Scalable Synthesis of Chiral Delta-Lactones for High-Purity Orlistat Production
Introduction to Advanced Delta-Lactone Manufacturing
The pharmaceutical industry continuously demands more efficient and sustainable pathways for producing complex active pharmaceutical ingredients (APIs) and their precursors. Patent CN100355740C introduces a groundbreaking methodology for the synthesis of 3,6-dialkyl-5,6-dihydro-4-hydroxy-pyran-2-one, a critical delta-lactone intermediate widely recognized for its role in the production of Orlistat (tetrahydrolipstatin). This novel process addresses long-standing challenges in stereochemical control and reaction safety, offering a robust alternative to traditional zinc-mediated cyclizations. By leveraging magnesium-based intramolecular Reformatsky reactions and advanced ruthenium-catalyzed asymmetric hydrogenation, this technology ensures high optical purity while mitigating environmental hazards. For global procurement teams, understanding this shift is vital for securing a reliable pharmaceutical intermediate supplier capable of meeting stringent regulatory and volume requirements.
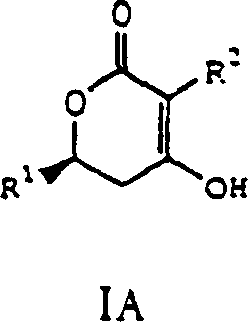
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of delta-lactones such as 3-hexyl-4-hydroxy-6-undecyl-5,6-dihydro-pyran-2-one has relied heavily on intramolecular Reformatsky reactions utilizing low-valent metals like zinc. While effective, these traditional protocols present significant drawbacks for industrial scale-up. The use of zinc generates heavy metal waste that is difficult and costly to treat, posing environmental compliance challenges for modern manufacturing facilities. Furthermore, conventional methods often necessitate the use of low-boiling ether solvents, which create high concentrations of flammable vapors within production equipment, increasing the risk of hazardous incidents. Additionally, achieving the necessary stereochemical purity often requires resolution of racemic mixtures, leading to substantial material loss and increased production costs due to the discarding of unwanted isomers.
The Novel Approach
The innovative process described in the patent data fundamentally shifts the paradigm by employing magnesium or Grignard reagents as the active species generator for the cyclization step. This substitution eliminates the environmental burden associated with zinc waste disposal and allows for the use of safer, higher-boiling solvents like tetrahydrofuran or toluene, thereby enhancing operational safety. Moreover, the integration of enantioselective hydrogenation using chiral ruthenium catalysts at the precursor stage ensures that the beta-hydroxy intermediates are produced with high optical purity from the outset. This eliminates the need for downstream resolution steps, drastically improving overall atom economy and yield. This approach represents a significant leap forward in cost reduction in pharmaceutical intermediate manufacturing by streamlining the synthetic route and minimizing waste generation.
Mechanistic Insights into Magnesium-Mediated Cyclization and Asymmetric Hydrogenation
The core of this technological advancement lies in the dual optimization of the cyclization mechanism and the stereoselective reduction step. In the cyclization phase, the reaction of an alpha-haloester with magnesium or a Grignard reagent leads to the formation of an active alpha-magnesium halide ester species. Contrary to earlier beliefs that Grignard species could not be formed from alpha-haloesters, this process demonstrates that metal-halide exchange occurs efficiently, facilitating an intramolecular nucleophilic attack on the ester carbonyl to close the lactone ring. The addition of excess Grignard reagent serves a dual purpose: generating the active species and quenching acidic protons in the initial cyclization product, thereby preventing decomposition and significantly boosting yield.
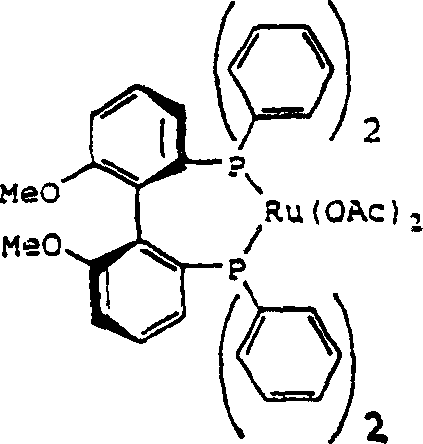
Complementing this is the highly sophisticated asymmetric hydrogenation of beta-keto esters. The process utilizes chiral ruthenium diphosphine complexes, specifically derivatives of Ru(OAc)2((R)-MeOBIPHEP). The mechanism involves the coordination of the beta-keto ester to the ruthenium center, followed by hydride transfer that is strictly controlled by the chiral environment of the MeOBIPHEP ligand. This ensures the preferential formation of the (R)-beta-hydroxy configuration. Remarkably, the catalyst system is robust enough to function effectively with technical grade hydrogen and does not require the ultra-high purity conditions typical of other asymmetric hydrogenations. This mechanistic resilience translates directly to process reliability and reduced operational complexity.
How to Synthesize 3,6-dialkyl-5,6-dihydro-4-hydroxy-pyran-2-one Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize the benefits of the novel catalytic systems. The process generally begins with the enantioselective reduction of a beta-keto ester, followed by esterification with an alpha-halo activated carbonyl compound, and concludes with the magnesium-mediated cyclization. Each step is optimized to maintain stereochemical integrity and minimize byproduct formation. The detailed standardized synthesis steps see the guide below.
- Enantioselective reduction of beta-keto esters using Ru-MeOBIPHEP catalysts to achieve high optical purity.
- Esterification of the resulting chiral beta-hydroxy compound with alpha-halo activated carbonyl compounds.
- Intramolecular cyclization via magnesium or Grignard reagents to form the target delta-lactone ring structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers tangible strategic advantages beyond mere technical superiority. The shift from zinc to magnesium chemistry fundamentally alters the cost structure of production by removing the need for expensive heavy metal waste treatment protocols. This simplification of the waste stream not only reduces direct disposal costs but also minimizes the regulatory burden associated with environmental compliance, ensuring smoother operations and fewer interruptions. Furthermore, the ability to use technical grade hydrogen and lower pressures reduces the capital expenditure required for specialized high-pressure reactor infrastructure, making the technology accessible for broader commercial scale-up.
- Cost Reduction in Manufacturing: The elimination of zinc waste treatment and the removal of racemic resolution steps lead to substantial cost savings. By achieving high enantiomeric excess directly through catalysis, the process avoids the 50% theoretical yield loss inherent in resolution methods. Additionally, the use of cheaper technical grade hydrogen and the avoidance of ultra-pure gas requirements further decrease raw material expenses, resulting in a more economically viable production model for high-volume API intermediates.
- Enhanced Supply Chain Reliability: The robustness of the magnesium-based cyclization and the ruthenium catalyst system ensures consistent batch-to-batch quality. The tolerance for technical grade reagents means that supply chain disruptions related to the availability of ultra-high purity gases are mitigated. This reliability is crucial for maintaining continuous production schedules and meeting the strict delivery timelines demanded by downstream pharmaceutical manufacturers, thereby strengthening the overall resilience of the supply network.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing solvents and reagents that are manageable in large-scale reactors without the safety risks associated with low-boiling ethers. The reduced environmental footprint, characterized by easier waste disposal and lower energy consumption for gas purification, aligns with modern green chemistry principles. This facilitates faster regulatory approvals and supports long-term sustainability goals, making it an ideal choice for partners committed to responsible manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis pathway. These insights are derived directly from the experimental data and embodiments provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing production lines.
Q: Why is Magnesium preferred over Zinc for this Reformatsky cyclization?
A: Magnesium waste is significantly easier to dispose of and less environmentally harmful than zinc waste. Additionally, magnesium allows for reactions in higher boiling solvents, reducing safety risks associated with volatile ether vapors in large-scale production.
Q: What level of enantiomeric excess (ee) can be achieved with this process?
A: The process utilizes chiral ruthenium catalysts, specifically Ru(OAc)2((R)-MeOBIPHEP), which can achieve enantiomeric excess values of at least 90% ee, often exceeding 99% ee after a single recrystallization step.
Q: Does this method require ultra-high purity hydrogen gas?
A: No, unlike conventional methods requiring 99.99% purity, this catalytic system operates effectively with technical grade hydrogen (approx. 99.5% purity), significantly lowering operational costs and infrastructure requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Orlistat Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the pharmaceutical value chain. Our expertise extends to mastering complex synthetic routes like the magnesium-mediated Reformatsky cyclization and asymmetric hydrogenation described in CN100355740C. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent, high-purity materials regardless of volume. Our rigorous QC labs and stringent purity specifications guarantee that every batch meets the exacting standards required for API synthesis, providing peace of mind for your regulatory filings.
We invite you to collaborate with us to optimize your supply chain for Orlistat and related pharmaceutical intermediates. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can drive efficiency and reliability in your operations.
