Industrial Scale Production of High-Purity 3-Aminopyrrolidine Salts via Novel Optical Resolution
Industrial Scale Production of High-Purity 3-Aminopyrrolidine Salts via Novel Optical Resolution
The pharmaceutical and agrochemical industries increasingly demand chiral building blocks with exceptional optical purity to ensure the efficacy and safety of final drug products. Patent CN101910124A introduces a groundbreaking methodology for the industrial production of optically active 3-aminopyrrolidine (3AP), a critical heterocyclic intermediate used in the synthesis of various bioactive molecules. This technology addresses the long-standing challenges associated with the low optical purity and complex processing inherent in previous manufacturing routes. By leveraging a specific diastereomeric salt formation strategy using optically active 2-methoxyphenylacetic acid (MPAA) in an aqueous environment, this process achieves high enantiomeric excess and yield simultaneously. For R&D directors and procurement specialists, this represents a significant shift towards more sustainable and cost-effective chiral synthesis, eliminating the need for hazardous reagents and complex multi-step protections often required in alternative pathways.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of optically active 3-aminopyrrolidine has been plagued by inefficient routes that compromise both economic viability and operational safety. One prominent conventional method involves the derivation from 4-hydroxyproline, which necessitates a cumbersome sequence of decarboxylation, N-Boc protection, O-mesylation, and subsequent azide reduction. 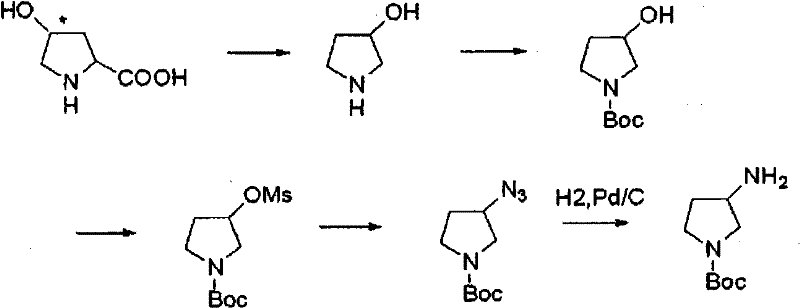 As illustrated in the reaction scheme, this pathway requires multiple isolation steps and the use of potentially explosive azide intermediates, creating significant safety liabilities and driving up production costs due to low overall throughput. Furthermore, enzymatic approaches using transaminases have demonstrated limited success, often yielding moderate enantiomeric excess (e.g., 88% ee) and requiring expensive cofactor regeneration systems. Another traditional resolution method utilizing L-tartaric acid suffers from unpredictable crystallization behavior, where mixed stereoisomer salts precipitate sequentially, making it difficult to isolate the target enantiomer with consistent high purity on a commercial scale.
As illustrated in the reaction scheme, this pathway requires multiple isolation steps and the use of potentially explosive azide intermediates, creating significant safety liabilities and driving up production costs due to low overall throughput. Furthermore, enzymatic approaches using transaminases have demonstrated limited success, often yielding moderate enantiomeric excess (e.g., 88% ee) and requiring expensive cofactor regeneration systems. Another traditional resolution method utilizing L-tartaric acid suffers from unpredictable crystallization behavior, where mixed stereoisomer salts precipitate sequentially, making it difficult to isolate the target enantiomer with consistent high purity on a commercial scale.
The Novel Approach
In stark contrast to these convoluted strategies, the novel approach detailed in the patent utilizes a direct optical resolution of racemic 3-aminopyrrolidine using (S)- or (R)-2-methoxyphenylacetic acid (MPAA). This method capitalizes on the formation of a highly stable diastereomeric salt with a defined 1:2 stoichiometry between the amine and the resolving agent. 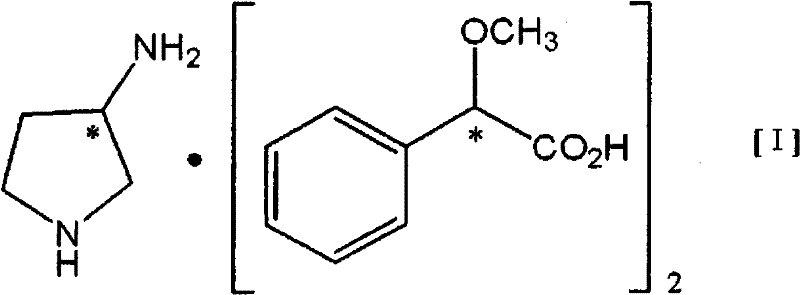 The innovation lies in the specific reaction conditions: conducting the resolution in water, preferably with the controlled addition of a mineral acid like hydrochloric acid. This aqueous system not only simplifies the workup by avoiding organic solvent extraction but also enhances the crystallization kinetics, allowing for the precipitation of the target salt with optical purities exceeding 98% directly from the reactor. The ability to tune the optical purity to over 99% through a simple water recrystallization step further underscores the robustness of this technique, providing a reliable supply of high-quality intermediates for downstream API synthesis.
The innovation lies in the specific reaction conditions: conducting the resolution in water, preferably with the controlled addition of a mineral acid like hydrochloric acid. This aqueous system not only simplifies the workup by avoiding organic solvent extraction but also enhances the crystallization kinetics, allowing for the precipitation of the target salt with optical purities exceeding 98% directly from the reactor. The ability to tune the optical purity to over 99% through a simple water recrystallization step further underscores the robustness of this technique, providing a reliable supply of high-quality intermediates for downstream API synthesis.
Mechanistic Insights into MPAA-Mediated Diastereomeric Salt Formation
The core mechanism driving this high-efficiency resolution is the preferential crystallization of a specific diastereomeric salt complex. When racemic 3-aminopyrrolidine interacts with optically active MPAA in the presence of an inorganic acid, the system favors the formation of a lattice structure comprising one mole of the amine and two moles of the acid. This 1:2 stoichiometry is critical; it creates a unique crystal packing arrangement that is significantly less soluble in the aqueous medium compared to the undesired enantiomeric salt or the free base. The presence of the methoxy group on the phenyl ring of the resolving agent likely contributes to specific steric and electronic interactions that stabilize this particular diastereomer, effectively locking the desired chirality into the solid phase while leaving the unwanted enantiomer in the mother liquor. This thermodynamic preference is what allows for such high initial optical purity without the need for iterative recrystallizations typically seen in less selective resolution processes.
Furthermore, the role of the inorganic acid, specifically hydrochloric acid, cannot be overstated in terms of process optimization. The patent data reveals that adding HCl adjusts the protonation state of the amine and the solubility product of the salt, acting as a 'yield booster' without compromising optical purity. In the absence of acid, while optical purity remains high, the yield drops precipitously because the salt remains too soluble. By carefully controlling the molar ratio of HCl to MPAA (optimally summing to roughly 2.0 equivalents relative to the amine), the process engineers can manipulate the supersaturation point. This ensures that the maximum amount of the target diastereomer precipitates out during the cooling phase (from roughly 60°C down to 20°C), thereby maximizing the resolution efficiency metric, which is a function of both yield and enantiomeric excess. This precise control over solution chemistry is what transforms a standard resolution into a highly efficient industrial process.
How to Synthesize Optically Active 3-Aminopyrrolidine Salt Efficiently
The synthesis of this high-value chiral intermediate follows a streamlined protocol designed for reproducibility and scale. The process begins with the preparation of an aqueous reaction mixture containing the racemic amine source and the chiral resolving agent, followed by precise thermal management to induce crystallization. The detailed standardized synthesis steps, including specific molar ratios, temperature ramps, and agitation speeds required for GMP compliance, are outlined in the technical guide below.
- React racemic 3-aminopyrrolidine (free base or dihydrochloride) with optically active 2-methoxyphenylacetic acid (MPAA) in water, utilizing hydrochloric acid to adjust pH and facilitate salt formation.
- Control the reaction temperature by initially heating to 50-70°C for dissolution, followed by slow cooling to 15-25°C to induce crystallization of the specific diastereomeric salt.
- Isolate the crystals via centrifugation or filtration, optionally recrystallize in water to achieve >99% optical purity, and liberate the free amine using inorganic base.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this MPAA-based aqueous resolution process offers tangible strategic advantages that directly impact the bottom line and operational resilience. The elimination of organic solvents like ethanol or methanol in favor of water drastically reduces raw material costs and mitigates the regulatory burdens associated with volatile organic compound (VOC) emissions. Additionally, the simplified downstream processing—requiring only filtration and washing rather than complex distillation or chromatography—shortens the production cycle time, allowing for faster turnaround on customer orders and improved inventory turnover rates. This efficiency translates into a more competitive pricing structure for the final 3-aminopyrrolidine intermediate, making it an attractive option for cost-sensitive generic drug manufacturing programs.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of expensive organic solvents with water and the reduction of unit operations. Traditional routes often require multiple protection and deprotection steps, each adding reagent costs and labor hours; this direct resolution bypasses those entirely. Furthermore, the high yield and optical purity achieved in the first crystallization reduce the need for costly recycling of mother liquors or extensive purification campaigns. By minimizing the consumption of energy-intensive distillation equipment and reducing the volume of hazardous waste requiring specialized disposal, the overall cost of goods sold (COGS) is significantly lowered, providing a clear margin advantage for large-scale production runs.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the use of readily available, commodity-grade starting materials. Racemic 3-aminopyrrolidine and 2-methoxyphenylacetic acid are commercially accessible from multiple global suppliers, reducing the risk of single-source bottlenecks. The robustness of the aqueous crystallization process also means that production is less susceptible to minor fluctuations in environmental conditions or raw material quality compared to sensitive enzymatic processes. This stability ensures that manufacturers can maintain consistent delivery schedules, a critical factor for pharmaceutical clients who require Just-In-Time inventory management to support their own clinical or commercial manufacturing timelines without interruption.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this methodology aligns perfectly with modern green chemistry principles. The use of water as the reaction medium eliminates fire hazards associated with flammable solvents, simplifying facility safety requirements and insurance costs. The waste stream is predominantly aqueous with low organic load, making wastewater treatment more straightforward and compliant with increasingly stringent environmental regulations. The process has been demonstrated to scale effectively from laboratory benchtops to 500-liter pilot reactors without loss of performance, indicating a smooth path to multi-ton commercial production. This scalability ensures that the supply chain can expand rapidly to meet market demand surges without requiring prohibitive capital investment in new specialized infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this optical resolution technology. These insights are derived directly from the experimental data and comparative examples provided in the patent literature, offering clarity on why this method outperforms legacy techniques. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this intermediate into their existing synthetic workflows.
Q: Why is 2-methoxyphenylacetic acid (MPAA) superior to tartaric acid for 3AP resolution?
A: According to patent CN101910124A, traditional tartaric acid resolution suffers from unstable crystallization sequences where different stereoisomers precipitate unpredictably. MPAA forms a stable 1:2 diastereomeric salt that crystallizes consistently with high optical purity (>98%) and yield, avoiding the co-precipitation issues seen with tartrates.
Q: What is the specific role of inorganic acid (HCl) in this resolution process?
A: The addition of mineral acids like hydrochloric acid is critical for maximizing both yield and optical purity. The patent data indicates that while MPAA alone can resolve the amine, the presence of HCl significantly improves the resolution efficiency by optimizing the solubility profile of the diastereomeric salt in the aqueous medium, allowing for higher recovery rates without sacrificing enantiomeric excess.
Q: Is this aqueous resolution method suitable for large-scale manufacturing?
A: Yes, the process is specifically designed for industrial scalability. By utilizing water as the primary solvent instead of volatile organic compounds like ethanol or methanol, the method reduces fire hazards and solvent recovery costs. The crystallization parameters (cooling from 60°C to 20°C) are easily manageable in standard 500L+ reactors, ensuring robust supply chain continuity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Aminopyrrolidine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex pharmaceutical intermediates requires more than just a patent; it demands deep process engineering expertise and a commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale discovery to full-scale manufacturing is seamless. We adhere to stringent purity specifications and operate rigorous QC labs equipped with advanced chiral HPLC and NMR capabilities to guarantee that every batch of 3-aminopyrrolidine meets the exacting standards required for API synthesis. Our facility is designed to handle the specific thermal and mixing requirements of aqueous crystallization processes, ensuring consistent particle size distribution and bulk density for optimal downstream handling.
We invite potential partners to engage with our technical procurement team to discuss how this innovative resolution technology can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this aqueous-based supply chain. We encourage you to contact us today to obtain specific COA data for our current inventory and to receive comprehensive route feasibility assessments that demonstrate how our manufacturing capabilities can support your long-term supply goals with reliability and precision.
