Advanced Manufacturing of Sitagliptin Intermediates via Asymmetric Hydrogenation
Advanced Manufacturing of Sitagliptin Intermediates via Asymmetric Hydrogenation
The global pharmaceutical landscape continues to demand more efficient and sustainable pathways for producing high-volume antidiabetic agents, particularly DPP-IV inhibitors like Sitagliptin. Patent CN106146340B introduces a transformative methodology for synthesizing the critical chiral intermediate required for Sitagliptin production, addressing long-standing inefficiencies in traditional manufacturing. This technical insight report analyzes the novel synthetic route disclosed in the patent, which leverages a highly selective asymmetric hydrogenation strategy to achieve exceptional stereochemical purity. By bypassing traditional resolution steps and utilizing a direct Heck coupling on unprotected acids, this technology offers a compelling value proposition for generic drug manufacturers and contract development organizations seeking to optimize their supply chains. The following analysis details the mechanistic advantages, operational parameters, and commercial implications of adopting this advanced synthetic protocol.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Sitagliptin intermediates has been plagued by significant economic and technical hurdles that hinder cost-effective mass production. Traditional routes often rely on the use of expensive chiral pool starting materials or require cumbersome resolution processes to separate enantiomers, which theoretically caps the maximum yield at 50% for the desired isomer. Furthermore, many legacy protocols necessitate the protection and deprotection of functional groups, such as carboxylic acids, adding multiple synthetic steps that increase solvent consumption, waste generation, and overall processing time. These multi-step sequences not only inflate the cost of goods sold (COGS) but also introduce additional points of failure where yield losses can accumulate. The reliance on harsh reaction conditions or difficult-to-remove heavy metal catalysts in older methods further complicates purification and regulatory compliance, creating bottlenecks for reliable pharmaceutical intermediates supplier networks trying to maintain consistent quality.
The Novel Approach
The methodology outlined in CN106146340B represents a paradigm shift by integrating a direct Heck reaction with a high-performance asymmetric hydrogenation step. Unlike conventional strategies, this approach allows the carboxylic acid moiety of the itaconic acid derivative to participate directly in the palladium-catalyzed coupling without prior protection, significantly streamlining the early stages of synthesis. The core innovation lies in the subsequent stereoselective reduction, which utilizes a specialized Iridium catalyst system to install the chiral center with precision. This eliminates the need for diastereomeric salt formation and crystallization, thereby doubling the theoretical yield compared to resolution-based methods. By condensing the synthetic sequence and improving atom economy, this novel approach facilitates cost reduction in pharmaceutical intermediates manufacturing while simultaneously enhancing the environmental profile of the production process through reduced solvent usage and waste generation.
Mechanistic Insights into Iridium-Catalyzed Asymmetric Hydrogenation
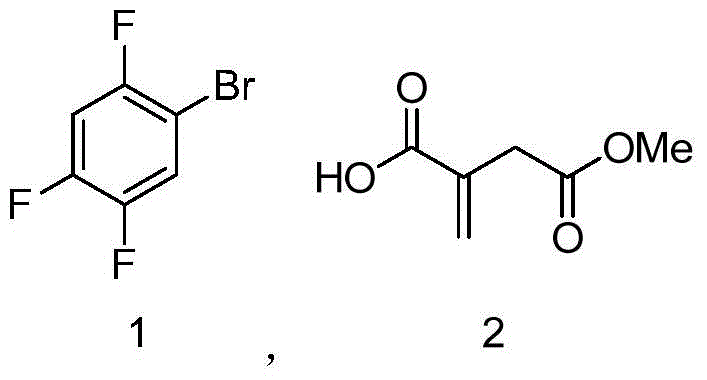
The cornerstone of this synthetic advancement is the enantioselective hydrogenation of the olefinic intermediate using the chiral catalyst [Ir-((R)-DTB-SIPHOX)(COD)]BArF. This specific Iridium complex features a SIPHOX ligand backbone modified with bulky 3,5-di-tert-butylphenyl (DTB) groups, which create a highly defined chiral pocket around the metal center. During the catalytic cycle, the substrate coordinates to the Iridium atom, and the steric bulk of the ligand directs the approach of molecular hydrogen to one specific face of the double bond. This precise spatial control ensures that the hydrogen atoms are added exclusively to generate the (R)-configuration required for biological activity. The use of the weakly coordinating BArF anion further enhances catalyst activity by preventing the formation of inactive ion pairs, allowing the reaction to proceed efficiently even at relatively low catalyst loadings of 0.001 to 1 mol%. This level of catalytic efficiency is critical for maintaining economic viability when using precious metal catalysts in large-scale operations.
From an impurity control perspective, this mechanism offers superior selectivity compared to non-catalytic or less selective reduction methods. The high enantiomeric excess (ee > 99%) achieved directly from the hydrogenation step means that downstream purification requirements are drastically reduced. In traditional synthesis, trace amounts of the wrong enantiomer can act as difficult-to-remove impurities that persist through multiple steps, potentially compromising the safety profile of the final API. By establishing the correct stereochemistry early in the sequence with such high fidelity, the risk of carrying over chiral impurities is virtually eliminated. Additionally, the mild reaction conditions (60-80°C) prevent thermal degradation or racemization of the sensitive chiral center, ensuring that the optical purity established during hydrogenation is maintained throughout the subsequent conversion to the acid chloride and Boc-protected amine derivatives.
How to Synthesize Sitagliptin Intermediate Efficiently
The implementation of this synthesis route involves a logical progression of well-defined chemical transformations that are amenable to standard reactor setups. The process begins with the formation of the carbon-carbon bond via Heck coupling, followed by the critical stereochemistry-setting hydrogenation, and concludes with functional group manipulations to install the amine protection. Each step has been optimized to balance reaction rate with product quality, utilizing common organic solvents like acetonitrile, methanol, and dichloromethane. The detailed standardized synthesis steps see the guide below, which outlines the specific stoichiometry, temperature profiles, and workup procedures necessary to replicate the high yields and purity reported in the patent data. Adhering to these parameters is essential for achieving the reported 99% yield in the hydrogenation step and the overall high purity of the final intermediate.
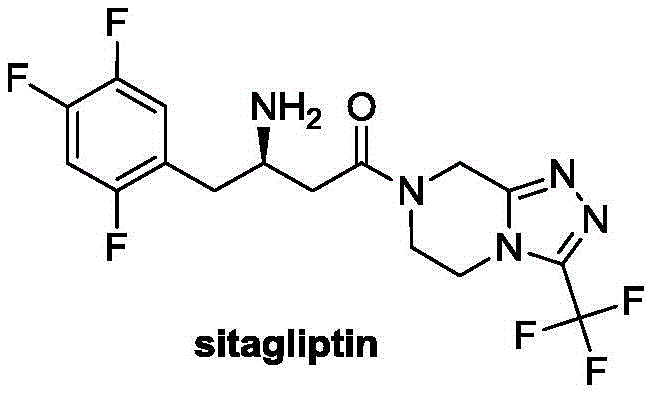
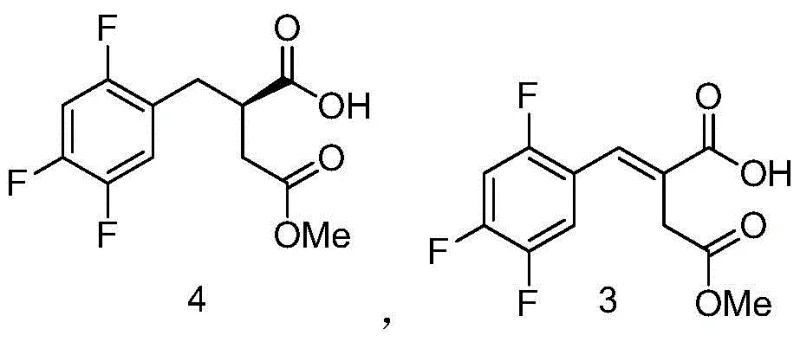
- Perform a Heck coupling reaction between 2,4,5-trifluorobromobenzene and monomethyl itaconic acid using a palladium catalyst and DBU base to form the alkene intermediate.
- Conduct asymmetric hydrogenation on the alkene intermediate using a chiral Iridium catalyst [Ir-((R)-DTB-SIPHOX)(COD)]BArF under hydrogen pressure to establish the chiral center with >99% ee.
- Convert the resulting chiral acid to the Boc-protected amine ester via acid chloride formation and azide substitution, followed by alkaline hydrolysis to yield the final amino acid intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route translates into tangible strategic benefits beyond simple unit cost metrics. The elimination of chiral resolution steps fundamentally alters the material balance of the process, effectively doubling the output from the same amount of starting chiral material or removing the need for expensive resolving agents entirely. This structural improvement in the process design leads to significant cost savings by reducing the volume of raw materials required per kilogram of finished intermediate. Furthermore, the robustness of the chemistry, which tolerates standard industrial conditions without requiring cryogenic temperatures or ultra-high pressures, enhances the reliability of supply. Manufacturers can scale this process with greater confidence, knowing that the reaction kinetics are predictable and the risk of batch failure due to sensitive operating windows is minimized.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this route is the removal of the resolution step, which traditionally discards half of the produced material. By achieving >99% ee directly through catalysis, the process maximizes atom economy and reduces the effective cost of the chiral building block. Additionally, the ability to perform the Heck reaction without protecting the carboxylic acid group saves the cost of protecting group reagents and the associated solvents and energy for their installation and removal. These cumulative efficiencies result in a leaner manufacturing process with lower variable costs, allowing for more competitive pricing in the generic pharmaceutical market without sacrificing margin.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by complex syntheses that rely on niche reagents or fragile reaction conditions. This methodology utilizes commodity chemicals such as 2,4,5-trifluorobromobenzene and monomethyl itaconic acid, which are readily available from multiple global sources, mitigating the risk of raw material shortages. The catalytic system, while specialized, is used in low quantities, reducing the logistical burden of sourcing large volumes of expensive chiral ligands. The operational simplicity of the steps, involving standard filtration and extraction workups, ensures that production timelines are predictable, reducing lead time for high-purity pharmaceutical intermediates and enabling manufacturers to respond more agilely to market demand fluctuations.
- Scalability and Environmental Compliance: Scaling chemical processes often exposes hidden inefficiencies related to heat transfer and waste management. This route is designed with scalability in mind, utilizing solvents like methanol and ethyl acetate that are easily recovered and recycled in standard distillation units. The reduction in synthetic steps directly correlates to a lower Process Mass Intensity (PMI), meaning less waste is generated per unit of product. This aligns with increasingly stringent environmental regulations and corporate sustainability goals. The absence of heavy metal contamination risks associated with less selective catalysts simplifies the purification train, ensuring that the final product meets rigorous heavy metal specifications required for pharmaceutical grade materials with less intensive treatment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for evaluating the feasibility of this route for your specific production needs. Understanding these nuances is critical for R&D teams planning technology transfer and for procurement teams assessing supplier capabilities.
Q: What is the enantiomeric excess (ee) achieved in this synthesis route?
A: The patented asymmetric hydrogenation step utilizing the specific Iridium catalyst system consistently achieves an ee value of greater than 99%, eliminating the need for chiral resolution.
Q: Does this process require protection of the carboxylic acid group during the initial coupling?
A: No, a key advantage of this methodology is that the carboxyl group of the starting itaconic acid derivative does not require protection during the Heck reaction, simplifying the workflow.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the process utilizes robust reaction conditions, standard solvents like methanol and dichloromethane, and avoids cryogenic temperatures, making it highly scalable for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sitagliptin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and robust infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this asymmetric hydrogenation route are fully realized in practice. We operate stringent purity specifications and maintain rigorous QC labs equipped to verify enantiomeric excess and impurity profiles at every stage of production. Our commitment to quality assurance means that every batch of Sitagliptin intermediate we produce is backed by comprehensive analytical data, giving our partners the confidence they need to file regulatory submissions and launch products without delay.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic impact of switching to this more efficient methodology. We encourage potential partners to contact us for specific COA data and route feasibility assessments tailored to your volume requirements. Let us help you optimize your API production strategy with a reliable partner dedicated to innovation and excellence in fine chemical manufacturing.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
