Advanced Catalytic Hydrogenation Strategy for Commercial Scale Ertapenem Sodium Production
Advanced Catalytic Hydrogenation Strategy for Commercial Scale Ertapenem Sodium Production
The pharmaceutical industry continuously seeks robust manufacturing pathways for critical antibiotics, particularly carbapenems, which are vital for treating resistant bacterial infections. A significant technological breakthrough in this domain is detailed in patent CN114105988B, which outlines a novel synthetic method for Ertapenem Sodium. This innovation addresses long-standing challenges associated with the instability of carbapenem intermediates and the stringent regulatory limits on heavy metal residues. By shifting the paradigm from unstable single-protection intermediates to a more robust Formula 1 precursor, this method achieves a remarkable purity of over 99% and reduces heavy metal content to less than 10 ppm. For global supply chain leaders and R&D directors, this represents a pivotal shift towards more reliable and compliant API intermediate manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Ertapenem has relied heavily on palladium-carbon hydrogenation followed by deprotection steps that are fraught with technical difficulties. The conventional route typically utilizes an intermediate with a single protection structure, which is inherently unstable under hydrogenation conditions. This instability leads to severe product degradation, resulting in a complex mixture of impurities that are difficult to separate. Furthermore, the post-treatment operations in these traditional methods are excessively complicated, often requiring multiple extraction and purification stages that still fail to adequately remove palladium residues. Consequently, manufacturers frequently face yields as low as 43.3% and purity levels hovering around 96.1%, which are suboptimal for modern pharmaceutical standards and create significant waste disposal burdens.
The Novel Approach
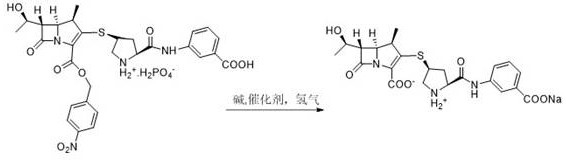
In stark contrast, the methodology disclosed in the patent introduces a highly efficient route centered on the hydrogenation of a specific compound, designated as Formula 1. This intermediate possesses superior stability compared to its predecessors, allowing the hydrogenation reaction to proceed thoroughly without significant degradation. The process employs a sophisticated mixed solvent system comprising water, methanol, n-propanol, and ethyl acetate, which facilitates a homogeneous reaction phase. This homogeneity ensures better mass transfer and reaction control, drastically reducing the formation of dimers and other oligomeric impurities. The result is a streamlined workflow where the product solution can be directly filtered and crystallized, bypassing the need for column chromatography and achieving a molar yield of 82.1% with exceptional purity.
Mechanistic Insights into Catalytic Hydrogenation and Solvent Engineering
The core of this technological advancement lies in the precise engineering of the reaction environment and the selection of the starting material. The hydrogenation step is conducted under a hydrogen pressure of 0.5 to 3 MPa, typically optimized between 1.0 and 2.0 MPa, using a catalyst such as palladium on carbon (Pd/C) with a specific loading of 5-50% by mass. The choice of the mixed solvent system is critical; the presence of methanol and n-propanol not only aids in dissolving the Formula 1 compound to form a homogeneous phase but also modulates the reaction kinetics to favor the desired deprotection pathway. This solvent engineering prevents the localized concentration spikes that often lead to side reactions in heterogeneous systems. Moreover, the inclusion of ethyl acetate in the mixture plays a dual role: it participates in the solvation of the reactants and later facilitates the liquid-liquid separation process, allowing for the effective removal of organic impurities before the final crystallization step.
Impurity control is further enhanced by the inherent stability of the Formula 1 intermediate. In conventional routes, the fragile nature of the intermediate often leads to ring-opening or beta-lactam degradation during the exothermic hydrogenation process. However, the structural integrity of Formula 1 withstands these conditions, ensuring that the primary reaction pathway dominates. Following the reaction, the pH of the aqueous phase is carefully adjusted to a range of 4 to 7, preferably between 5.5 and 6.0. This precise pH control is essential for converting the product into its acidic form suitable for crystallization while preventing acid-catalyzed degradation of the sensitive beta-lactam ring. The final crystallization from a methanol and n-propanol mixture yields a product with a crystal lattice that effectively excludes residual solvents and trace metals, guaranteeing the high quality required for parenteral administration.
How to Synthesize Ertapenem Sodium Efficiently
The synthesis of Ertapenem Sodium via this patented route involves a two-stage process that prioritizes yield and purity. First, the stable intermediate (Formula 1) is prepared by condensing the carbapenem parent nucleus MAP with the Ertapenem side chain hydrochloride in a polar aprotic solvent like DMF, using a strong organic base such as tetramethylguanidine at low temperatures. Once this stable intermediate is isolated, it undergoes the critical hydrogenation step. The detailed operational parameters, including specific solvent ratios and catalyst loadings, are crucial for replicating the high success rates reported in the patent data. For a comprehensive guide on executing this synthesis with industrial precision, please refer to the standardized protocol below.
- Condense Carbapenem parent nucleus MAP with Ertapenem side chain hydrochloride in DMF using tetramethylguanidine to form the stable Formula 1 intermediate.
- Perform catalytic hydrogenation on Formula 1 in a mixed solvent system (water/methanol/n-propanol/ethyl acetate) using Pd/C catalyst under controlled pressure.
- Filter the reaction mixture, adjust pH to 4-7, and crystallize directly from a methanol/n-propanol solvent system to obtain pure Ertapenem Sodium.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis method offers transformative benefits that extend beyond mere technical metrics. The elimination of complex purification steps, such as column chromatography, translates directly into significant cost reduction in pharmaceutical intermediate manufacturing. By simplifying the workflow to filtration, extraction, and crystallization, the process reduces solvent consumption, labor hours, and equipment occupancy time. This efficiency gain allows for a more competitive pricing structure without compromising on the quality of the final API. Furthermore, the drastic improvement in yield—from roughly 43% in older methods to over 82% in this new process—means that less raw material is required to produce the same amount of finished goods, substantially lowering the cost of goods sold (COGS) and enhancing overall margin potential.
- Cost Reduction in Manufacturing: The streamlined nature of this process removes the need for expensive silica gel columns and the large volumes of solvents associated with chromatographic purification. Additionally, the high yield implies that the consumption of high-value starting materials like the carbapenem parent nucleus is nearly halved per unit of output. This efficiency creates a leaner manufacturing model where resource utilization is maximized, leading to substantial cost savings that can be passed down the supply chain or reinvested into further R&D initiatives.
- Enhanced Supply Chain Reliability: The stability of the Formula 1 intermediate is a game-changer for inventory management and logistics. Unlike previous intermediates that required immediate processing or specialized cold-chain storage to prevent degradation, this robust precursor can be stockpiled and transported with greater ease. This flexibility reduces the risk of production stoppages due to material spoilage and allows for more agile response times to market demand fluctuations. The simplified post-treatment also shortens the overall production cycle time, enabling faster turnaround from raw material intake to finished product release.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this method is exceptionally clean. The reduction in heavy metal residues to below 10 ppm ensures compliance with strict international pharmacopoeia standards without the need for additional metal scavenging steps, which often generate hazardous waste. The use of common, recoverable solvents like ethyl acetate and alcohols simplifies waste treatment and solvent recovery systems. This alignment with green chemistry principles not only mitigates environmental risk but also future-proofs the manufacturing site against increasingly stringent environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method outperforms legacy processes in terms of purity, yield, and operational simplicity.
Q: How does this new synthesis method improve heavy metal control compared to traditional routes?
A: Traditional methods often struggle with palladium residue exceeding standards due to complex post-treatment. This novel approach utilizes a specific mixed solvent system and direct filtration after hydrogenation, effectively reducing heavy metal residues to below 10 ppm without requiring expensive scavenging resins or column chromatography.
Q: What is the stability advantage of the Formula 1 intermediate?
A: Unlike the 'single protection structure' intermediates used in conventional methods which degrade easily during hydrogenation, the Formula 1 intermediate described in Patent CN114105988B exhibits superior chemical stability. This stability minimizes degradation byproducts, directly contributing to the achieved yield increase from roughly 43% to over 82%.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process is designed for scalability. It eliminates the need for column chromatography and uses common industrial solvents like ethyl acetate, methanol, and n-propanol. The simplified work-up involving liquid separation and direct crystallization makes it highly viable for multi-kilogram to ton-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ertapenem Sodium Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes to maintain competitiveness in the global antibiotic market. Our team of expert chemists has extensively evaluated the pathway described in Patent CN114105988B and confirmed its viability for large-scale production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to industrial manufacturing is seamless. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the detection of trace heavy metals and specific impurities, guaranteeing that every batch of Ertapenem Sodium meets the highest international standards.
We invite pharmaceutical partners to collaborate with us to optimize their supply chains using this superior technology. By leveraging our expertise, you can secure a stable source of high-quality intermediates that drive down your overall production costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term strategic goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
