Scalable Manufacturing of Indolinobenzodiazepine Derivatives via Novel Iron-Mediated Cyclization
The pharmaceutical industry's relentless pursuit of potent Antibody-Drug Conjugates (ADCs) has placed indolinobenzodiazepine derivatives at the forefront of cytotoxic payload development. Patent CN110573510B, filed in 2022, introduces a transformative methodology for synthesizing these complex heterocyclic monomers and their synthetic precursors. Unlike traditional routes that often rely on harsh conditions or expensive noble metal catalysts for every transformation, this invention leverages a strategic combination of iron-mediated reductive cyclization and transfer hydrogenation. This technical breakthrough addresses critical bottlenecks in the commercial production of high-purity pharmaceutical intermediates, specifically targeting the needs of process chemists who require robust, scalable, and safe manufacturing protocols. The disclosed methods facilitate the construction of the benzodiazepine core under mild conditions, significantly mitigating the risks associated with high-pressure hydrogenation while ensuring excellent control over stereochemistry and impurity profiles.
For procurement managers and supply chain directors, the implications of this technology are profound. By shifting the reliance from exclusive dependence on high-pressure hydrogenation infrastructure to more accessible chemical reduction methods, the barrier to entry for manufacturing these high-value intermediates is substantially lowered. The process utilizes commodity chemicals such as iron powder and ammonium chloride, which are globally available and cost-stable, thereby insulating the supply chain from the volatility often seen with specialized catalytic reagents. Furthermore, the emphasis on crystallization-based purification rather than chromatographic separation aligns perfectly with the requirements for multi-kilogram to ton-scale production, ensuring that the cost of goods sold (COGS) remains competitive even for complex molecular architectures.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of indolinobenzodiazepine scaffolds has been plagued by significant process safety hazards and economic inefficiencies. Conventional routes frequently necessitate the use of high-pressure hydrogen gas (H2) in conjunction with palladium or platinum catalysts to effect the reduction of nitro groups and subsequent cyclization. This requirement mandates the use of specialized autoclaves and rigorous safety protocols, which drastically increases capital expenditure (CAPEX) and limits the number of qualified Contract Development and Manufacturing Organizations (CDMOs) capable of executing the chemistry. Additionally, traditional methods often struggle with chemoselectivity, where the reduction conditions might inadvertently affect other sensitive functional groups present in the molecule, leading to complex impurity profiles that are difficult to purge. The reliance on chromatographic purification for intermediates further exacerbates cost issues, as silica gel consumption and solvent usage become prohibitive at commercial scales, rendering many promising candidates economically unviable for widespread therapeutic application.
The Novel Approach
The methodology outlined in CN110573510B represents a paradigm shift by introducing a mild, iron-mediated reductive cyclization strategy. This novel approach utilizes elemental iron and ammonium chloride in a mixed solvent system to simultaneously reduce the nitro functionality and induce cyclization to form the seven-membered diazepine ring. This transformation proceeds efficiently at moderate temperatures, typically around 65°C, eliminating the need for high-pressure equipment. Moreover, the patent details a sophisticated debenzylation protocol using transfer hydrogenation with 1,4-cyclohexadiene as a hydrogen donor in the presence of palladium on alumina. This technique allows for the removal of benzyl protecting groups without handling gaseous hydrogen, thereby enhancing operational safety. The integration of these steps creates a streamlined workflow that prioritizes safety and scalability, making it an ideal candidate for reliable pharmaceutical intermediate supplier networks aiming to deliver consistent quality.
Mechanistic Insights into Iron-Mediated Reductive Cyclization
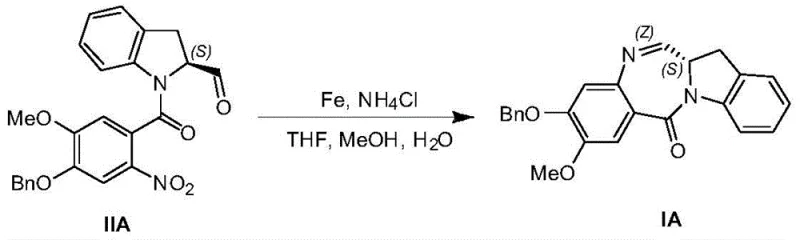
The core innovation of this patent lies in the mechanistic elegance of the iron-mediated transformation. The reaction begins with the single-electron transfer from the surface of the iron powder to the nitro group of the benzamide precursor, generating a nitro radical anion. In the presence of ammonium chloride, which acts as a proton source, this species undergoes a series of reductions to form the corresponding hydroxylamine and eventually the amine. Crucially, the proximity of the newly formed amine to the electrophilic carbonyl of the amide linkage facilitates an intramolecular nucleophilic attack. This cyclization event is thermodynamically driven by the formation of the stable aromatic system within the indoline moiety and the relief of ring strain. The use of a THF, methanol, and water solvent mixture is critical; it ensures the solubility of the organic substrate while maintaining the necessary ionic environment for the iron surface to remain active. This delicate balance prevents the passivation of the iron surface, ensuring high conversion rates and minimizing the formation of azo or hydrazo byproducts that are common in poorly optimized nitro reductions.
Impurity control is meticulously managed through the physical properties of the intermediates. The patent specifies that the crude product from the cyclization step can be purified via precipitation or recrystallization using specific solvent pairs like dichloromethane and ethanol or toluene and acetonitrile. This selectivity is rooted in the distinct solubility differences between the planar, conjugated benzodiazepine product and the non-planar, polar iron salt byproducts. By optimizing the temperature and solvent ratios, the process forces the desired product out of the solution while keeping impurities dissolved. For instance, dissolving the crude material in hot toluene and adding acetonitrile as an anti-solvent induces the formation of a highly ordered crystalline lattice. This not only removes residual iron salts but also enriches the enantiomeric excess if chiral starting materials are used, as the crystal packing often favors one stereoisomer. Such robust purification protocols are essential for meeting the stringent purity specifications required for clinical-grade active pharmaceutical ingredients.
How to Synthesize Indolinobenzodiazepine Derivatives Efficiently
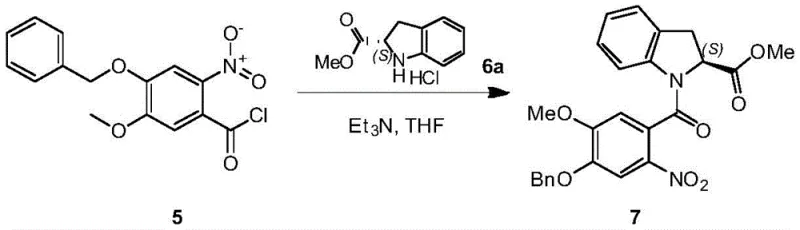
The synthesis of these complex molecules requires precise control over reaction parameters to ensure reproducibility and high yield. The process generally initiates with the coupling of a functionalized benzoic acid derivative with a chiral indoline amine to establish the requisite amide bond. Following this assembly, the key cyclization step is performed under inert atmosphere to prevent oxidation of the sensitive intermediates. The reaction mixture is typically heated to promote the cyclization kinetics, followed by a workup that involves filtration to remove bulk iron solids. The filtrate is then concentrated, and the residue is subjected to the aforementioned crystallization protocols. For the final deprotection, the benzyl group is removed using the transfer hydrogenation method, which requires careful monitoring of the hydrogen donor addition to prevent over-reduction or side reactions. Detailed standardized synthesis steps see the guide below.
- Preparation of the nitro-benzamide precursor via coupling of a substituted benzoic acid chloride with an indoline derivative.
- Execution of the key reductive cyclization using iron powder and ammonium chloride in a THF/MeOH/Water solvent system to form the benzodiazepine core.
- Final deprotection of the benzyl group using palladium-catalyzed transfer hydrogenation or acid-mediated cleavage to yield the target phenol derivative.
Commercial Advantages for Procurement and Supply Chain Teams
The adoption of the processes described in CN110573510B offers tangible benefits for the commercial viability of indolinobenzodiazepine manufacturing. From a procurement perspective, the substitution of high-pressure hydrogenation with iron-mediated reduction significantly reduces the dependency on specialized infrastructure. This broadens the pool of potential manufacturing partners, fostering a more competitive sourcing environment. The raw materials involved, particularly iron powder and ammonium chloride, are commodity chemicals with stable pricing and abundant global supply, mitigating the risk of raw material shortages that can plague projects relying on exotic catalysts. Furthermore, the elimination of high-pressure gas handling simplifies the regulatory compliance landscape, as facilities do not need to maintain the same level of certification for pressure vessels, thereby accelerating the timeline from process validation to commercial production.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the drastic simplification of the reaction setup and purification workflow. By avoiding high-pressure autoclaves, manufacturers save on both equipment depreciation and maintenance costs. The use of iron as a stoichiometric reductant is exponentially cheaper than using catalytic hydrogenation with precious metals, especially when considering the turnover numbers required for large batches. Additionally, the shift towards crystallization-based purification eliminates the massive solvent and silica gel costs associated with flash chromatography. This reduction in consumable usage directly lowers the variable cost per kilogram, allowing for substantial cost savings in the overall manufacturing budget without compromising the quality of the final API intermediate.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available reagents and standard laboratory equipment. The process does not rely on single-source specialty catalysts that might face supply disruptions. The robustness of the iron reduction step means that minor fluctuations in reagent quality can be tolerated without catastrophic failure of the reaction, ensuring consistent batch-to-batch performance. Moreover, the ability to perform the debenzylation using transfer hydrogenation means that production can continue even in facilities lacking hydrogen gas infrastructure, providing greater flexibility in choosing manufacturing sites and reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scaling this process from gram to ton quantities is straightforward due to the exothermic nature of the iron reduction being manageable in standard jacketed reactors. The waste profile is also improved; iron oxides generated during the reaction are non-toxic and easily disposed of compared to heavy metal waste from other reduction methods. The solvent systems recommended, such as ethyl acetate and ethanol, are greener alternatives to chlorinated solvents often used in older methodologies. This alignment with green chemistry principles simplifies environmental permitting and waste treatment, ensuring long-term sustainability and compliance with increasingly strict global environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this synthesis route. They are derived from the specific embodiments and experimental data provided in the patent documentation, focusing on practical aspects of reaction execution and purification. Understanding these details is crucial for process engineers evaluating the feasibility of technology transfer.
Q: What is the primary advantage of the iron-mediated cyclization method described in CN110573510B?
A: The primary advantage is the replacement of expensive and potentially hazardous catalytic hydrogenation conditions with a mild, cost-effective iron powder reduction. This method operates at atmospheric pressure and moderate temperatures (e.g., 65°C), significantly reducing equipment costs and safety risks associated with high-pressure hydrogen gas while maintaining high chemoselectivity for the nitro group reduction and subsequent cyclization.
Q: How does the patent address the removal of benzyl protecting groups safely?
A: The patent discloses a transfer hydrogenation protocol using palladium on alumina (Pd/Alox) or palladium on carbon (Pd/C) with 1,4-cyclohexadiene as the hydrogen donor. This approach eliminates the need for flammable hydrogen gas cylinders, enhancing operational safety in large-scale manufacturing environments while ensuring efficient debenzylation under mild thermal conditions.
Q: What purification strategies are recommended for the intermediate benzodiazepine compounds?
A: The documentation emphasizes recrystallization and precipitation techniques over chromatography to ensure scalability. Specific solvent systems such as dichloromethane/ethanol or toluene/acetonitrile mixtures are utilized to precipitate the product with high purity, effectively removing iron salts and organic impurities without the need for resource-intensive column chromatography.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indolinobenzodiazepine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality linkers and payloads play in the success of next-generation ADC therapies. Our technical team has extensively analyzed the methodology presented in CN110573510B and possesses the expertise to implement these advanced synthetic routes effectively. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from clinical trials to market launch. Our state-of-the-art facilities are equipped to handle the specific requirements of iron-mediated chemistries and transfer hydrogenation, supported by stringent purity specifications and rigorous QC labs that guarantee every batch meets the highest international standards.
We invite you to collaborate with us to optimize your supply chain for these vital pharmaceutical intermediates. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our implementation of this patent technology can reduce your overall COGS. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us partner with you to accelerate the development of life-saving medicines through superior chemical manufacturing excellence.
