Advanced Esterification Technology for High-Purity Pharmaceutical Intermediates and Commercial Scale-Up
The pharmaceutical industry constantly seeks robust synthetic routes that balance high purity with economic efficiency, particularly for critical intermediates like those used in the synthesis of Telmisartan. Patent CN114835584A introduces a transformative method for the post-treatment of the esterification reaction between 3-methyl-4-nitrobenzoic acid and alcohol, addressing long-standing inefficiencies in traditional organic synthesis. This technology focuses on the recovery and recycling of acid catalysts and solvents, which are typically major contributors to production costs and environmental waste in fine chemical engineering. By implementing a closed-loop system for mother liquor and solvent management, this approach not only enhances the yield of the esterified product but also significantly mitigates the burden of waste acid water treatment. For R&D directors and procurement managers, understanding the nuances of this patent is essential for optimizing supply chains and reducing the overall cost of goods sold for high-value API intermediates. The strategic integration of such recycling protocols represents a shift towards more sustainable and economically viable manufacturing practices in the competitive landscape of global pharmaceutical production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for producing esterified derivatives of 3-methyl-4-nitrobenzoic acid often rely heavily on the use of large quantities of sulfuric acid as a catalyst, followed by dilution with vast amounts of water to induce crystallization. This conventional methodology presents several critical drawbacks that impact both operational efficiency and environmental compliance. Firstly, the addition of large volumes of water to quench the reaction generates substantial amounts of acidic wastewater, which requires complex and expensive treatment processes before disposal. Secondly, the conventional filtration and isolation steps often fail to effectively recover unreacted starting materials, leading to significant material loss and reduced overall yield. Furthermore, the presence of residual heavy metals or organic by-products from the raw materials can poison downstream catalysts, such as palladium on carbon used in subsequent reduction steps, thereby compromising the quality of the final API. These inefficiencies create a bottleneck in production scalability and increase the total cost of manufacturing, making it difficult for suppliers to remain competitive in a price-sensitive market.
The Novel Approach
In stark contrast to these outdated practices, the novel approach detailed in the patent utilizes a sophisticated post-treatment strategy that maximizes resource utilization through systematic recycling. Instead of diluting the reaction mixture with water, the process employs direct cooling crystallization, which allows for the separation of the ester product while keeping the acid catalyst and unreacted materials in the mother liquor. This mother liquor is then concentrated under reduced pressure to recover the alcohol solvent, and the remaining acidic residue is recycled directly into the next batch of production. This innovation effectively breaks the cycle of waste generation by transforming what was previously considered waste into a valuable resource for subsequent reactions. By maintaining the catalytic activity of the acid and recovering the solvent, the process ensures a consistent reaction environment that promotes higher conversion rates. This method not only simplifies the operational workflow but also aligns with modern green chemistry principles, offering a compelling value proposition for manufacturers seeking to reduce their environmental footprint while enhancing production economics.
Mechanistic Insights into Acid-Catalyzed Esterification and Recycling
The core of this technological advancement lies in the precise control of the esterification mechanism under protic acid catalysis. In the presence of a strong acid catalyst such as concentrated sulfuric acid, the carbonyl group of the 3-methyl-4-nitrobenzoic acid becomes protonated, making it more electrophilic and susceptible to nucleophilic attack by the alkyl alcohol. 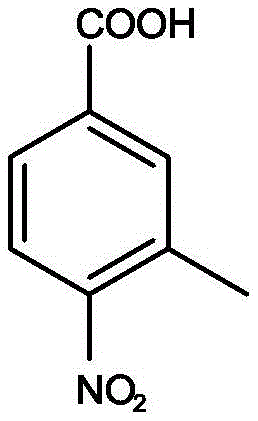 . This reaction is inherently reversible, and the equilibrium is traditionally driven towards the ester product by the removal of water. However, the patented method leverages the dehydrating property of concentrated sulfuric acid to shift this equilibrium without the need for excessive water addition during the workup phase. The recycling of the acidic mother liquor ensures that the catalytic species remain active in the system, reducing the need for fresh acid addition in subsequent batches. This continuous catalytic cycle maintains a high reaction rate and ensures that the conversion of the carboxylic acid to the ester proceeds efficiently, even with reduced fresh reagent inputs. The mechanistic stability provided by this recycling loop is crucial for maintaining batch-to-batch consistency, a key requirement for GMP-compliant pharmaceutical manufacturing.
. This reaction is inherently reversible, and the equilibrium is traditionally driven towards the ester product by the removal of water. However, the patented method leverages the dehydrating property of concentrated sulfuric acid to shift this equilibrium without the need for excessive water addition during the workup phase. The recycling of the acidic mother liquor ensures that the catalytic species remain active in the system, reducing the need for fresh acid addition in subsequent batches. This continuous catalytic cycle maintains a high reaction rate and ensures that the conversion of the carboxylic acid to the ester proceeds efficiently, even with reduced fresh reagent inputs. The mechanistic stability provided by this recycling loop is crucial for maintaining batch-to-batch consistency, a key requirement for GMP-compliant pharmaceutical manufacturing.
Impurity control is another critical aspect where this novel mechanism excels, particularly concerning the removal of organic by-products and potential catalyst poisons. In conventional water crystallization, impurities often co-precipitate with the product or remain trapped in the crystal lattice, affecting the purity profile required for downstream synthesis. The patented method utilizes organic solvent phase crystallization combined with controlled cooling, which selectively precipitates the desired ester while leaving many impurities in the mother liquor. Since the mother liquor is recycled, these impurities are either degraded in subsequent reaction cycles or managed through periodic purging, preventing their accumulation to detrimental levels. Furthermore, the removal of residual heavy metals from the raw materials is facilitated by the specific solubility characteristics of the ester in the organic phase, ensuring that the final product meets stringent purity specifications. This level of impurity management is vital for protecting sensitive downstream catalysts and ensuring the safety and efficacy of the final pharmaceutical product, thereby reducing the risk of batch failures and regulatory non-compliance.
How to Synthesize Methyl 3-Methyl-4-Nitrobenzoate Efficiently
Implementing this synthesis route requires a disciplined approach to reaction conditions and separation techniques to fully realize the benefits of the recycling protocol. The process begins with the mixing of 3-methyl-4-nitrobenzoic acid and a selected alcohol solvent, followed by the controlled addition of an acid catalyst under heating conditions to initiate the esterification. The reaction is monitored closely to ensure that the residual acid content drops below a specific threshold, indicating high conversion. Following the reaction, the mixture is cooled to induce crystallization, and the solid product is separated via filtration. The critical step involves the treatment of the filtrate, where the solvent is recovered via distillation, and the acidic residue is preserved for reuse. This sequence ensures that the valuable catalytic components are not lost, and the solvent is returned to the process in a dry, usable state. Detailed standardized synthesis steps see the guide below.
- Mix 3-methyl-4-nitrobenzoic acid with a solvent and heat with alkyl alcohol under acid catalysis to generate the ester.
- Cool the reaction liquid to crystallize the product, then filter and separate to obtain the purified esterification product.
- Concentrate the filtered mother liquor to recover solvent and recycle the acidic mother liquor for the next batch production.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented post-treatment method offers substantial strategic advantages that extend beyond simple technical metrics. The primary benefit lies in the drastic simplification of the waste management infrastructure required for production. By recycling the acid catalyst and solvent, the volume of hazardous waste requiring external treatment is significantly reduced, leading to lower disposal fees and reduced regulatory burden. This efficiency translates directly into cost reduction in pharmaceutical intermediate manufacturing, allowing suppliers to offer more competitive pricing without compromising on quality. Additionally, the ability to recover and reuse unreacted raw materials minimizes the consumption of expensive starting materials, further enhancing the economic viability of the process. These factors combine to create a more resilient supply chain that is less susceptible to fluctuations in raw material costs and waste disposal regulations.
- Cost Reduction in Manufacturing: The elimination of excessive water usage and the recycling of high-boiling acid catalysts remove the need for expensive neutralization and wastewater treatment steps. This qualitative shift in process design means that the operational expenditure associated with environmental compliance is drastically lowered. Furthermore, the recovery of unreacted benzoic acid derivatives ensures that raw material utilization is maximized, reducing the effective cost per kilogram of the final product. By avoiding the purchase of fresh catalyst for every batch, the cumulative savings over a production year become substantial, providing a clear margin advantage for manufacturers adopting this technology.
- Enhanced Supply Chain Reliability: The simplified workflow reduces the complexity of the production schedule, minimizing the risk of delays associated with waste handling and solvent procurement. Since the process recycles its own solvents and catalysts, the dependency on external suppliers for these specific consumables is reduced, mitigating supply chain disruptions. The robustness of the crystallization method also ensures consistent product quality, reducing the likelihood of batch rejections that can disrupt delivery timelines. This reliability is crucial for maintaining long-term contracts with pharmaceutical companies that demand just-in-time delivery of high-purity intermediates.
- Scalability and Environmental Compliance: The mild reaction conditions and the absence of complex quenching steps make this process highly scalable from pilot plant to commercial production volumes. The reduction in waste acid generation aligns with increasingly strict environmental regulations, future-proofing the manufacturing facility against tighter compliance standards. This environmental stewardship enhances the corporate reputation of the supplier and reduces the risk of operational shutdowns due to environmental violations. The ease of scaling ensures that supply can be ramped up quickly to meet market demand without significant capital investment in new waste treatment infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this esterification post-treatment method. These answers are derived directly from the technical disclosures and experimental data provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details helps stakeholders evaluate the feasibility of integrating this technology into their existing manufacturing frameworks. The focus is on practical application, yield optimization, and regulatory compliance, which are the primary concerns for decision-makers in the pharmaceutical supply chain.
Q: How does this patent method improve yield compared to conventional esterification?
A: The method recycles unreacted raw materials along with the acidic mother liquor back into the next batch, significantly increasing overall product yield and reducing material loss compared to single-pass methods.
Q: What are the environmental benefits of this post-treatment process?
A: By recycling acid catalysts like sulfuric acid and solvents, the process drastically reduces the volume of waste acid water requiring treatment, lowering environmental impact and disposal costs.
Q: Is this process suitable for large-scale Telmisartan intermediate production?
A: Yes, the process features mild reaction conditions and simple operation steps like cooling crystallization and filtration, making it highly adaptable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Methyl-4-Nitrobenzoic Acid Supplier
The technical potential of this esterification route is immense, offering a pathway to more sustainable and cost-effective production of critical pharmaceutical intermediates. NINGBO INNO PHARMCHEM, as a leading CDMO expert, possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring such innovative processes to life. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, ensuring that every batch of 3-methyl-4-nitrobenzoic acid derivatives meets the highest industry standards. We understand the complexities of managing acid-catalyzed reactions and solvent recycling at scale, and our team is dedicated to optimizing these parameters to deliver maximum value to our partners. By leveraging our technical expertise, clients can accelerate their development timelines and secure a stable supply of high-quality intermediates.
We invite you to explore how our capabilities can enhance your supply chain efficiency and reduce your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs. We encourage you to contact us to request specific COA data and route feasibility assessments for your projects. By collaborating with us, you gain access to a partner committed to innovation, quality, and reliability, ensuring that your critical drug development programs proceed without interruption. Let us help you navigate the complexities of fine chemical synthesis with confidence and precision.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
