Advanced Manufacturing of Ertapenem Sodium: Overcoming Purity Barriers in Carbapenem Synthesis
The pharmaceutical landscape for broad-spectrum antibiotics continues to evolve, with carbapenems remaining a cornerstone of modern antimicrobial therapy. Specifically, the preparation of ertapenem sodium, a potent agent developed through collaborative efforts by major pharmaceutical entities, presents unique synthetic challenges that directly impact global supply chains. The patent CN102690266B introduces a transformative methodology that addresses the longstanding issues of purity and yield in ertapenem manufacturing. By redefining the handling of the critical intermediate, this technology offers a pathway to high-quality active pharmaceutical ingredients (APIs) that is both robust and scalable. For R&D directors and procurement specialists alike, understanding the nuances of this process is essential for securing a reliable ertapenem sodium supplier capable of meeting stringent regulatory standards.
Traditionally, the synthesis of carbapenem antibiotics has been fraught with complexity, often requiring multi-step protection and deprotection strategies that inflate costs and extend lead times. Conventional routes frequently employed three-protection or two-protection structures involving allyl or nitrobenzyl groups, which necessitated harsh conditions for removal. Furthermore, earlier attempts at streamlining the process via one-pot reactions often resulted in the carryover of inorganic salts and phosphonic acid byproducts into the final hydrogenation stage. These impurities not only complicated the downstream purification processes but also posed significant risks to the final product's purity profile, often necessitating additional extraction steps that reduced overall efficiency.
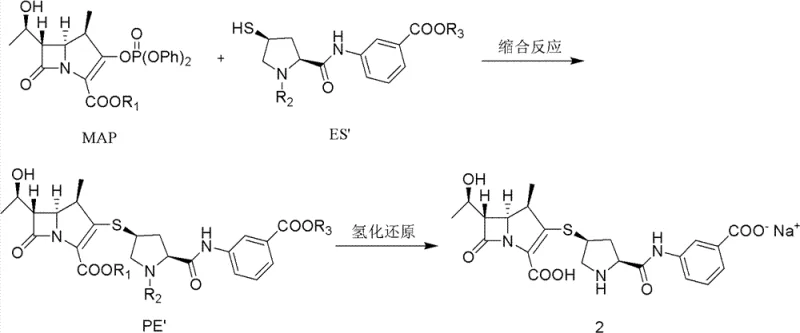
In stark contrast, the novel approach detailed in the patent leverages a single-protection strategy that significantly simplifies the molecular architecture of the intermediate. By utilizing a specifically designed intermediate (PE) that retains a single carboxyl-protecting group, such as p-nitrobenzyl, the synthetic route achieves superior atom economy. The innovation lies not just in the structure, but in the physical handling of this intermediate prior to the final reduction. Instead of subjecting the sensitive beta-lactam scaffold to rigorous vacuum drying at elevated temperatures, which can induce decomposition, the process allows for the direct use of undried intermediates or those dried gently at ambient temperatures. This subtle yet profound shift prevents the formation of degradation products that typically plague high-temperature drying protocols, thereby ensuring a cleaner reaction profile for the subsequent hydrogenation.
Mechanistic Insights into Controlled Moisture Hydrogenation
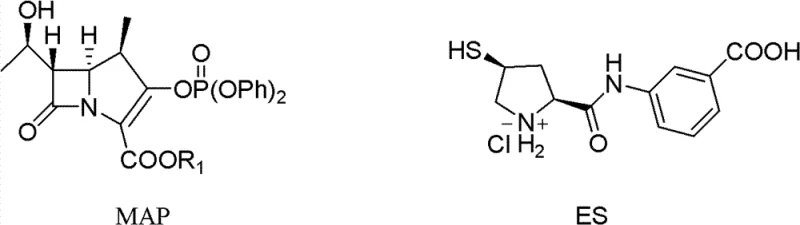
The core of this technological advancement rests on the delicate interplay between the intermediate's physical state and the catalytic hydrogenation environment. The condensation reaction between the 1-beta-methyl carbapenem parent nucleus (MAP) and the ertapenem side chain (ES) is facilitated by strong bases like tetramethyl guanidine (TMG) and catalysts such as 4-dimethylaminopyridine (DMAP). This step forms the protected intermediate PE, which contains a thioether linkage crucial for the antibiotic's activity. However, the stability of this intermediate is paramount; exposure to heat during isolation can trigger hydrolysis or rearrangement of the fragile bicyclic system.
Upon entering the hydrogenation phase, the presence of controlled moisture acts as a stabilizing factor rather than a contaminant. The patent data indicates that intermediates with a water content ranging from 5% to 65%, or even undried cakes with up to 85% moisture, yield superior results compared to overly dried materials. When the intermediate is dried at temperatures exceeding 35°C, the purity of the final ertapenem drops precipitously, often falling below 90% due to the formation of polymeric impurities and degradation byproducts. Conversely, maintaining the moisture content within the specified range allows the palladium charcoal catalyst to function optimally in the mixed solvent system of tetrahydrofuran (THF) and water. This environment facilitates the selective removal of the protecting group without compromising the integrity of the beta-lactam ring, resulting in a final product with purity levels consistently exceeding 96%.
How to Synthesize Ertapenem Sodium Efficiently
The synthesis of ertapenem sodium via this optimized route requires precise control over reaction parameters, particularly during the isolation and hydrogenation stages. The process begins with the condensation of MAP and the side chain ES in a polar aprotic solvent like DMF at cryogenic temperatures to ensure stereochemical control. Following the reaction, the intermediate is precipitated into an acidic aqueous solution, filtered, and washed. Crucially, the subsequent drying step is either bypassed entirely or conducted under mild vacuum at 20-30°C to retain specific moisture levels. The detailed standardized synthesis steps for implementing this high-efficiency protocol are outlined below.
- Condense 1-beta-methyl carbapenem parent nucleus (MAP) with the ertapenem side chain (ES) in the presence of a base like tetramethyl guanidine and a reducing agent to form the protected intermediate PE.
- Isolate the intermediate PE either as an undried wet cake or dry it at a controlled low temperature of 20-30°C to achieve a water content between 5% and 65%.
- Subject the intermediate PE to catalytic hydrogenation using palladium charcoal in a mixed solvent system of THF and water at 15-20°C to yield the final ertapenem sodium product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis method translates into tangible operational efficiencies and risk mitigation. The ability to utilize undried or partially dried intermediates eliminates the energy-intensive and time-consuming vacuum drying steps typically required at high temperatures. This reduction in processing time directly contributes to cost reduction in pharmaceutical intermediates manufacturing by lowering utility consumption and increasing batch throughput. Furthermore, the simplified purification workflow, which avoids complex extraction procedures to remove phosphonic acid impurities, reduces the consumption of organic solvents and minimizes waste generation, aligning with modern green chemistry initiatives.
- Cost Reduction in Manufacturing: The elimination of harsh drying conditions and the associated degradation products means that fewer resources are spent on reprocessing or discarding off-spec batches. By preventing the formation of difficult-to-remove impurities early in the process, the overall yield of the final API is preserved, leading to substantial cost savings in raw material utilization. The streamlined workflow reduces the number of unit operations, thereby decreasing labor costs and equipment occupancy time, which is critical for maximizing facility productivity.
- Enhanced Supply Chain Reliability: The robustness of this method against variations in intermediate moisture content provides a buffer against production delays. Traditional methods that require bone-dry intermediates are susceptible to bottlenecks if drying equipment is unavailable or malfunctioning. This flexible approach ensures that production schedules can be maintained even if the intermediate isolation step varies slightly, thereby reducing lead time for high-purity carbapenem antibiotics and ensuring a steady flow of materials to downstream formulation partners.
- Scalability and Environmental Compliance: The use of milder conditions and the avoidance of aggressive extraction steps make this process highly amenable to commercial scale-up of complex pharmaceutical intermediates. The reduced solvent usage and lower energy demand for drying contribute to a smaller environmental footprint, facilitating easier compliance with increasingly stringent environmental regulations. This scalability ensures that suppliers can meet surging global demand for essential antibiotics without compromising on quality or sustainability metrics.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this specific ertapenem sodium preparation method. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering clarity on the operational parameters that define success in this synthesis.
Q: Why is controlling the moisture content of the intermediate critical in this synthesis?
A: The patent reveals that excessive drying temperatures (above 35°C) cause the intermediate to decompose, significantly increasing impurities in the final hydrogenation step. Maintaining moisture between 5-65% or using the undried intermediate preserves structural integrity and maximizes purity.
Q: How does this method improve upon traditional one-pot synthesis routes?
A: Traditional one-pot methods often carry over inorganic salts and phosphonic acid impurities into the hydrogenation stage, complicating purification. This novel approach isolates the intermediate first, allowing for the removal of these contaminants before the critical hydrogenation step, resulting in a cleaner final product.
Q: What are the optimal conditions for the catalytic hydrogenation step?
A: The process utilizes a mixed solvent system of tetrahydrofuran (THF) and water with a volume ratio of roughly 1.05:1 to 1.5:1. The reaction is conducted at a mild temperature of 15-20°C under a hydrogen pressure of 10-17 atm using palladium charcoal as the catalyst.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ertapenem Sodium Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires more than just a patent; it demands deep process engineering expertise. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the nuanced requirements of carbapenem synthesis are met with precision. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of ertapenem sodium meets the highest international pharmacopoeia standards, providing our partners with unwavering confidence in their supply chain.
We invite you to collaborate with us to leverage this advanced synthesis technology for your antibiotic portfolios. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to technical excellence can drive value for your organization.
