Revolutionizing Antibiotic Intermediate Production via One-Bacterium Multi-Enzyme Whole-Cell Catalysis
The pharmaceutical and veterinary industries rely heavily on the consistent supply of high-purity chiral building blocks, particularly for the synthesis of broad-spectrum antibiotics like florfenicol and thiamphenicol. A pivotal advancement in this domain is detailed in patent CN110951799B, which introduces a groundbreaking method for the whole-cell asymmetric synthesis of (2S,3R)-p-methylsulfonylphenylserine. This technology leverages a sophisticated "one-bacterium multi-enzyme" strategy, utilizing engineered Escherichia coli BL21(DE3) to co-express L-threonine transaldolase (PsLTTA), alcohol dehydrogenase (ApADH), and formate dehydrogenase (CbFDH). Unlike traditional chemical methods that struggle with harsh conditions and low theoretical yields, this biocatalytic approach operates under mild normal temperature and pressure conditions. It represents a paradigm shift towards greener, more efficient manufacturing, effectively addressing the critical bottlenecks of stereoselectivity and process complexity that have long plagued the production of beta-amino alcohol antibiotics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of (2S,3R)-p-methylsulfonylphenylserine has been dominated by chemical synthesis routes that are fraught with significant inefficiencies and environmental burdens. As illustrated in the reaction scheme below, the conventional pathway typically involves an aldol condensation between p-methylsulfonyl benzaldehyde and glycine in the presence of copper sulfate and ammonia water, followed by esterification and a final resolution step using D-tartaric acid. This multi-step process necessitates the use of strong acids and bases, creating severe safety hazards and generating substantial hazardous waste streams. Furthermore, the reliance on chiral resolution imposes a fundamental theoretical yield limit of 50%, meaning half of the synthesized material is discarded or requires energy-intensive recycling. The use of heavy metal catalysts like copper also introduces stringent purification requirements to meet residual metal specifications for pharmaceutical grades, adding further cost and complexity to the supply chain.
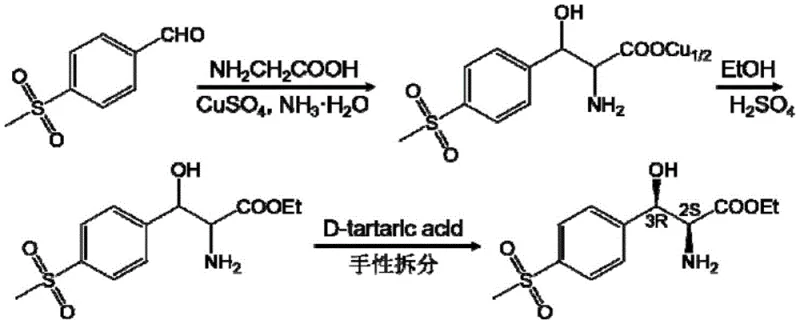
The Novel Approach
In stark contrast, the novel biocatalytic method disclosed in the patent circumvents these limitations through an elegant enzymatic cascade performed within a single microbial host. By integrating three distinct enzymatic functions into one organism, the process achieves a one-step conversion of readily available substrates—p-methylsulfonylbenzaldehyde and L-threonine—directly into the target chiral amino acid. This approach completely eliminates the need for toxic heavy metal catalysts and corrosive reagents, operating instead in a benign aqueous buffer system at physiological pH. Most critically, because the synthesis is asymmetric and enzyme-driven, it bypasses the 50% yield ceiling of resolution methods, driving substrate conversion to near completion (>99%). This transition from a linear, wasteful chemical process to a circular, high-efficiency biological system offers a compelling value proposition for manufacturers seeking to optimize both their environmental footprint and their bottom line.
Mechanistic Insights into the Tri-Enzyme Cascade System
The core innovation of this technology lies in the synergistic interaction of the three co-expressed enzymes within the engineered E. coli cells. The primary catalyst, L-threonine transaldolase (PsLTTA), is responsible for the stereoselective formation of the carbon-carbon bond between the aldehyde and the amino acid precursor, establishing the critical (2S,3R) configuration with exceptional precision. However, a major challenge in transaldolase reactions is the accumulation of acetaldehyde, a byproduct that acts as a potent inhibitor of the enzyme, stalling the reaction. The patent solves this by co-expressing alcohol dehydrogenase (ApADH), which acts as a scavenger, rapidly reducing the inhibitory acetaldehyde into harmless ethanol. This in situ removal of the inhibitor not only protects the PsLTTA activity but also shifts the reaction equilibrium towards the product side according to Le Chatelier's principle, thereby maximizing yield.
Completing this metabolic loop is the third enzyme, formate dehydrogenase (CbFDH), which plays a vital role in cofactor regeneration. The reduction of acetaldehyde by ApADH consumes the expensive reduced coenzyme I (NADH). To make the process economically viable on an industrial scale, NADH must be continuously regenerated. CbFDH achieves this by oxidizing inexpensive sodium formate into carbon dioxide, simultaneously reducing NAD+ back to NADH. This self-sustaining cofactor cycle ensures that only catalytic amounts of the expensive cofactor are needed, drastically reducing raw material costs. The result is a robust, self-regulating biocatalytic system that maintains high activity over extended periods, delivering product with an enantiomeric excess (ee) greater than 99.9% and a diastereomeric excess (de) of 94.1%, metrics that are difficult to achieve consistently with chemical catalysis.
How to Synthesize (2S,3R)-p-Methylsulfonylphenylserine Efficiently
Implementing this advanced biocatalytic route requires precise control over strain construction and fermentation parameters to ensure optimal enzyme expression ratios. The process begins with the transformation of the host bacteria with dual plasmids carrying the genes for the three target enzymes, followed by a controlled fermentation phase to build up biomass and induce protein expression. Once the whole-cell biocatalyst is prepared, the synthesis itself is remarkably straightforward, involving the mixing of substrates with the wet cell paste in a buffered solution. For a comprehensive understanding of the specific operational parameters, including induction temperatures, substrate feeding strategies, and downstream isolation techniques, please refer to the detailed technical guide below.
- Construct double-plasmid engineering bacteria by transforming E. coli BL21(DE3) with pET28a-PsLTTA and pETDuet-ApADH/CbFDH plasmids.
- Culture the engineered bacteria in LB medium with antibiotics, induce protein expression with IPTG at OD600 0.5, and harvest cells via centrifugation.
- Perform the asymmetric synthesis by reacting p-methylsulfonylbenzaldehyde and L-threonine with the wet whole cells in Tris-HCl buffer containing sodium formate and cofactors.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this whole-cell biocatalytic technology translates into tangible strategic advantages beyond mere technical superiority. The shift from a multi-step chemical synthesis involving hazardous reagents to a single-step aqueous enzymatic process fundamentally alters the cost structure and risk profile of manufacturing this key antibiotic intermediate. By removing the need for expensive chiral resolving agents like D-tartaric acid and eliminating the complex purification steps associated with heavy metal removal, the overall production cost is significantly reduced. Furthermore, the high stereoselectivity of the enzymatic route minimizes the formation of unwanted isomers, leading to a much cleaner crude product that requires less solvent and energy for final purification, thereby enhancing the overall process mass intensity.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the elimination of costly resolution steps and the maximization of raw material utilization. Unlike chemical resolution which inherently wastes 50% of the material, this asymmetric synthesis converts nearly all starting materials into the desired product, effectively doubling the theoretical output from the same amount of feedstock. Additionally, the use of whole cells obviates the need for expensive enzyme purification protocols; the bacteria serve as their own reaction vessels, saving significant capital and operational expenditure on chromatography columns and purification buffers. The regeneration of the NADH cofactor via the formate dehydrogenase system further ensures that the consumption of high-value reagents remains minimal, contributing to a leaner and more cost-effective manufacturing model.
- Enhanced Supply Chain Reliability: From a supply chain perspective, the simplicity and robustness of the whole-cell process offer greater predictability and shorter lead times. The reaction conditions are mild (normal temperature and pressure), reducing the dependency on specialized high-pressure reactors or extreme cooling systems that can be prone to maintenance issues and downtime. The starting materials, p-methylsulfonylbenzaldehyde and L-threonine, are commodity chemicals with stable global supply chains, mitigating the risk of raw material shortages. Moreover, the high conversion rate (>99%) ensures consistent batch-to-batch quality and yield, allowing for more accurate production planning and inventory management, which is crucial for meeting the rigorous delivery schedules of multinational pharmaceutical clients.
- Scalability and Environmental Compliance: Scaling biocatalytic processes is often more straightforward than scaling complex chemical syntheses involving hazardous intermediates. The aqueous nature of the reaction mixture simplifies heat transfer and mixing in large-scale fermenters, facilitating a smoother transition from pilot plant to commercial production volumes. Environmentally, this technology aligns perfectly with modern green chemistry principles by replacing toxic solvents and heavy metals with water and biodegradable enzymes. This significantly reduces the burden on wastewater treatment facilities and lowers the costs associated with hazardous waste disposal and regulatory compliance. For companies aiming to reduce their carbon footprint and meet stringent ESG (Environmental, Social, and Governance) goals, this biosynthetic route offers a clear pathway to sustainable manufacturing without compromising on productivity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this whole-cell biocatalytic technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific production needs. Understanding these nuances is essential for making informed decisions about technology transfer and process optimization.
Q: How does the 'one-bacterium multi-enzyme' system improve yield compared to traditional chemical resolution?
A: Traditional chemical resolution using D-tartaric acid is theoretically limited to a maximum 50% yield for the desired enantiomer. In contrast, the patented whole-cell asymmetric synthesis utilizes a cascade of L-threonine transaldolase, alcohol dehydrogenase, and formate dehydrogenase to drive the reaction stereo-specifically, achieving substrate conversion rates exceeding 99% and eliminating the 50% yield ceiling inherent in resolution processes.
Q: What are the specific advantages of using whole-cell catalysis over purified enzyme systems?
A: Using whole cells eliminates the complex, time-consuming, and costly downstream processing steps required to separate and purify individual enzymes. The cellular matrix provides a natural protective environment for the enzymes, enhancing their stability and allowing for direct reuse or simplified handling, which significantly reduces operational expenditure and simplifies the manufacturing workflow.
Q: How does the system manage the inhibitory byproduct acetaldehyde?
A: The system employs a clever metabolic coupling strategy where the co-expressed alcohol dehydrogenase (ApADH) immediately converts the inhibitory byproduct acetaldehyde into ethanol. This not only prevents the accumulation of acetaldehyde which would otherwise inhibit the primary transaldolase enzyme but also drives the reaction equilibrium forward, ensuring high efficiency and continuous catalytic turnover.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (2S,3R)-p-Methylsulfonylphenylserine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced biocatalysis in the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate complex laboratory innovations like the "one-bacterium multi-enzyme" system into robust, commercial-scale manufacturing processes. Our facilities are equipped to handle diverse synthetic pathways, ranging from traditional chemical synthesis to cutting-edge enzymatic cascades, with the capability to scale production seamlessly from 100 kgs to 100 MT/annual commercial production. We maintain stringent purity specifications and operate rigorous QC labs to ensure that every batch of (2S,3R)-p-methylsulfonylphenylserine meets the exacting standards required for antibiotic synthesis, guaranteeing consistency and reliability for your supply chain.
We invite you to collaborate with us to explore how this innovative technology can optimize your production costs and enhance your product quality. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact our technical procurement team today to request specific COA data, discuss route feasibility assessments, and discover how we can support your long-term strategic goals in the competitive antibiotic market.
