Advanced Resolution Technology for Enantiopure Homocysteine-gamma-thiolactone: Scalable Manufacturing for Global Pharma Supply Chains
The pharmaceutical industry constantly seeks robust methods for producing enantiomerically pure intermediates, a critical requirement for ensuring drug safety and efficacy. Patent CN101622239A introduces a groundbreaking resolution method for preparing enantiomer-pure homocysteine-gamma-thiolactone (HCT), a vital building block in the synthesis of various bioactive compounds. This technology addresses the longstanding challenges associated with obtaining high optical purity without incurring prohibitive costs. The core innovation lies in a dynamic kinetic resolution strategy that combines classical diastereomeric salt formation with an in-situ racemization step. By utilizing chiral organic acids to selectively precipitate one isomer and catalytic amounts of aromatic aldehydes to recycle the other, this process fundamentally alters the economic landscape of HCT production. 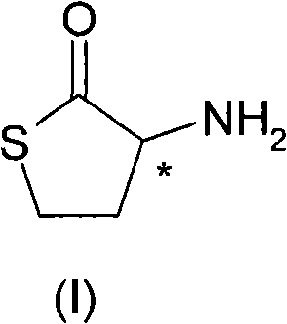 As depicted in the structure, the molecule contains a specific chiral carbon atom marked with an asterisk, necessitating precise stereochemical control during manufacturing to avoid the formation of inactive or harmful enantiomers.
As depicted in the structure, the molecule contains a specific chiral carbon atom marked with an asterisk, necessitating precise stereochemical control during manufacturing to avoid the formation of inactive or harmful enantiomers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of enantiomerically pure HCT has relied heavily on the demethylation of enantiomerically pure methionine, a pathway fraught with significant industrial disadvantages. This traditional route is not only costly due to the high price of the starting chiral amino acid but also presents severe difficulties when attempting to implement it on a large industrial scale. Alternative methods disclosed in prior art, such as those found in Japanese patents JP 2001-199980 and JP 2000-351776, involve reacting racemic HCT with chiral acids to form diastereomeric amides. However, these approaches suffer from low yields and require complex multi-step separation processes. Furthermore, the cleavage of the resulting amides to recover the free thiolactone is chemically difficult and often requires harsh conditions that can degrade the product. These inefficiencies create bottlenecks in the supply chain, leading to inconsistent availability and inflated costs for downstream pharmaceutical manufacturers who rely on this key intermediate for their synthesis pipelines.
The Novel Approach
The methodology outlined in CN101622239A offers a transformative solution by shifting the paradigm from linear synthesis to a cyclic resolution and recycling process. Instead of discarding the unwanted enantiomer, this novel approach recovers one isomer as a diastereomeric salt with a chiral organic acid while subjecting the remaining isomer in the mother liquor to racemization. This is achieved using catalytic amounts of aromatic aldehydes, such as salicylaldehyde, which facilitate the conversion of the unwanted enantiomer back into the racemic mixture. This dynamic equilibrium allows the process to theoretically convert nearly all starting material into the desired enantiomer, drastically improving overall atom economy. The method operates under relatively mild conditions, utilizing common solvents like acetone and methanol, and avoids the need for expensive transition metal catalysts or hazardous reagents. This simplicity translates directly into operational excellence, making it highly suitable for reliable agrochemical intermediate supplier networks and pharmaceutical supply chains alike.
Mechanistic Insights into Chiral Acid-Catalyzed Resolution and Racemization
The heart of this technology lies in the delicate interplay between solubility equilibria and catalytic racemization kinetics. When a racemic solution of HCT is treated with an enantiomerically pure chiral acid, such as di(toluoyl)tartaric acid or mandelic acid, two diastereomeric salts are formed. Due to their distinct physical properties, one salt is significantly less soluble and precipitates out of the solution upon cooling or concentration. This precipitation drives the equilibrium forward, effectively pulling the desired enantiomer out of the mixture. The patent details two main embodiments, with the second being particularly advantageous for industrial application. In this embodiment, the optical resolution is conducted in the presence of a catalytic amount of an aromatic aldehyde. This additive ensures that the more soluble diastereomeric salt, which remains in the mother liquor, undergoes continuous racemization. 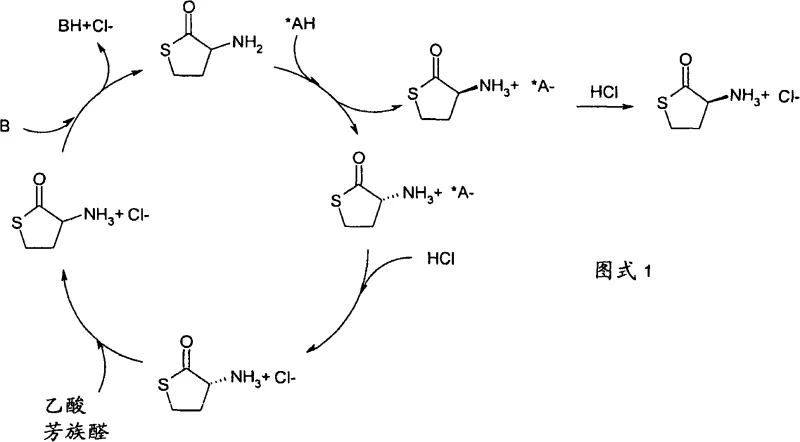 As illustrated in the reaction scheme, the base (B) and chiral acid (*AH) facilitate the salt formation, while the subsequent treatment with HCl and aromatic aldehyde regenerates the racemic starting material from the mother liquor. This creates a closed loop where the 'waste' stream is continuously fed back into the process, maximizing resource utilization.
As illustrated in the reaction scheme, the base (B) and chiral acid (*AH) facilitate the salt formation, while the subsequent treatment with HCl and aromatic aldehyde regenerates the racemic starting material from the mother liquor. This creates a closed loop where the 'waste' stream is continuously fed back into the process, maximizing resource utilization.
From an impurity control perspective, this mechanism offers superior purity profiles compared to traditional methods. Because the unwanted enantiomer is constantly being converted back to the racemate rather than accumulating as a byproduct, the risk of cross-contamination in the final crystalline product is minimized. The use of specific aromatic aldehydes like salicylaldehyde is critical, as they form transient intermediates with the amine group of the HCT, lowering the energy barrier for racemization without causing degradation of the sensitive thiolactone ring. The process continues until an equilibrium is reached between the solubility product (Ksp) of the precipitating salt and the kinetics of the racemization reaction. This thermodynamic control ensures that the final product, typically isolated as the hydrochloride salt, meets stringent purity specifications required for high-purity OLED material or pharmaceutical intermediate applications. The ability to tune this balance by adjusting temperature and solvent composition provides process chemists with robust control over the final quality attributes.
How to Synthesize Homocysteine-gamma-thiolactone Efficiently
The synthesis protocol derived from this patent provides a clear roadmap for manufacturing teams aiming to implement this technology. The process begins with the preparation of a racemic HCT solution, typically generated by treating commercially available racemic HCT hydrochloride with a base like triethylamine in a solvent such as acetone. To this solution, a stoichiometric amount of the chosen chiral resolving agent is added. For instance, when using mandelic acid, the mixture is stirred until the less soluble diastereomeric salt precipitates completely. The solid is then filtered and washed, while the mother liquor is retained for the crucial recycling step. This streamlined approach eliminates the need for complex chromatographic separations, relying instead on the fundamental principles of crystallization and solubility differences to achieve high optical purity.
- Prepare a solution of racemic HCT in a suitable solvent such as acetone or methanol and add an enantiomerically pure chiral organic acid like mandelic acid or di(toluoyl)tartaric acid.
- Induce precipitation of the less soluble diastereomeric salt by cooling or heating, then separate the solid via filtration to isolate the desired enantiomer precursor.
- Treat the mother liquor containing the unwanted isomer with a catalytic amount of aromatic aldehyde and acid to racemize it, allowing for recycling and further resolution cycles.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this resolution technology represents a significant opportunity for cost reduction in pharmaceutical intermediates manufacturing. The most compelling economic driver is the drastic improvement in yield. Traditional resolution methods are capped at a maximum theoretical yield of 50% for the desired enantiomer unless a racemization step is included. However, this patent demonstrates that by integrating in-situ racemization, yields of enantiomerically pure HCT hydrochloride can exceed 140% based on the theoretical amount of the desired enantiomer in the initial racemate. This means that for every ton of raw material purchased, the output of valuable product is nearly tripled compared to static resolution methods. Such efficiency gains directly translate to lower unit costs, allowing companies to maintain competitive pricing even in volatile raw material markets. Furthermore, the reagents used—such as mandelic acid, tartaric acid derivatives, and salicylaldehyde—are commodity chemicals with stable global supply chains, reducing the risk of procurement bottlenecks.
- Cost Reduction in Manufacturing: The elimination of expensive starting materials like enantiopure methionine is a primary cost-saving factor. By starting with cheap racemic HCT and recycling the mother liquor, the process minimizes raw material waste. The removal of transition metal catalysts, which often require costly removal steps to meet regulatory limits, further simplifies the downstream processing. This reduction in processing steps leads to substantial cost savings in terms of labor, energy, and solvent consumption. Additionally, the ability to recover and reuse the chiral resolving acid from the mother liquor streams adds another layer of economic efficiency, ensuring that high-value chiral auxiliaries are not lost to waste streams.
- Enhanced Supply Chain Reliability: Relying on a synthesis route that depends on the demethylation of methionine exposes the supply chain to fluctuations in the amino acid market. In contrast, this resolution method utilizes widely available bulk chemicals, insulating the production process from upstream supply shocks. The robustness of the crystallization-based purification also means that the process is less sensitive to minor variations in reaction conditions, leading to higher batch-to-batch consistency. This reliability is crucial for reducing lead time for high-purity pharmaceutical intermediates, ensuring that downstream drug manufacturers receive their materials on schedule without delays caused by failed batches or complex purification struggles.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from laboratory benchtop to commercial scale-up of complex pharmaceutical intermediates. The use of standard solvents like acetone and methanol facilitates easy solvent recovery and recycling, aligning with modern green chemistry principles. By converting the unwanted enantiomer back into the process rather than discarding it, the overall waste generation is significantly reduced, lowering the environmental footprint and disposal costs. This compliance with environmental regulations is increasingly important for maintaining operating licenses and meeting the sustainability goals of multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this resolution technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for R&D teams evaluating the feasibility of adopting this route for their specific production needs. By addressing these points early, organizations can better assess the fit of this technology within their existing manufacturing infrastructure.
Q: What is the primary advantage of the dynamic resolution method described in CN101622239A?
A: The primary advantage is the ability to exceed the theoretical 50% yield limit of traditional resolution. By continuously racemizing the unwanted enantiomer in the mother liquor using catalytic aromatic aldehydes, the process converts the undesired isomer back into the racemic mixture, allowing for yields often exceeding 140% based on the initial desired enantiomer content.
Q: Which chiral resolving agents are most effective for this process?
A: The patent identifies di(toluoyl)tartaric acid and mandelic acid as the most preferred resolving agents. These acids form diastereomeric salts with distinct solubility profiles in solvents like acetone and methanol, facilitating efficient separation of the target enantiomer through crystallization.
Q: How does this method improve supply chain sustainability compared to methionine demethylation?
A: Unlike the conventional demethylation of enantiopure methionine, which relies on expensive starting materials and difficult industrial scaling, this resolution method utilizes readily available racemic HCT. The recycling of mother liquors significantly reduces waste generation and raw material consumption, leading to a more sustainable and cost-effective supply chain.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Homocysteine-gamma-thiolactone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug development programs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in the clinical trial phase or full-scale commercialization. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our expertise in chiral resolution and dynamic kinetic processes allows us to optimize the synthesis of homocysteine-gamma-thiolactone, guaranteeing consistent optical purity and minimal impurity profiles that satisfy global regulatory standards.
We invite you to collaborate with us to leverage these advanced manufacturing capabilities for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized processes can enhance your supply chain efficiency. Let us be your partner in navigating the complexities of fine chemical manufacturing, ensuring a secure and cost-effective supply of this vital intermediate.
