Revolutionizing Fragrance Manufacturing: High-Efficiency Enzymatic Synthesis of Optically Pure Lactones
The landscape of fine chemical manufacturing, particularly within the high-value fragrance sector, is undergoing a paradigm shift driven by the urgent demand for sustainable, high-purity intermediates. Patent CN113201511A introduces a groundbreaking advancement in this domain: a novel (R)-5-carbonyl decanoate (ester) reductase mutant, designated as SmCR, which offers unprecedented efficiency in the preparation of optically pure (R)-gamma/delta-lactones. These lactone compounds, renowned for their creamy, fruity, and coconut-like aromas, are critical building blocks for premium perfumery and food flavorings. Traditionally, obtaining these molecules with high optical purity has been a bottleneck, often relying on inefficient extraction or chemically intensive processes. This new biocatalytic technology addresses these historical challenges by leveraging protein engineering and directed evolution to create an enzyme variant with superior catalytic performance, stability, and enantioselectivity. For industry leaders seeking a reliable flavor intermediate supplier, this technology represents a significant leap forward in process capability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of lactone fragrance compounds has been fraught with significant technical and economic hurdles that limit scalability and product quality. Early methods relied heavily on the extraction of these compounds from natural plant sources, a process inherently limited by low yields and seasonal variability, resulting in exorbitant costs that restrict their application to niche luxury markets. While fermentation technologies using Saccharomyces cerevisiae offered a biological alternative, they suffer from severe metabolic bottlenecks; specifically, the accumulation of lactone products and their hydroxy-acid precursors often exhibits cytotoxicity towards the yeast strains themselves, capping the achievable titers and necessitating complex in-situ product removal strategies. Furthermore, chemical synthesis routes, while capable of higher volumes, frequently employ harsh conditions and stoichiometric amounts of chiral auxiliaries or transition metal catalysts. These traditional chemical pathways often generate unwanted color impurities and racemic by-products that are difficult to separate, posing serious risks for food and beverage applications where regulatory compliance and sensory purity are paramount. The reliance on heavy metal catalysts also introduces substantial environmental liabilities and downstream purification costs associated with removing trace metal residues to meet safety standards.
The Novel Approach
In stark contrast to these legacy methods, the novel approach disclosed in the patent utilizes a highly engineered SmCR mutant to catalyze the asymmetric reduction of prochiral 5-carbonyl compounds with exceptional precision. This biocatalytic route bypasses the toxicity issues of whole-cell fermentation by employing isolated enzyme systems or resting cells that can tolerate significantly higher substrate concentrations, reaching up to 200 mM without loss of activity. The process operates under mild physiological conditions, typically around 30°C and neutral pH, which preserves the integrity of sensitive functional groups and prevents the formation of thermal degradation by-products. By coupling the SmCR mutant with a glucose dehydrogenase (GDH) system, the process achieves efficient in-situ regeneration of the expensive NADPH cofactor, drastically reducing the overall cost of goods. This enzymatic cascade ensures that the reduction step proceeds with >98% conversion and >99% enantiomeric excess (ee), delivering a hydroxy-acid intermediate that is primed for the subsequent cyclization step. This level of control allows manufacturers to produce high-purity API intermediates and fragrance components with a consistency that chemical synthesis simply cannot match.
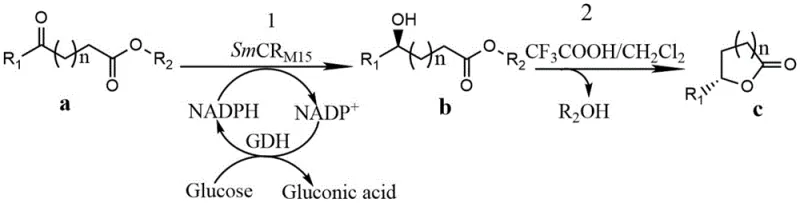
Mechanistic Insights into SmCR-Mediated Asymmetric Reduction
The core of this technological breakthrough lies in the rational design and directed evolution of the SmCR enzyme, specifically targeting the active site to accommodate long-chain aliphatic substrates. The wild-type enzyme, derived from Serratia marcescens, originally showed limited activity towards 5-carbonyl substrates. Through a combination of error-prone PCR and iterative saturation mutagenesis, specific amino acid residues surrounding the substrate binding pocket—such as positions 23, 61, 127, and 145—were modified to optimize steric fit and electronic interactions. For instance, the M15 mutant variant incorporates multiple substitutions (E23K, K61V, E104K, L107I, K127E, N145R, A149T, R172S) that collectively reshape the hydrophobic channel, allowing the long alkyl chain of the 5-carbonyl decanoate to bind more deeply and securely. This precise positioning ensures that the hydride transfer from the NADPH cofactor occurs exclusively to the Re-face of the carbonyl group, resulting in the exclusive formation of the (R)-configured hydroxy acid. The mechanistic fidelity of this enzyme is further enhanced by its robust thermal stability, with a half-life significantly extended compared to the parent enzyme, allowing for prolonged batch operations without frequent enzyme replenishment.
Beyond the primary reduction mechanism, the process demonstrates remarkable versatility in handling a broad spectrum of substrates, as defined by the general formula where n varies to produce either gamma or delta lactones. The enzyme's ability to accept various R1 alkyl chains (from butyl to octyl and even aromatic groups) indicates a flexible active site that does not strictly discriminate against chain length variations. This broad substrate tolerance is crucial for industrial applications where a single biocatalyst platform can be deployed to manufacture a portfolio of different fragrance molecules, such as delta-nonalactone, delta-decalactone, and delta-undecalactone, simply by changing the feedstock. Furthermore, the impurity profile of the enzymatic reaction is exceptionally clean; unlike chemical hydrogenation which might reduce other unsaturated bonds or cause racemization, the SmCR mutant is highly chemoselective for the ketone functionality. This specificity minimizes the formation of side products like hemiacetals or over-reduced alcohols, simplifying the downstream workup to a straightforward acidification and extraction, thereby enhancing the overall space-time yield of the manufacturing process.
How to Synthesize Delta-Lactone Efficiently
The implementation of this biocatalytic route involves a streamlined workflow that integrates recombinant protein expression with a robust reaction protocol. The process begins with the cultivation of E. coli hosts expressing the optimized SmCR mutant, followed by cell disruption to release the intracellular enzyme or the use of permeabilized resting cells. The reduction reaction is conducted in an aqueous buffer system supplemented with glucose as a sacrificial electron donor, ensuring continuous cofactor recycling. Once the asymmetric reduction is complete, the reaction mixture is acidified to induce spontaneous or catalyzed lactonization of the resulting chiral hydroxy acid. Detailed standardized synthesis steps for this high-efficiency pathway are provided below.
- Express the recombinant SmCR mutant (e.g., M15) in E. coli BL21(DE3) and prepare crude enzyme powder or resting cells.
- Perform asymmetric reduction of the 5-carbonyl substrate (20-200 mM) in phosphate buffer with glucose dehydrogenase for NADPH regeneration.
- Quench the reaction with acid, extract the hydroxy-acid intermediate, and perform acid-catalyzed cyclization to form the final lactone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this SmCR mutant technology translates into tangible strategic advantages that extend far beyond simple yield improvements. The shift from metal-catalyzed chemistry or low-titer fermentation to high-efficiency biocatalysis fundamentally alters the cost structure and risk profile of fragrance manufacturing. By eliminating the need for precious metal catalysts and the associated rigorous purification steps required to meet ppm-level metal specifications, the process significantly reduces raw material costs and waste disposal expenses. Moreover, the high substrate tolerance of the mutant enzyme means that reactors can be charged with much higher concentrations of starting material, effectively increasing the volumetric productivity of existing infrastructure without the need for capital-intensive expansion. This intensification of the process leads to a drastic simplification of the manufacturing workflow, reducing the number of unit operations and the overall processing time required to bring a batch to completion.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the elimination of expensive chiral resolving agents and the reduction of solvent usage due to higher reaction concentrations. Because the enzyme operates with high turnover numbers and the cofactor is recycled efficiently via the glucose dehydrogenase couple, the consumption of auxiliary reagents is minimized. This leaner reagent profile, combined with the avoidance of cryogenic conditions or high-pressure hydrogenation equipment, results in substantially lower utility and operational expenditures. The simplified downstream processing, requiring only basic extraction and distillation rather than complex chromatography, further drives down the cost per kilogram of the final high-purity lactone product.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the robustness of the recombinant enzyme system, which can be produced at scale using standard fermentation techniques for E. coli, ensuring a consistent and reliable source of biocatalyst. Unlike natural extraction methods which are vulnerable to agricultural fluctuations, or chemical routes dependent on volatile petrochemical feedstocks, this biocatalytic route relies on stable, commercially available keto-acid precursors. The high stability of the SmCR mutant also allows for the potential formulation of the enzyme into stable liquid or solid preparations that can be stored and transported with ease, reducing the risk of production delays caused by catalyst degradation or supply shortages.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process offers a greener alternative that aligns with increasingly stringent global regulations. The aqueous nature of the reaction medium and the absence of toxic heavy metals eliminate the generation of hazardous waste streams, facilitating easier compliance with environmental discharge permits. The scalability of the process is proven by its ability to handle high substrate loads (up to 200 mM) in standard stirred-tank reactors, demonstrating that the technology is ready for commercial scale-up of complex fragrance intermediates. This environmental compatibility not only reduces regulatory risk but also enhances the brand value of the final consumer products by enabling 'natural' or 'bio-based' labeling claims in certain jurisdictions.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented biocatalytic technology. These insights are derived directly from the experimental data and embodiments disclosed in the patent documentation, providing a clear understanding of the process capabilities for potential licensees or manufacturing partners.
Q: What are the advantages of the SmCR mutant over wild-type enzymes for lactone synthesis?
A: The SmCR mutant exhibits significantly improved catalytic activity, thermal stability, and enantioselectivity towards 5-carbonyl compounds compared to the parent enzyme, allowing for higher substrate loading (up to 200 mM) and faster reaction times.
Q: How is the cofactor NADPH regenerated in this process?
A: The process utilizes a coupled enzyme system where glucose dehydrogenase (GDH) oxidizes glucose to gluconic acid, simultaneously regenerating NADPH from NADP+, ensuring a sustainable and cost-effective catalytic cycle.
Q: Can this method produce both gamma and delta lactones?
A: Yes, the engineered SmCR mutant has an expanded substrate spectrum capable of reducing various long-chain aliphatic keto acids with carbonyls at gamma and delta positions, yielding corresponding optically pure lactones after cyclization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Delta-Lactone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced biocatalysis in the production of high-value fragrance intermediates like delta-lactones. As a leading CDMO partner, we possess the technical expertise to adapt and optimize such enzymatic routes for your specific production needs, ensuring seamless technology transfer from lab bench to commercial plant. Our facilities are equipped with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, guaranteeing that the high purity and stereochemical integrity demonstrated in the patent are maintained at an industrial scale. We operate stringent purity specifications and maintain rigorous QC labs to verify that every batch meets the exacting standards required by the global flavor and fragrance industry.
We invite you to explore how this innovative SmCR mutant technology can optimize your supply chain and reduce your manufacturing footprint. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your current production volumes. We are prepared to provide specific COA data for similar lactone intermediates and conduct comprehensive route feasibility assessments to demonstrate how we can become your trusted partner in delivering high-quality, cost-effective fragrance solutions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
