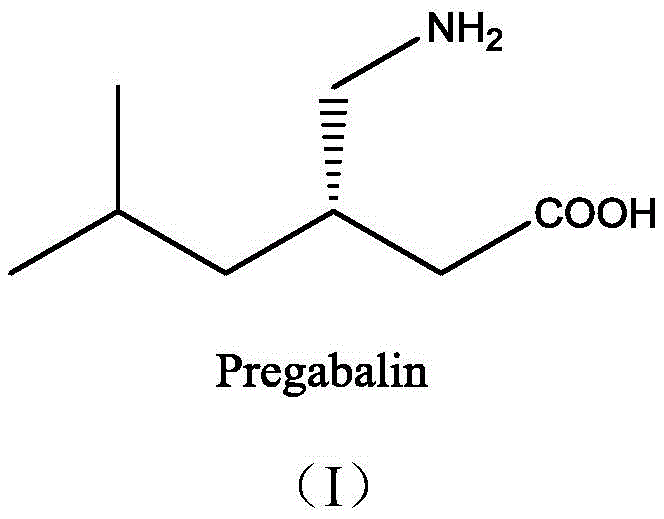
Advanced Biocatalytic Route for High-Purity Pregabalin Chiral Intermediate Manufacturing
The global demand for Pregabalin, a potent GABA analog used extensively in the treatment of neuropathic pain and epilepsy, continues to drive innovation in its manufacturing supply chain. As regulatory standards tighten and cost pressures mount, the industry is shifting away from traditional chemical resolution methods toward more sustainable and precise biocatalytic solutions. Patent CN108715881B introduces a groundbreaking regio- and stereoselective biocatalytic synthesis method that addresses the critical bottlenecks of yield and optical purity in producing the key chiral intermediate, (S)-3-cyano-5-methylhexanoic acid. This technology leverages a sophisticated dual-enzyme system comprising specific nitrilases and amidases to convert racemic isobutyl succinonitrile (IBSN) directly into the target acid under mild aqueous conditions. By achieving a conversion rate approaching the theoretical maximum of 50% for kinetic resolution and an exceptional enantiomeric excess (e.e.) of 99.5%, this process represents a significant leap forward for reliable pharmaceutical intermediate supplier networks seeking to optimize their API production lines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for Pregabalin intermediates have long been plagued by inefficiencies that inflate production costs and complicate waste management. Conventional chemical hydrolysis often requires harsh acidic or basic conditions, elevated temperatures, and the use of heavy metal catalysts, which necessitate complex downstream purification to meet stringent residual metal specifications for pharmaceutical ingredients. Furthermore, earlier biocatalytic attempts using single nitrilase enzymes frequently suffered from the accumulation of the intermediate amide, (S)-3-cyano-5-methylhexanamide. This byproduct not only reduces the overall yield of the desired carboxylic acid but also creates a challenging separation problem, as the amide and acid possess similar physicochemical properties. Previous patents, such as CN1942587B, reported yields struggling to exceed 43% even after extensive optimization, with optical purity often fluctuating below the 99% threshold required for high-grade API manufacturing, thereby forcing manufacturers to implement costly recrystallization steps.
The Novel Approach
The innovative methodology disclosed in the patent data overcomes these historical hurdles through a synergistic one-pot dual-enzyme cascade. By strictly coupling a highly stereoselective nitrilase with a complementary amidase, the process ensures that any amide intermediate formed is immediately hydrolyzed into the final carboxylic acid product. This eliminates the accumulation of the amide byproduct entirely, simplifying the reaction profile to a clean conversion of substrate to product. The reaction operates under remarkably mild conditions, typically between 20°C and 50°C, in a simple buffered aqueous medium, which drastically reduces energy consumption compared to thermal chemical processes. Most critically, this approach achieves a conversion rate of 49.5% with an e.e. of 99.5%, effectively maximizing the theoretical yield of the kinetic resolution while delivering a product of exceptional optical purity that minimizes the need for further chiral enrichment.
Mechanistic Insights into Dual-Enzyme Cascade Catalysis
The core of this technological advancement lies in the precise selection and pairing of enzyme variants that exhibit complementary activities and stereoselectivities. The nitrilase component, specifically variants like AaNIT derived from Arabis alpina, demonstrates a profound preference for the (S)-enantiomer of the dinitrile substrate, initiating the hydrolysis of one cyano group to form the chiral acid. However, nitrilases inherently possess varying degrees of nitrile hydratase activity, which can lead to the formation of the corresponding amide. In this novel system, the addition of a robust amidase, such as Pa-Ami from Pantoea sp., acts as a scavenger for this amide intermediate. The amidase rapidly hydrolyzes the amide bond, converting the potential impurity into additional quantities of the desired (S)-3-cyano-5-methylhexanoic acid. This cooperative catalysis not only drives the reaction equilibrium forward but also ensures that the final product stream is virtually free of amide contaminants, streamlining the isolation process.
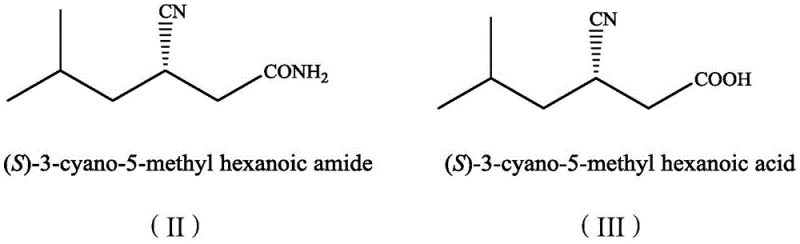
From an impurity control perspective, the mechanism offers distinct advantages for maintaining high product quality throughout the batch cycle. In single-enzyme systems, the accumulation of the amide can lead to competitive inhibition or non-selective hydrolysis that degrades optical purity over time. By maintaining a low steady-state concentration of the amide through immediate conversion by the amidase, the dual-enzyme system preserves the stereochemical integrity of the chiral center. Experimental data confirms that optimizing the mass ratio of nitrilase-containing cells to amidase-containing cells (e.g., ratios between 1:1 and 12:1) allows for fine-tuning of the reaction kinetics. This ensures that the rate of amide formation never exceeds the rate of its consumption, resulting in a clean reaction profile where the only significant organic components remaining at the end of the 4 to 10-hour cycle are the unreacted (R)-substrate and the high-purity (S)-product acid.
How to Synthesize (S)-3-cyano-5-methylhexanoic acid Efficiently
Implementing this biocatalytic route requires careful preparation of the biocatalysts and precise control of the reaction environment to maximize the synergistic effects of the enzyme pair. The process begins with the cultivation of recombinant E. coli strains expressing the specific nitrilase and amidase genes, followed by harvesting the wet cell biomass which serves as the source of catalytic activity. The transformation is conducted in a buffered aqueous system, preferably using Tris-HCl at a pH between 7.5 and 9.0, which maintains optimal enzyme stability and activity. Substrate loading is a critical parameter, with the patent demonstrating the capability to handle initial IBSN concentrations up to 150 g/L without significant loss of efficiency, a feature that is vital for commercial viability. The detailed standardized synthesis steps for replicating this high-efficiency protocol are outlined in the guide below.
- Prepare recombinant E. coli cells expressing specific nitrilase (e.g., AaNIT) and amidase (e.g., Pa-Ami) enzymes.
- Mix wet cell biomass with racemic isobutyl succinonitrile (IBSN) in a Tris-HCl buffer system at pH 7.5-9.0.
- Maintain reaction temperature between 20-50°C for 4-10 hours to achieve near-theoretical conversion with >99% optical purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this dual-enzyme biocatalytic process translates into tangible strategic benefits regarding cost structure and supply reliability. The elimination of harsh chemical reagents and heavy metal catalysts removes the need for expensive scavenging resins and complex wastewater treatment protocols associated with metal removal, leading to substantial cost savings in utility and waste disposal. Furthermore, the ability to operate at high substrate concentrations significantly improves volumetric productivity, meaning that the same reactor volume can produce more product per batch, effectively lowering the fixed cost allocation per kilogram of API intermediate. This efficiency gain is compounded by the simplified downstream processing; because the reaction does not accumulate difficult-to-separate amide byproducts, the isolation of the final acid can be achieved with fewer unit operations, reducing both processing time and solvent consumption.
- Cost Reduction in Manufacturing: The primary driver for cost reduction in pharmaceutical intermediate manufacturing with this technology is the drastic simplification of the purification train. By preventing the formation of the amide byproduct, the process eliminates the need for specialized chromatographic separations or multiple recrystallization steps that are typically required to remove structurally similar impurities. Additionally, the use of whole-cell biocatalysts avoids the high capital expenditure associated with immobilizing purified enzymes on expensive carriers, allowing for a more flexible and lower-cost catalyst replacement strategy. The mild reaction temperatures also contribute to lower energy overheads compared to thermal chemical hydrolysis methods.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the robustness of the enzymatic process, which tolerates high substrate loadings and utilizes readily available racemic starting materials. Unlike processes dependent on scarce chiral pool precursors or unstable organometallic catalysts, this biocatalytic route relies on fermentable enzymes and bulk chemicals, insulating the supply chain from volatility in specialty reagent markets. The high stereoselectivity ensures consistent product quality batch-after-batch, reducing the risk of production delays caused by out-of-specification results that require re-processing or scrapping.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the use of aqueous media and standard fermentation-derived biocatalysts, which are inherently safer and easier to handle than pyrophoric or toxic chemical reagents. The environmental profile is significantly improved due to the absence of heavy metals and the reduction in organic solvent usage during workup, aligning with increasingly strict global environmental regulations and corporate sustainability goals. This green chemistry profile not only mitigates regulatory risk but also enhances the marketability of the final API to environmentally conscious partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this dual-enzyme biocatalytic technology. These insights are derived directly from the experimental data and process parameters detailed in the patent documentation, providing a clear understanding of the method's capabilities and limitations. Understanding these specifics is crucial for R&D teams evaluating the feasibility of technology transfer and for procurement teams assessing the long-term viability of the supply source.
Q: How does the dual-enzyme system improve optical purity compared to single nitrilase catalysis?
A: Single nitrilase catalysis often results in the accumulation of the amide intermediate ((S)-3-cyano-5-methylhexanamide), which complicates purification. The patented dual-enzyme system couples nitrilase with a specific amidase that immediately hydrolyzes this amide byproduct into the desired acid, preventing accumulation and driving the reaction toward higher yield and 99.5% e.e.
Q: What are the substrate loading capabilities of this biocatalytic process?
A: Unlike many biocatalytic processes that suffer from substrate inhibition at high concentrations, this method supports initial substrate concentrations of racemic IBSN up to 150 g/L. This high loading capacity significantly improves volumetric productivity, making it highly suitable for industrial-scale manufacturing.
Q: Why is the selection of specific enzyme variants like AaNIT and Pa-Ami critical?
A: Screening data indicates that enzyme variants vary significantly in stereoselectivity and side-reaction profiles. AaNIT demonstrates superior stereoselectivity for the (S)-enantiomer with minimal amide production, while Pa-Ami exhibits high activity for converting the residual amide. This specific pairing ensures the process avoids the formation of difficult-to-remove impurities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pregabalin Intermediate Supplier
As the pharmaceutical industry continues to evolve towards more sustainable and efficient manufacturing paradigms, NINGBO INNO PHARMCHEM stands at the forefront of translating advanced biocatalytic research into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising metrics observed in laboratory settings are faithfully reproduced in large-scale reactors. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify enantiomeric excess and impurity profiles, guaranteeing that every batch of Pregabalin intermediate meets the exacting standards required for global regulatory filings.
We invite forward-thinking partners to collaborate with us to leverage this cutting-edge technology for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this biocatalytic route for your specific production volumes. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that enhance both the profitability and sustainability of your API manufacturing operations.
