Advanced Biocatalytic Synthesis of Sitagliptin Intermediates Using Engineered Aminotransferases
The pharmaceutical industry is currently witnessing a paradigm shift towards sustainable and highly selective manufacturing processes, particularly for complex chiral molecules like Sitagliptin, a leading DPP-IV inhibitor for type II diabetes. Patent CN107384887B introduces a groundbreaking biocatalytic approach utilizing a novel ω-aminotransferase derived from Burkholderia gladioli ZJB-12126. This technology addresses critical bottlenecks in traditional synthesis, offering a robust alternative to metal-catalyzed hydrogenation. By leveraging directed evolution, the inventors have developed specific enzyme mutants capable of operating efficiently under industrially relevant conditions, including high substrate loading and the presence of organic cosolvents. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, this enzymatic route represents a significant advancement in process chemistry, promising enhanced purity profiles and streamlined regulatory compliance through the elimination of toxic reagents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Sitagliptin and its key chiral intermediates has relied heavily on transition metal-catalyzed asymmetric hydrogenation or complex chiral pool strategies. Patents such as US6699871 and WO2004087650 describe routes utilizing expensive Ruthenium or Germanium catalysts, often requiring high-pressure hydrogen atmospheres and rigorous safety protocols. These chemical methods frequently suffer from moderate stereoselectivity, with ee values often hovering around 94-96%, necessitating additional recrystallization steps that erode overall yield. Furthermore, alternative chemical routes involving cyanide substitution, as seen in CN102838511, introduce severe environmental and safety hazards due to the toxicity of the reagents. The cumulative effect of these limitations is a manufacturing process with high operational expenditure, significant waste generation, and a complex impurity profile that complicates downstream purification and quality control.
The Novel Approach
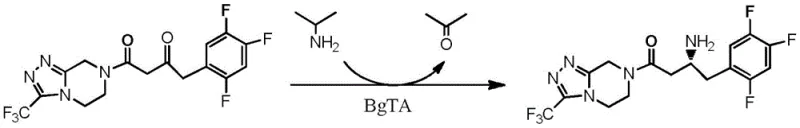
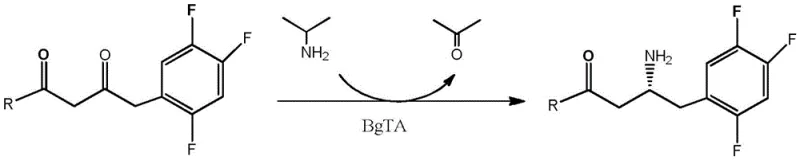
In stark contrast, the novel approach detailed in CN107384887B employs a biocatalytic transamination strategy that operates under mild, ambient conditions. The core innovation lies in the use of engineered aminotransferase mutants that exhibit exceptional activity towards the bulky Sitagliptin precursor ketone. Unlike the wild-type enzyme, which shows negligible conversion at high substrate concentrations, the evolved mutants maintain high catalytic efficiency even at loadings of 50g/L. This method utilizes isopropylamine as a cheap and readily available amino donor, driving the equilibrium towards product formation without the need for high-pressure equipment. The result is a process that achieves a total yield of 76% with an optical purity exceeding 99% ee, effectively bypassing the need for chiral resolution and significantly simplifying the production workflow for cost reduction in API manufacturing.
Mechanistic Insights into BgTA-Catalyzed Asymmetric Transamination
The catalytic mechanism relies on the pyridoxal-5'-phosphate (PLP) dependent transfer of an amino group from isopropylamine to the prochiral ketone substrate. The enzyme forms a Schiff base intermediate with the cofactor, facilitating the stereospecific abstraction of a proton and subsequent amination to generate the (R)-configured amine. The patent highlights specific amino acid substitutions in Mutant 1 (SEQ ID NO: 4) and Mutant 2 (SEQ ID NO: 6), such as H53T, Y113M, and W115T, which are critical for expanding the active site volume. These mutations alleviate steric hindrance caused by the trifluorophenyl group of the substrate, allowing for tighter binding and more efficient turnover. Understanding these structural modifications is vital for R&D teams aiming to further optimize reaction kinetics or adapt the enzyme for analogous substrates within the same chemical class.
Furthermore, the process demonstrates remarkable tolerance to dimethyl sulfoxide (DMSO), used as a cosolvent to enhance substrate solubility. The ability of the mutant enzymes to retain structural integrity and catalytic function in up to 40% (v/v) DMSO is a significant technical achievement, as organic solvents typically denature proteins. This solvent tolerance ensures that the reaction can proceed at high substrate concentrations without precipitation, maintaining a homogeneous system that is easier to monitor and control. The high enantioselectivity (>99% ee) is intrinsic to the enzyme's active site geometry, which strictly discriminates against the formation of the (S)-enantiomer, thereby ensuring that the final product meets the stringent purity specifications required for global pharmaceutical markets without extensive chromatographic separation.
How to Synthesize Sitagliptin Efficiently
The implementation of this biocatalytic route involves a straightforward fermentation and conversion protocol that is amenable to scale-up. The process begins with the cultivation of recombinant E. coli expressing the mutant aminotransferase, followed by the preparation of wet cell biomass which serves as the whole-cell biocatalyst. The reaction is conducted in a triethanolamine buffer system at pH 8.5, providing a stable environment for enzyme activity. Detailed standard operating procedures regarding induction times, cell lysis options, and specific work-up protocols involving acid-base extraction are essential for maximizing recovery. The following guide outlines the critical operational parameters derived from the patent data to ensure successful replication of these high-efficiency results.
- Prepare the biocatalyst by fermenting recombinant E. coli containing the BgTA mutant gene, harvesting wet cells, and resuspending in triethanolamine buffer.
- Establish the reaction system with sitagliptin precursor ketone (up to 50g/L), isopropylamine as amino donor, PLP coenzyme, and DMSO cosolvent at pH 8.5.
- Incubate at 35°C with stirring for 36 hours, then proceed to acid-base extraction and purification to isolate high-purity sitagliptin.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this enzymatic technology offers tangible strategic benefits beyond mere technical performance. The primary advantage lies in the drastic simplification of the supply chain for raw materials. By replacing scarce and price-volatile precious metal catalysts with fermentable biological systems, manufacturers can insulate themselves from geopolitical fluctuations in metal markets. Additionally, the elimination of high-pressure hydrogenation steps reduces the capital expenditure required for specialized reactor infrastructure, allowing for production in standard stainless steel vessels. This flexibility enhances supply chain reliability and reduces lead time for high-purity pharmaceutical intermediates by enabling faster batch turnover and simplified maintenance schedules.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the removal of expensive chiral ligands and transition metals like Ruthenium and Germanium from the bill of materials. Since the biocatalyst is produced via fermentation, the marginal cost of the enzyme decreases significantly with scale, unlike stoichiometric chemical reagents. Furthermore, the high conversion rates and excellent stereoselectivity minimize the loss of valuable starting materials and reduce the consumption of solvents and energy associated with recycling and purification steps. These factors collectively contribute to substantial cost savings in the overall manufacturing budget without compromising on product quality.
- Enhanced Supply Chain Reliability: Relying on biological catalysts diversifies the sourcing strategy away from single-source chemical suppliers who may face production disruptions. The genetic information for the enzyme can be stored indefinitely and expressed on-demand, ensuring a continuous and secure supply of the critical processing agent. Moreover, the mild reaction conditions (30-45°C, atmospheric pressure) lower the risk of safety incidents that could halt production lines. This operational stability is crucial for maintaining consistent delivery schedules to downstream API manufacturers and mitigating the risk of inventory shortages in the global market.
- Scalability and Environmental Compliance: From an environmental perspective, this aqueous-based biocatalytic process generates significantly less hazardous waste compared to traditional organic synthesis. The absence of heavy metals simplifies wastewater treatment and reduces the burden of regulatory compliance regarding effluent discharge. The process is inherently scalable, as demonstrated by the successful conversion at substrate concentrations of 50g/L, which translates well to commercial scale-up of complex pharmaceutical intermediates. This alignment with green chemistry principles not only improves the corporate sustainability profile but also future-proofs the manufacturing process against increasingly stringent environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this aminotransferase technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on performance metrics and operational feasibility. Understanding these details is essential for stakeholders evaluating the integration of this biocatalytic route into their existing production portfolios.
Q: What are the advantages of using BgTA mutants over wild-type enzymes for sitagliptin synthesis?
A: The engineered mutants (specifically Mutant 2, SEQ ID NO: 6) demonstrate significantly improved tolerance to high substrate concentrations (up to 50g/L) and organic cosolvents like DMSO, achieving conversion rates of 91% compared to less than 1% for the wild-type under identical high-load conditions.
Q: Does this biocatalytic process require expensive metal catalysts?
A: No, this method completely eliminates the need for precious metal catalysts such as Ruthenium, Platinum, or Germanium, which are traditionally used in asymmetric hydrogenation routes, thereby reducing raw material costs and heavy metal contamination risks.
Q: What is the optical purity of the sitagliptin produced via this method?
A: The process consistently yields sitagliptin with an enantiomeric excess (ee) value greater than 99%, meeting the stringent stereochemical requirements for pharmaceutical-grade active ingredients without the need for complex chiral resolution steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sitagliptin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the biocatalytic synthesis routes described in CN107384887B for the production of Sitagliptin intermediates. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory successes are seamlessly translated into industrial reality. Our facilities are equipped with state-of-the-art fermentation and downstream processing capabilities, supported by rigorous QC labs that guarantee stringent purity specifications for every batch. We are committed to helping our partners navigate the complexities of enzyme engineering and process optimization to achieve maximum efficiency.
We invite you to collaborate with us to leverage this advanced technology for your supply chain needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this enzymatic route can improve your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a sustainable, cost-effective, and high-quality supply of Sitagliptin intermediates that meets the demands of the global pharmaceutical market.
