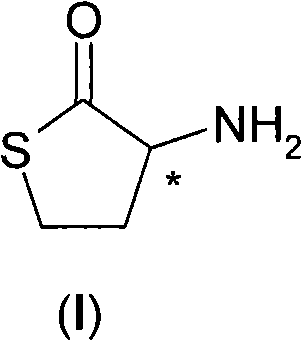
Scalable Production of Enantiopure Homocysteine-Gamma-Thiolactone via Dynamic Resolution
The pharmaceutical industry continuously demands high-purity chiral building blocks to ensure the safety and efficacy of active pharmaceutical ingredients (APIs). A critical intermediate in this domain is homocysteine-gamma-thiolactone (HCT), a compound characterized by its reactive thiolactone ring and chiral center. As detailed in patent CN101622239B, a groundbreaking methodology has been established for the preparation of enantiomerically pure HCT, overcoming the significant limitations of traditional synthesis routes. This innovation leverages a sophisticated combination of optical resolution using chiral organic acids and catalytic racemization mediated by aromatic aldehydes. By transforming the unwanted enantiomer back into the racemic mixture, this process打破了 the theoretical yield barrier of standard resolution techniques. For global procurement teams and R&D directors, this represents a pivotal shift towards more sustainable and cost-effective manufacturing of sulfur-containing amino acid derivatives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of enantiomerically pure homocysteine derivatives has been fraught with economic and technical challenges. The first known method involves the demethylation of enantiomerically pure methionine, a process that is inherently costly due to the high price of the starting chiral material. Furthermore, the subsequent closure of the lactone ring requires harsh conditions that can compromise product integrity and lead to significant impurity profiles. Alternative approaches, such as those disclosed in Japanese patents JP 2001-199980 and JP 2000-351776, rely on forming diastereomeric amides. While chemically feasible, these methods suffer from low overall yields and require difficult cleavage reactions to liberate the free amine. The multi-step nature of these conventional pathways introduces multiple points of failure, increases solvent consumption, and generates substantial chemical waste, rendering them suboptimal for modern, green chemistry-compliant industrial operations.
The Novel Approach
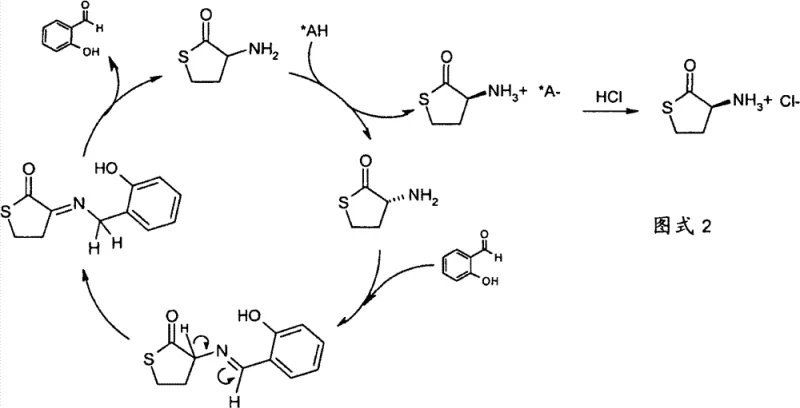
The methodology described in CN101622239B introduces a paradigm shift by integrating resolution with dynamic racemization. Instead of discarding the unwanted enantiomer, which typically constitutes 50% of the starting racemic material, this novel approach recycles it. The process begins with the formation of a diastereomeric salt between the racemic HCT and a chiral organic acid, such as mandelic acid or di(toluoyl)tartaric acid. The key innovation lies in the treatment of the mother liquor—the solution containing the soluble, unwanted isomer—with a catalytic amount of an aromatic aldehyde. This triggers a rapid racemization reaction, effectively converting the waste isomer back into a racemic mixture that can be subjected to resolution again. In the most advantageous embodiment, this racemization occurs simultaneously with the resolution, creating a dynamic system where the desired isomer is continuously pulled out of equilibrium as a precipitate, driving the reaction towards near-quantitative conversion of the starting material into the single desired enantiomer.
Mechanistic Insights into Chiral Acid Resolution and Aldehyde-Catalyzed Racemization
The core of this technology relies on the precise interplay between thermodynamics and kinetics within the reaction vessel. When a chiral resolving agent like L-(+)-mandelic acid is introduced to the racemic HCT base, two diastereomeric salts are formed. Due to differences in crystal lattice energy and solvation, one salt (e.g., the S-HCT mandelate) exhibits significantly lower solubility in solvents like acetone and precipitates out of the solution. This precipitation is the driving force for the separation. However, without intervention, the process would stop at 50% yield. The introduction of an aromatic aldehyde, preferably salicylaldehyde, changes the game entirely. The aldehyde reacts with the free amine group of the HCT remaining in the mother liquor to form a Schiff base intermediate. This imine formation renders the alpha-proton adjacent to the chiral center acidic, allowing for easy deprotonation and reprotonation, which results in the loss of stereochemical information (racemization). Once the Schiff base is hydrolyzed back to the amine, the previously unwanted enantiomer has been converted into a 50:50 mixture, ready to re-enter the resolution cycle.
From an impurity control perspective, this mechanism is exceptionally robust. Because the racemization is catalytic and occurs in the solution phase, it minimizes the formation of side products often associated with stoichiometric derivatization methods. The use of mild conditions, typically heating to moderate temperatures (e.g., 50°C) in acetic acid or simply stirring at room temperature in the presence of the aldehyde, ensures that the sensitive thiolactone ring remains intact. The patent data highlights that yields of enantiomerically pure HCT hydrochloride can exceed 140% based on the theoretical amount of the desired enantiomer in the starting racemate. This figure is a direct testament to the efficiency of the recycling mechanism, proving that the process successfully harvests material that would otherwise be lost. For R&D teams, this implies a cleaner reaction profile with fewer byproducts to manage during downstream purification.
How to Synthesize Homocysteine-Gamma-Thiolactone Efficiently
Implementing this resolution strategy requires careful control of solvent systems and stoichiometry to maximize the precipitation of the desired diastereomer while maintaining the solubility of the racemizing species. The process generally initiates by neutralizing commercially available racemic HCT hydrochloride with a tertiary amine base, such as triethylamine, in a polar aprotic or alcoholic solvent. Following the addition of the chiral acid, the system is allowed to equilibrate, often with cooling to enhance crystal growth. The solid salt is filtered, and the filtrate is not discarded but instead treated with the racemization catalyst. This cyclic nature of the synthesis allows for a continuous or semi-continuous operation mode, which is highly desirable for process chemistry. The following guide outlines the standardized operational parameters derived from the patent examples to ensure reproducibility and high optical purity.
- Prepare a solution of racemic HCT hydrochloride in a suitable solvent like acetone or methanol and neutralize with a base such as triethylamine.
- Add an enantiomerically pure chiral organic acid, such as mandelic acid or di(toluoyl)tartaric acid, to precipitate the less soluble diastereomeric salt.
- Treat the mother liquor containing the unwanted isomer with a catalytic amount of an aromatic aldehyde (e.g., salicylaldehyde) to induce racemization for recycling.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this resolution-based methodology offers profound strategic benefits compared to legacy synthesis routes. The primary advantage is the drastic reduction in raw material costs. By utilizing cheap, readily available racemic HCT hydrochloride as the starting feedstock instead of expensive enantiopure methionine, the cost of goods sold (COGS) is significantly lowered. Furthermore, the ability to recycle the mother liquor means that the effective utilization of the starting material approaches 100%, eliminating the waste associated with the 50% loss typical of classical resolutions. This efficiency translates directly into margin improvement and reduced exposure to volatile pricing of chiral pool starting materials. The supply chain becomes more resilient as it relies on commodity chemicals rather than specialized, low-volume chiral precursors.
- Cost Reduction in Manufacturing: The elimination of expensive chiral starting materials like enantiopure methionine removes a major cost driver from the production budget. Additionally, the catalytic nature of the racemization step means that only small amounts of aromatic aldehydes are required, further reducing reagent costs. The process avoids the need for complex amide formation and hydrolysis steps, which typically require additional reagents, energy, and time, thereby streamlining the overall manufacturing expense structure substantially.
- Enhanced Supply Chain Reliability: Relying on racemic starting materials simplifies sourcing logistics, as these compounds are produced on a much larger industrial scale than their chiral counterparts. The robustness of the resolution process, which tolerates standard industrial solvents like acetone and methanol, ensures that production is not bottlenecked by the availability of exotic reagents. This stability allows for consistent lead times and reliable inventory planning, crucial for maintaining uninterrupted API production schedules for downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work efficiently with simple filtration and crystallization unit operations that are standard in any multipurpose chemical plant. The reduction in waste generation, achieved by recycling the unwanted isomer rather than discarding it, aligns perfectly with modern environmental regulations and sustainability goals. This reduces the burden on waste treatment facilities and lowers the environmental compliance costs associated with hazardous waste disposal, making the facility more attractive for audits and regulatory inspections.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this resolution technology. They are derived directly from the experimental data and claims within the patent documentation to provide accurate guidance for potential partners. Understanding these nuances is essential for evaluating the feasibility of integrating this route into your existing manufacturing portfolio.
Q: What is the primary advantage of using aromatic aldehydes in this resolution process?
A: The use of catalytic aromatic aldehydes, such as salicylaldehyde, enables the in situ racemization of the unwanted enantiomer remaining in the mother liquor. This converts the waste stream back into the racemic mixture, allowing it to re-enter the resolution cycle and significantly boosting the overall yield beyond the theoretical 50% limit of standard resolution.
Q: Which chiral resolving agents are most effective for HCT separation?
A: The patent data identifies di(toluoyl)tartaric acid and mandelic acid as the preferred resolving agents. These acids form diastereomeric salts with distinct solubility profiles in solvents like acetone and methanol, facilitating efficient precipitation and isolation of the desired enantiomer with high optical purity.
Q: Can this process be scaled for industrial manufacturing?
A: Yes, the process is designed for scalability. It utilizes commercially available racemic starting materials and common organic solvents. The ability to recycle the mother liquor through catalytic racemization reduces raw material consumption, making it economically viable for large-scale production of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Homocysteine-Gamma-Thiolactone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the drug development pipeline. Our technical team has extensively analyzed the resolution process described in CN101622239B and possesses the expertise to scale this pathway from laboratory benchtop to full commercial production. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee the enantiomeric excess required for your specific application. We are committed to delivering a product that meets the highest international standards for pharmaceutical intermediates.
We invite you to discuss how our optimized manufacturing capabilities can support your project goals. By leveraging our expertise in chiral resolution and process optimization, we can help you achieve significant efficiencies in your supply chain. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our production of homocysteine-gamma-thiolactone can add value to your operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
