Advanced Biocatalytic Synthesis of (2S,3R)-p-Methylsulfonylphenylserine for Antibiotic Production
Advanced Biocatalytic Synthesis of (2S,3R)-p-Methylsulfonylphenylserine for Antibiotic Production
The pharmaceutical industry is constantly seeking more efficient and sustainable routes for the production of critical antibiotic intermediates, particularly for broad-spectrum agents like thiamphenicol and florfenicol. Patent CN110577948B introduces a groundbreaking biocatalytic approach utilizing a novel L-threonine aldolase derived from Actinocorallia herbida to synthesize (2S,3R)-p-methylsulfonylphenylserine with exceptional stereoselectivity. This technology represents a significant leap forward from traditional chemical synthesis, offering a pathway that is not only environmentally benign but also capable of achieving high conversion rates and purity levels essential for modern Good Manufacturing Practice (GMP) standards. By leveraging genetic engineering to express this specific enzyme in Escherichia coli, the process facilitates a direct asymmetric aldol condensation between p-methylsulfonylbenzaldehyde and glycine, effectively constructing two adjacent chiral centers in a single enzymatic step.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of chiral intermediates for beta-amino alcohol antibiotics has relied heavily on chemical resolution strategies, which are inherently inefficient and environmentally burdensome. The traditional route typically involves an aldol condensation between p-methylsulfonylbenzaldehyde and glycine catalyzed by copper sulfate and ammonia water at elevated temperatures around 70°C, followed by esterification and chiral resolution using D-tartaric acid. This multi-step process suffers from a fundamental theoretical yield limitation of merely 50%, as the resolution step discards the unwanted enantiomer, leading to significant material waste. Furthermore, the reliance on heavy metal catalysts like copper sulfate generates substantial quantities of toxic wastewater, creating severe environmental compliance challenges and increasing the cost of waste treatment for manufacturers. The harsh reaction conditions and the need for multiple purification steps also contribute to higher operational costs and longer production cycles, making the supply chain vulnerable to disruptions and regulatory scrutiny regarding heavy metal residues in the final active pharmaceutical ingredients.
The Novel Approach
In stark contrast, the biocatalytic method disclosed in the patent utilizes a highly specific L-threonine aldolase to drive the asymmetric synthesis under mild, aqueous conditions, effectively bypassing the limitations of chemical resolution. This enzymatic route operates at a neutral pH of 7.0 and a moderate temperature of 30°C, eliminating the need for hazardous reagents and extreme thermal inputs. The use of recombinant E. coli whole cells as the biocatalyst simplifies the downstream processing significantly, as there is no requirement for cell disruption or enzyme purification; the substrates penetrate the cell membrane to react with the intracellular enzyme, and the product is excreted into the supernatant. This whole-cell catalysis strategy not only enhances the stability of the enzyme but also streamlines the manufacturing workflow, reducing the number of unit operations and minimizing the risk of contamination. The result is a robust, green chemistry solution that aligns perfectly with the industry's shift towards sustainable manufacturing practices while delivering superior stereochemical control.
Mechanistic Insights into L-Threonine Aldolase Catalyzed Asymmetric Aldol Reaction
The core of this technological advancement lies in the unique catalytic mechanism of the L-threonine aldolase (EC 4.1.2.5), a pyridoxal phosphate (PLP)-dependent enzyme that facilitates the formation of carbon-carbon bonds with high precision. The enzyme functions by forming a Schiff base intermediate with the amino group of glycine, generating a nucleophilic quinonoid species that attacks the carbonyl carbon of the aldehyde substrate, specifically p-methylsulfonylbenzaldehyde in this application. The active site of the L-threonine aldolase derived from Actinocorallia herbida is structurally tuned to favor the formation of the (2S,3R) configuration, which is the bioactive isomer required for the synthesis of thiamphenicol and florfenicol. This stereospecificity is achieved through precise spatial arrangement within the enzyme's binding pocket, which restricts the approach of the substrate and stabilizes the transition state leading to the desired diastereomer. The patent data indicates that this specific enzyme variant exhibits a remarkable enantiomeric excess (ee) of greater than 99.9% and a diastereomeric excess (de) of 62.3%, demonstrating its superior ability to discriminate between potential stereoisomers compared to other reported aldolases.
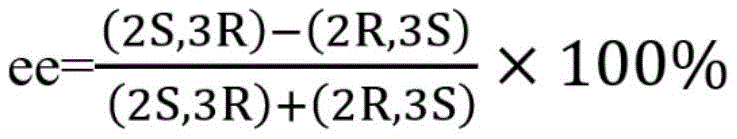
Furthermore, the impurity profile of the reaction is tightly controlled by the enzyme's inherent selectivity, minimizing the formation of the (2S,3S) diastereomer and other undesired byproducts that are common in non-enzymatic aldol reactions. The presence of organic co-solvents, specifically 30% acetonitrile, plays a crucial role in enhancing the solubility of the hydrophobic aldehyde substrate while maintaining the structural integrity and activity of the biocatalyst. Optimization studies revealed that maintaining a substrate concentration of 500mM and a cell concentration of 12.5mg/mL allows the system to achieve a substrate conversion rate of up to 75.7% within just 6 hours. This high efficiency is attributed to the engineered stability of the recombinant enzyme and the optimized mass transfer characteristics of the whole-cell system, ensuring that the reaction proceeds rapidly to completion without significant accumulation of toxic intermediates or side products that could inhibit the catalyst.
How to Synthesize (2S,3R)-p-Methylsulfonylphenylserine Efficiently
The implementation of this biocatalytic process requires a systematic approach to strain construction and reaction engineering to ensure consistent quality and yield at scale. The patent outlines a clear methodology starting with the codon optimization of the L-threonine aldolase gene to enhance expression levels in the E. coli host, followed by the construction of a stable recombinant plasmid vector. Once the production strain is established, the focus shifts to optimizing the fermentation and biotransformation conditions, where parameters such as induction temperature, pH, and co-solvent ratio are fine-tuned to maximize catalytic performance. For detailed operational protocols regarding the specific media compositions, induction timelines, and downstream isolation techniques, please refer to the standardized synthesis guide below.
- Construct a recombinant expression plasmid containing the optimized L-threonine aldolase gene derived from Actinocorallia herbida and transform it into E. coli BL21(DE3) competent cells.
- Culture the recombinant bacteria in LB medium with kanamycin, induce protein expression with IPTG at 20°C, and harvest the whole cells via centrifugation.
- Perform the asymmetric aldol reaction in a phosphate buffer containing 30% acetonitrile, p-methylsulfonylbenzaldehyde, glycine, and pyridoxal phosphate at 30°C for 6 hours.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this enzymatic technology offers profound strategic advantages that extend beyond simple technical metrics, fundamentally altering the cost structure and reliability of the supply chain for antibiotic intermediates. By transitioning from a resolution-based chemical process to a direct asymmetric biosynthesis, manufacturers can effectively double the theoretical yield of the target isomer, as the process is not limited by the 50% ceiling inherent in racemic resolution. This dramatic improvement in atom economy translates directly into substantial raw material savings, as less starting aldehyde and glycine are required to produce the same amount of final product, thereby insulating the supply chain from volatility in precursor pricing. Additionally, the elimination of heavy metal catalysts like copper sulfate removes the need for expensive and complex wastewater treatment systems designed to handle toxic metal effluents, resulting in significant operational expenditure reductions and a smaller environmental footprint.
- Cost Reduction in Manufacturing: The shift to a whole-cell biocatalytic system eliminates several costly unit operations associated with traditional chemical synthesis, including high-temperature heating, hazardous esterification steps, and the consumption of chiral resolving agents like D-tartaric acid. Since the enzyme acts as a reusable biological catalyst within the whole cell, the recurring cost of chiral auxiliaries is removed entirely, and the mild reaction conditions reduce energy consumption for heating and cooling. The simplified downstream processing, which avoids the need for enzyme purification and complex extraction steps required to remove heavy metals, further drives down the cost of goods sold, making the final intermediate more price-competitive in the global market.
- Enhanced Supply Chain Reliability: Relying on a genetically engineered microbial host for production provides a highly scalable and consistent source of the catalyst, independent of the fluctuations often seen in the supply of specialized chemical reagents or natural extracts. The robustness of the recombinant E. coli strain ensures that production can be ramped up quickly to meet surges in demand for thiamphenicol and florfenicol, which are critical medicines in both human and veterinary sectors. Furthermore, the high stereoselectivity of the process reduces the risk of batch failures due to poor optical purity, ensuring a steady flow of compliant material that meets stringent regulatory specifications without the need for re-processing or rejection.
- Scalability and Environmental Compliance: The aqueous nature of the reaction system, supplemented with a manageable amount of acetonitrile, aligns perfectly with green chemistry principles, facilitating easier regulatory approval and reducing the burden of environmental compliance reporting. The process generates significantly less hazardous waste compared to the copper-laden sludge produced by conventional methods, simplifying disposal logistics and lowering the risk of environmental liabilities. This eco-friendly profile not only enhances the corporate sustainability image of the manufacturer but also future-proofs the supply chain against increasingly strict global environmental regulations regarding industrial effluent and heavy metal discharge.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of integrating this biocatalytic route into their existing manufacturing frameworks, we have compiled answers to common inquiries regarding the process specifics and performance metrics. These responses are derived directly from the experimental data and technical disclosures within the patent documentation, providing a transparent view of the technology's capabilities and limitations. Understanding these details is crucial for R&D directors and process engineers who are responsible for validating new synthetic routes and ensuring they meet the rigorous quality standards required for pharmaceutical production.
Q: What are the stereochemical advantages of using L-threonine aldolase over chemical resolution?
A: The enzymatic route achieves an enantiomeric excess (ee) of greater than 99.9% and a diastereomeric excess (de) of 62.3% for the (2S,3R) isomer, significantly surpassing the theoretical 50% yield limit of traditional chemical resolution methods.
Q: How does the whole-cell catalysis method simplify the production process?
A: By using recombinant E. coli whole cells as biocatalysts, the process eliminates the need for complex cell disruption and enzyme purification steps, allowing substrates to penetrate the cell membrane and react directly with the intracellular enzyme.
Q: What are the optimal reaction conditions for maximizing substrate conversion?
A: Optimal conditions include a substrate concentration of 500mM p-methylsulfonylbenzaldehyde, a cell concentration of 12.5mg/mL, 30% acetonitrile as a co-solvent, and a reaction temperature of 30°C maintained for 6 hours.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (2S,3R)-p-Methylsulfonylphenylserine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this L-threonine aldolase technology in securing a sustainable and cost-effective supply of critical antibiotic intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory-scale enzymatic synthesis to industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced fermentation capabilities designed to handle sensitive biocatalytic processes, guaranteeing that every batch of (2S,3R)-p-methylsulfonylphenylserine meets stringent purity specifications and regulatory requirements for global markets.
We invite pharmaceutical companies and agrochemical manufacturers to collaborate with us to leverage this innovative green synthesis route for their supply chains. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this enzymatic process for your specific volume needs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that enhance both your product quality and your bottom line.
