Advanced Ionic Liquid Catalysis for High-Purity Cyanoacetate Production and Commercial Scale-Up
Advanced Ionic Liquid Catalysis for High-Purity Cyanoacetate Production and Commercial Scale-Up
The chemical industry is currently witnessing a paradigm shift towards greener, more sustainable synthesis pathways, particularly in the production of critical pharmaceutical intermediates like cyanoacetates. Patent CN102786436A introduces a groundbreaking methodology that replaces traditional, corrosive mineral acid catalysts with novel acidic ionic liquids, fundamentally altering the efficiency and environmental profile of esterification reactions. This technology leverages the unique physicochemical properties of imidazolium, pyrrolidone, and pyridine-based ionic liquids to catalyze the reaction between cyanoacetic acid and various alcohols under mild conditions. By eliminating the need for excessive amounts of dehydrating agents and avoiding the generation of hazardous waste streams, this process addresses long-standing pain points in fine chemical manufacturing. For R&D directors and process engineers, this represents a significant opportunity to optimize impurity profiles and streamline downstream processing. Furthermore, the ability to recycle the catalytic medium offers a compelling value proposition for procurement teams focused on long-term cost stability and supply chain resilience in the competitive landscape of API intermediate production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of cyanoacetates, such as the widely used ethyl cyanoacetate, has relied heavily on the Fischer esterification method catalyzed by concentrated sulfuric acid. This traditional approach is fraught with significant operational and environmental drawbacks that hinder modern manufacturing efficiency. The use of strong mineral acids necessitates the employment of specialized, corrosion-resistant reactor materials, driving up capital expenditure and maintenance costs for production facilities. Moreover, the reaction equilibrium in sulfuric acid-catalyzed systems often requires a large excess of alcohol to drive the conversion, leading to increased raw material consumption and complex recovery processes for unreacted solvents. Perhaps most critically, the post-reaction workup involves neutralization and washing steps that generate substantial volumes of acidic wastewater, creating a heavy burden on waste treatment infrastructure and complicating regulatory compliance. The difficulty in separating the product from the catalyst mixture often results in lower selectivity and purity, requiring energy-intensive distillation steps that further erode profit margins and increase the carbon footprint of the manufacturing process.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes task-specific acidic ionic liquids that function simultaneously as the reaction medium and the catalyst, effectively resolving the inefficiencies of the legacy technology. These ionic liquids, characterized by their negligible vapor pressure and tunable acidity, provide a homogeneous environment that enhances the interaction between cyanoacetic acid and the alcohol substrate. The process operates at moderate temperatures ranging from 40°C to 100°C, significantly reducing energy consumption compared to the harsh conditions often required by traditional methods. A key advantage lies in the phase behavior of the reaction system; upon completion, the ester product often separates spontaneously or can be easily induced to separate by the addition of a small amount of water, simplifying isolation to a mere decantation or separation funnel operation. This inherent ease of separation not only improves the overall yield but also preserves the integrity of the ionic liquid phase, which can be regenerated and reused, thereby establishing a closed-loop system that minimizes waste generation and maximizes atom economy in the synthesis of high-value cyanoacetate derivatives.
Mechanistic Insights into Ionic Liquid Catalyzed Esterification
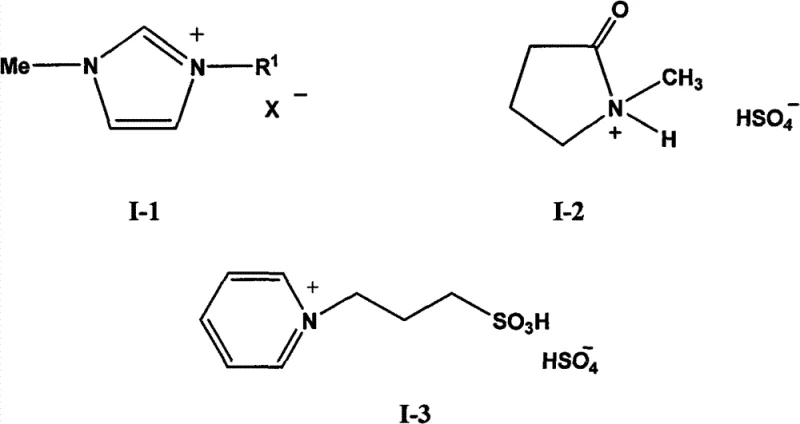
The efficacy of this synthesis route is rooted in the Brønsted acidic nature of the selected ionic liquids, which facilitate the protonation of the carbonyl oxygen in cyanoacetic acid, thereby increasing its electrophilicity towards nucleophilic attack by the alcohol. The specific structures employed, such as the sulfonic acid-functionalized imidazolium salts or the protonated pyrrolidone derivatives, possess strong acidic protons that mimic the catalytic activity of sulfuric acid but within a structured, non-volatile ionic matrix. This dual functionality eliminates the mass transfer limitations often observed in biphasic systems and ensures a high concentration of active catalytic sites throughout the reaction volume. The stability of the ionic liquid framework prevents degradation under the reaction conditions, ensuring consistent catalytic performance over multiple cycles. Furthermore, the hydrophobicity or hydrophilicity of the ionic liquid can be tuned by modifying the anion (e.g., HSO4⁻ or BF4⁻) or the cation side chains, allowing for precise control over the partition coefficient of the product and facilitating the spontaneous phase separation that is critical for efficient downstream processing.
Controlling the impurity profile is another critical aspect where this mechanistic approach excels, particularly for applications in pharmaceutical intermediates where strict purity specifications are mandatory. The mild acidic environment provided by the ionic liquid suppresses side reactions such as polymerization or decomposition of the sensitive cyano group, which are common pitfalls in strongly acidic mineral acid media. The absence of oxidizing species, which can sometimes be present as impurities in industrial grade sulfuric acid, further protects the integrity of the cyanoacetate product. Additionally, the recyclability of the ionic liquid means that any potential accumulation of byproducts can be managed through periodic purification of the catalyst phase, rather than introducing fresh contaminants with every batch as happens when using stoichiometric amounts of mineral acids. This results in a cleaner crude product that requires less rigorous purification, reducing the load on distillation columns and minimizing thermal stress on the final API intermediate, ultimately delivering a higher quality material to the end-user.
How to Synthesize Cyanoacetate Efficiently
The practical implementation of this technology involves a straightforward protocol that integrates seamlessly into existing batch reactor setups with minimal modification. The process begins with the precise metering of cyanoacetic acid, the target alcohol, and the selected ionic liquid catalyst into the reaction vessel, adhering to the optimized molar ratios defined in the patent to ensure maximum conversion efficiency. The mixture is then heated to the specified temperature range and maintained under agitation for the requisite time to drive the esterification to completion. Following the reaction, the system is allowed to cool and settle, leveraging the density differences between the ionic liquid phase and the organic ester phase to achieve separation. For a comprehensive understanding of the specific operational parameters, safety considerations, and workup procedures required to replicate these results in a pilot or production setting, please refer to the standardized synthesis guide below.
- Mix cyanoacetic acid, the chosen alcohol (C1-C8, cyclohexyl, or benzyl), and the acidic ionic liquid catalyst in a molar ratio ranging from 0.1: 1:1 to 5:1:5.
- Heat the reaction mixture to a temperature between 40°C and 100°C and stir continuously for a duration of 2 to 15 hours to ensure complete esterification.
- Allow the reaction system to stand for phase separation; isolate the upper ester layer directly, or add water to induce layering if the system is homogeneous, then recover and recycle the ionic liquid layer.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this ionic liquid catalyzed process offers transformative benefits that extend far beyond the laboratory bench, directly impacting the bottom line and operational reliability of chemical supply chains. The elimination of corrosive sulfuric acid removes the need for expensive Hastelloy or glass-lined reactors, allowing for the use of more cost-effective stainless steel equipment in many cases, which represents a significant capital cost avoidance for manufacturers scaling up production. The simplified workup procedure, which often requires only phase separation rather than complex neutralization and extraction sequences, drastically reduces the consumption of auxiliary chemicals like bases and wash waters, leading to substantial reductions in operating expenses. Furthermore, the ability to recycle the ionic liquid catalyst multiple times without significant loss of activity transforms the catalyst from a consumable cost center into a durable asset, amortizing the initial investment over a much larger production volume and stabilizing the variable cost per kilogram of the final product.
- Cost Reduction in Manufacturing: The economic model of this process is driven by the drastic simplification of the downstream processing train. By avoiding the generation of large volumes of acidic wastewater, manufacturers save significantly on effluent treatment costs, which are a major component of operational expenditure in the fine chemical sector. The high selectivity of the reaction minimizes the formation of tars and heavy ends, improving the yield of the desired ester and reducing the loss of valuable raw materials. Additionally, the reduced reaction time and lower temperature requirements translate directly into lower energy consumption for heating and stirring, contributing to a leaner and more cost-efficient manufacturing process that enhances competitiveness in the global market for pharma intermediates.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the regulatory and logistical challenges associated with handling and transporting hazardous corrosive acids. By substituting sulfuric acid with stable, non-volatile ionic liquids, the supply chain becomes more robust and less susceptible to disruptions caused by hazardous material transport restrictions. The simplicity of the process also reduces the risk of batch failures due to operator error or equipment malfunction, ensuring a more consistent and reliable output of high-purity cyanoacetates. This reliability is crucial for downstream customers in the pharmaceutical industry who require just-in-time delivery of critical intermediates to maintain their own production schedules without interruption.
- Scalability and Environmental Compliance: As regulatory pressures regarding environmental protection intensify globally, the green chemistry credentials of this process provide a strategic advantage for long-term sustainability. The negligible vapor pressure of ionic liquids eliminates VOC emissions, helping facilities meet stringent air quality standards without additional abatement equipment. The scalability of the process is enhanced by the ease of heat management and phase separation, which are often bottlenecks in scaling up traditional exothermic esterifications. This allows for a smoother transition from pilot scale to multi-ton commercial production, ensuring that the supply of cyanoacetates can grow in tandem with market demand while maintaining full compliance with evolving environmental regulations.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating the feasibility of integrating this technology into their operations, we have compiled a set of frequently asked questions based on the technical disclosures and experimental data provided in the patent documentation. These inquiries address common concerns regarding catalyst lifecycle, substrate scope, and process safety, offering clarity on how this method compares to incumbent technologies. Understanding these nuances is essential for conducting a thorough techno-economic analysis and making informed sourcing decisions for your chemical supply chain.
Q: What are the primary advantages of using ionic liquids over traditional sulfuric acid for cyanoacetate synthesis?
A: Unlike traditional concentrated sulfuric acid methods which cause severe equipment corrosion and generate difficult-to-treat waste acid, ionic liquids act as both catalyst and solvent, allowing for simple product separation, catalyst recyclability, and significantly reduced environmental impact.
Q: Can the ionic liquid catalyst be reused in this synthesis process?
A: Yes, the ionic liquid layer can be recovered after the reaction by water extraction and washing, followed by heating under reduced pressure to remove water and organic matter, allowing it to be reused multiple times without significant loss of catalytic activity.
Q: What types of alcohols are compatible with this ionic liquid catalyzed esterification method?
A: The method is highly versatile and supports a wide range of alcohols including straight or branched C1 to C8 monohydric saturated alcohols, as well as cyclic alcohols like cyclohexanol and aromatic alcohols like benzyl alcohol.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyanoacetate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the evolving demands of the global pharmaceutical and fine chemical industries. Our team of expert chemists and process engineers possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory techniques like the ionic liquid catalyzed esterification described in CN102786436A can be successfully translated into robust industrial processes. We are committed to delivering high-purity cyanoacetates that meet stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the highest international standards. Our state-of-the-art facilities are equipped to handle the specific requirements of ionic liquid chemistry, providing a secure and efficient platform for the manufacture of these valuable intermediates.
We invite procurement managers and R&D leaders to collaborate with us to leverage these technological advancements for your specific projects. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this greener synthesis route for your supply chain. We encourage you to contact our technical procurement team today to request specific COA data for our cyanoacetate portfolio and to discuss route feasibility assessments tailored to your unique production needs, ensuring a partnership that drives both innovation and profitability.
