temperatures (30°C), allowing for easier scale-up in standard stainless steel reactors. Moreover, the use of water as the primary solvent aligns with Green Chemistry principles, significantly reducing the volume of organic solvent waste. This environmental advantage facilitates smoother regulatory approvals and supports corporate sustainability goals, making the supply chain more compliant with increasingly strict global environmental regulations.
Frequently Asked Questions (FAQ)
Understanding the technical specifics of this new manufacturing route is essential for making informed sourcing decisions. The following questions address common inquiries regarding the feasibility, quality, and operational parameters of the biocatalytic synthesis of (R)-2-(2,5-difluorophenyl)pyrrolidine. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a transparent view of the technology's capabilities.
Q: What are the advantages of using imine reductase over traditional metal catalysts for this synthesis?
A: The biocatalytic method described in patent CN111057729A eliminates the need for expensive precious metal catalysts (like Pd or Ru) and avoids heavy metal contamination issues common in chemical hydrogenation. It achieves superior stereoselectivity (>94% ee) under mild aqueous conditions, significantly simplifying downstream purification and reducing environmental impact.
Q: What represents the optimal reaction conditions for maximum conversion?
A: According to the patent data, optimal conversion rates exceeding 99% are achieved at a substrate concentration of 0.05 g/mL, a reaction temperature of 30°C, and a controlled pH range of 6.4 to 6.6. The system utilizes a coupled enzyme approach with glucose dehydrogenase to regenerate the NADPH cofactor efficiently.
Q: Is this process suitable for large-scale industrial production of Larotretinib intermediates?
A: Yes, the process is specifically designed for industrial scalability. It uses water as the primary solvent, operates at ambient temperatures (30°C), and utilizes inexpensive glucose as a reductant. These factors lower operational costs and safety risks associated with high-pressure hydrogenation or hazardous chemical reductants like sodium borohydride.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (R)-2-(2,5-difluorophenyl)pyrrolidine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of next-generation oncology drugs like Larotretinib depends on the availability of high-quality, cost-effective intermediates. Our technical team has thoroughly analyzed the biocatalytic pathways described in recent patents, including CN111057729A, and we possess the expertise to implement these green chemistry solutions at an industrial scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of materials that meet stringent purity specifications. Our rigorous QC labs are equipped to verify enantiomeric purity and residual solvent levels, guaranteeing that every batch aligns with global regulatory standards.
We invite pharmaceutical companies and contract research organizations to collaborate with us on optimizing the supply chain for chiral pyrrolidine derivatives. By leveraging our advanced biocatalytic capabilities, we can offer a Customized Cost-Saving Analysis tailored to your specific project needs. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Let us partner with you to accelerate your drug development timeline while achieving significant efficiencies in your manufacturing operations.
