Advanced Photo-Catalytic Synthesis of 3-Methyl-4-Nitrobenzoic Acid for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust and environmentally sustainable pathways for synthesizing critical antihypertensive drug intermediates, and recent intellectual property developments highlight significant strides in this domain. Specifically, patent CN113480433A discloses a groundbreaking preparation method for 3-methyl-4-nitrobenzoic acid, a pivotal building block in the manufacture of Telmisartan. This technical insight report analyzes the transformative potential of this photo-catalytic oxidation strategy, which shifts away from harsh traditional oxidants toward a greener, light-driven process utilizing molecular oxygen. By leveraging a synergistic catalyst system comprising hematoporphyrin derivatives and nickel oxide under illumination, the process achieves exceptional conversion rates exceeding 98 percent while maintaining mild reaction temperatures. For global procurement leaders and R&D directors, understanding this shift is crucial for securing a reliable pharmaceutical intermediates supplier capable of delivering high-purity materials with a reduced environmental footprint. The following analysis details how this innovation addresses long-standing bottlenecks in selectivity and operational safety, offering a viable route for cost reduction in pharma manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-methyl-4-nitrobenzoic acid has been plagued by inefficient and hazardous chemical processes that struggle to meet modern green chemistry standards. Conventional routes often rely on the use of dilute nitric acid as a stoichiometric oxidant, a method that generates substantial quantities of nitrogen oxide emissions and acidic wastewater, creating severe burdens for waste treatment facilities and environmental compliance teams. Alternatively, prior art describes aerobic oxidation methods utilizing cobalt acetate and butanone in an acetic acid medium, which necessitate aggressive reaction conditions including temperatures around 85 degrees Celsius and elevated air pressures of 0.5 MPa. Despite these energy-intensive requirements, the performance of these legacy methods remains suboptimal, with reported maximum conversion rates hovering around 58 percent and selectivity for the desired 3-methyl-4-nitrobenzoic acid isomer dropping as low as 50 percent. Consequently, the actual isolated yield in these traditional processes is often a dismal 29 percent, leading to excessive raw material consumption and inflated production costs that erode profit margins for downstream API manufacturers.
The Novel Approach
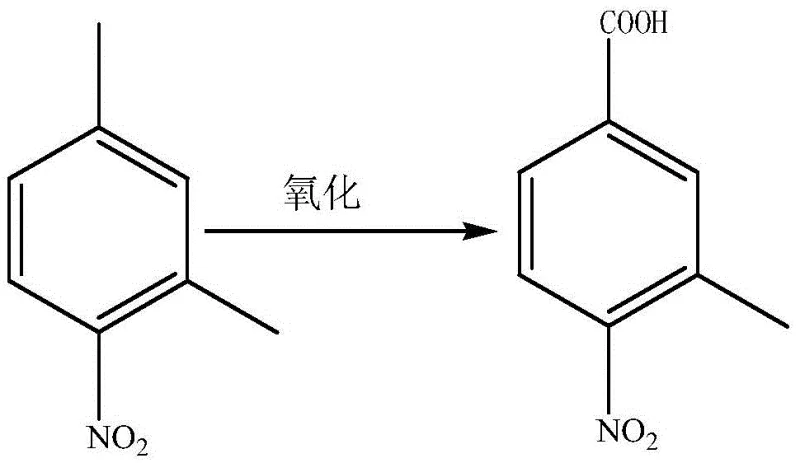
In stark contrast to these cumbersome legacy techniques, the novel approach detailed in the patent data introduces a highly selective photo-catalytic oxidation system that fundamentally redefines the efficiency landscape for this transformation. By employing 2,4-dimethylnitrobenzene as the starting material and subjecting it to oxygen introduction under stirring and specific illumination in an acetonitrile solvent, the reaction proceeds with remarkable precision at a mild temperature of 30 degrees Celsius. The integration of a dual-catalyst system, featuring hematoporphyrin or its monomethyl ether alongside NiO, facilitates the activation of molecular oxygen under light irradiation, driving the selective oxidation of the 4-methyl group to the carboxylic acid functionality without over-oxidizing the remaining methyl group. This methodology not only eliminates the need for high-pressure reactors and corrosive nitric acid but also boosts the conversion rate of the starting material to greater than or equal to 98 percent, with product yields reaching approximately 65 percent. Such a dramatic improvement in efficiency underscores the value of partnering with a reliable pharmaceutical intermediates supplier who adopts cutting-edge technologies to ensure supply stability and quality consistency.
Mechanistic Insights into Photo-Catalytic Selective Oxidation
The core of this technological breakthrough lies in the sophisticated interplay between the photo-sensitive porphyrin catalyst and the nickel oxide co-catalyst under visible or near-UV light irradiation. When the reaction mixture is exposed to light from a high-pressure mercury lamp with a filtering wavelength of less than 380 nanometers, the hematoporphyrin molecules absorb photon energy and transition to an excited state, effectively acting as photosensitizers. In this excited state, the catalyst transfers energy to molecular oxygen dissolved in the acetonitrile solvent, generating reactive oxygen species such as singlet oxygen or superoxide radicals that are potent yet controllable oxidants. The presence of NiO likely serves to stabilize these reactive intermediates or facilitate electron transfer processes, ensuring that the oxidation occurs specifically at the benzylic position of the 4-methyl group rather than indiscriminately attacking the aromatic ring or the nitro group. This precise mechanistic control is what allows the process to achieve such high selectivity, avoiding the formation of dicarboxylic acid byproducts or other oxidative degradation impurities that typically plague non-selective radical oxidations.
Furthermore, the mechanism inherently supports superior impurity control, which is a primary concern for R&D directors focused on the purity profile of API intermediates. Because the reaction conditions are mild (30 degrees Celsius) and the oxidant is generated in situ via a catalytic cycle rather than added as a harsh bulk reagent, side reactions such as nitration or ring chlorination are virtually eliminated. The heterogeneous nature of the NiO component also aids in the purification process; post-reaction, the solid catalyst can be removed via simple filtration, preventing metal contamination in the final filtrate. Following solvent recovery via rotary evaporation, the crude product is precipitated by adding water and adjusting the pH to approximately 3.8, a step that further purifies the material by keeping soluble impurities in the aqueous phase while crystallizing the target acid. This multi-layered approach to impurity management ensures that the resulting 3-methyl-4-nitrobenzoic acid meets stringent purity specifications, often exceeding 96 percent, thereby reducing the burden on downstream purification steps and enhancing the overall commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize 3-Methyl-4-Nitrobenzoic Acid Efficiently
Implementing this advanced synthesis route requires careful attention to the photoreactor setup and the precise dosing of the catalyst system to maximize the quantum yield of the oxidation. The process begins by charging a specialized photoreactor, equipped with a filter tube to control wavelength exposure, with acetonitrile solvent and initiating a flow of oxygen gas from the lower section to ensure adequate saturation. Once the system is stabilized, the substrate 2,4-dimethylnitrobenzene is introduced alongside the specific catalyst loading of hematoporphyrin and NiO, after which the high-pressure mercury lamp is activated to drive the reaction at a controlled temperature of 30 degrees Celsius for a duration of 5 hours. Upon completion, the workflow involves a straightforward workup procedure consisting of filtration to remove the solid catalyst, solvent recovery, and acidification to isolate the pure product, representing a significant simplification over multi-step traditional syntheses. The detailed standardized synthetic steps, including exact reagent quantities and equipment specifications, are outlined in the guide below for technical reference.
- Charge a photoreactor with acetonitrile solvent and introduce oxygen gas from the bottom while stirring.
- Add 2,4-dimethylnitrobenzene substrate along with hematoporphyrin (or its monomethyl ether) and NiO catalyst.
- Illuminate with a high-pressure mercury lamp (wavelength <380nm) at 30°C for 5 hours, then filter and acidify to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this photo-catalytic technology translates into tangible strategic benefits that extend far beyond simple chemical yield improvements. The shift from high-pressure, high-temperature batch processes to a mild, ambient-pressure photochemical reaction drastically reduces the energy consumption associated with heating and compression, leading to significant operational expenditure savings over the lifecycle of the product. Moreover, the elimination of corrosive nitric acid and the reduction of heavy metal usage (replacing soluble cobalt with recoverable nickel oxide) simplifies the waste management protocol, lowering the costs associated with hazardous waste disposal and environmental remediation. These factors collectively contribute to a more resilient supply chain, as the process is less dependent on specialized high-pressure equipment that often creates bottlenecks in manufacturing capacity, thereby reducing lead time for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The economic advantages of this method are driven primarily by the drastic simplification of the reaction conditions and the efficiency of the catalyst system. By operating at 30 degrees Celsius instead of 85 degrees Celsius, the facility saves substantial amounts of steam or electrical heating energy, while the atmospheric pressure operation removes the need for expensive autoclaves and the safety protocols associated with pressurized vessels. Additionally, the ability to recover and reuse the NiO catalyst means that the recurring cost of catalyst consumption is minimized, and the high conversion rate ensures that expensive raw materials like 2,4-dimethylnitrobenzene are utilized with maximum efficiency, minimizing waste loss. This holistic reduction in input costs allows for a more competitive pricing structure without compromising on the quality of the final intermediate.
- Enhanced Supply Chain Reliability: From a logistics and planning perspective, the robustness of this new method offers superior reliability compared to the finicky nature of traditional cobalt-catalyzed oxidations. The reaction is described as stable and easy to control, which reduces the risk of batch failures or off-spec material that can disrupt downstream API production schedules. Furthermore, the starting materials and solvents involved, such as acetonitrile and oxygen, are commodity chemicals with stable global supply lines, reducing the risk of raw material shortages. The simplicity of the workup—filtration, evaporation, and precipitation—also means that the throughput time per batch can be optimized, allowing manufacturers to respond more agilely to fluctuations in market demand for Telmisartan intermediates.
- Scalability and Environmental Compliance: Scaling photochemical reactions has historically been a challenge due to light penetration limits, but the specific reactor design implied in the patent suggests a viable path for commercial scale-up of complex pharmaceutical intermediates. The process generates minimal hazardous byproducts, aligning perfectly with increasingly strict global environmental regulations regarding VOC emissions and heavy metal discharge. By avoiding the generation of nitrogen oxides and acidic wastewater, the facility reduces its regulatory burden and the risk of shutdowns due to compliance violations. This environmental stewardship not only future-proofs the supply chain against tightening regulations but also enhances the brand reputation of the supplier as a sustainable partner for green chemistry initiatives.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of integrating this intermediate into their supply chain, we have compiled answers to common inquiries based on the specific technical parameters of the patent. These responses address critical concerns regarding catalyst toxicity, scalability of photo-reactors, and the comparative purity profiles against incumbent methods. Understanding these nuances is essential for making informed sourcing decisions that balance cost, quality, and risk.
Q: What are the advantages of this photo-catalytic method over traditional nitric acid oxidation?
A: Unlike traditional methods using dilute nitric acid which generate significant acidic waste, or cobalt-catalyzed air oxidation requiring high temperature (85°C) and pressure (0.5MPa) with low yields (29%), this novel method operates at mild conditions (30°C) with high conversion (>98%) and uses recoverable catalysts.
Q: Is the catalyst system reusable in this synthesis?
A: Yes, the process utilizes NiO which can be separated and recovered after the reaction, contributing to lower operational costs and reduced metal waste compared to homogeneous cobalt systems.
Q: What is the purity profile of the resulting 3-methyl-4-nitrobenzoic acid?
A: The optimized process yields 3-methyl-4-nitrobenzoic acid with high purity, demonstrated at approximately 96.7% in experimental examples, making it suitable for downstream pharmaceutical applications like Telmisartan synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Methyl-4-Nitrobenzoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes like the photo-catalytic oxidation of 2,4-dimethylnitrobenzene requires a partner with deep technical expertise and proven manufacturing capabilities. As a dedicated CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent can be realized in a real-world industrial setting. Our facilities are equipped with state-of-the-art photo-reactors and rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 3-methyl-4-nitrobenzoic acid meets the exacting standards required for antihypertensive drug synthesis. We are committed to bridging the gap between innovative academic research and reliable commercial supply.
We invite global pharmaceutical partners to engage with our technical procurement team to explore how this optimized route can enhance your specific supply chain dynamics. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the potential economic impact of switching to this greener, more efficient method. We encourage you to contact us directly to obtain specific COA data from our pilot batches and to discuss route feasibility assessments tailored to your volume requirements, ensuring a seamless integration of this high-value intermediate into your production pipeline.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
