Advanced Manufacturing of Photoinitiator 1173 via Green Catalytic Routes
Introduction to Patent CN108892605B
The chemical manufacturing landscape is undergoing a significant transformation driven by the urgent need for safer and more sustainable synthetic routes, a shift clearly exemplified by the technological breakthroughs detailed in patent CN108892605B. This specific intellectual property discloses a revolutionary preparation process for Photoinitiator 1173, chemically known as 2-hydroxy-2-methyl-1-phenyl-1-acetone, which serves as a critical component in UV-curing applications across the coatings and ink industries. Unlike conventional methodologies that rely on a fragmented four-step sequence involving hazardous halogenating agents, this invention consolidates the synthesis into a streamlined two-stage operation. The core innovation lies in the substitution of traditional acyl chloride formation and Friedel-Crafts acylation with a direct high-temperature ketone synthesis using solid acid catalysts. For R&D directors and technical leaders, this represents a paradigm shift towards heterogeneous catalysis that promises not only higher selectivity but also a fundamental restructuring of the production safety profile. By integrating a one-pot chlorination and hydrolysis strategy in the second stage, the process effectively bypasses the handling of elemental chlorine gas, thereby addressing some of the most persistent pain points in fine chemical manufacturing regarding operator safety and environmental compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional industrial pathways for synthesizing Photoinitiator 1173 have long been plagued by inherent safety hazards and complex waste management challenges that strain both operational budgets and regulatory compliance frameworks. The legacy process typically necessitates a four distinct working sections, beginning with the reaction of isobutyric acid with phosphorus trichloride to generate isobutyryl chloride, a step that produces corrosive byproducts and requires rigorous moisture control. Subsequently, this intermediate undergoes a Friedel-Crafts reaction with benzene catalyzed by aluminum trichloride, a procedure that generates substantial quantities of acidic aluminum wastewater and consumes large volumes of benzene, a known carcinogen with strict exposure limits. The downstream processing further exacerbates these issues by employing elemental chlorine gas for alpha-chlorination, introducing severe toxicity risks and requiring specialized gas handling infrastructure. Finally, the alkaline hydrolysis step generates additional saline waste streams, culminating in a cumulative three-waste profile that includes high-phosphorus wastewater, aluminum-containing sludge, and hydrogen chloride emissions. These factors collectively impose a heavy financial burden on manufacturers through elevated costs for waste treatment facilities, hazardous material storage, and personal protective equipment, while simultaneously creating bottlenecks in production continuity due to the batch nature of these reactive steps.
The Novel Approach
In stark contrast to the fragmented and hazardous legacy workflows, the novel approach outlined in the patent data introduces a cohesive and environmentally benign strategy that fundamentally reimagines the molecular construction of the target ketone. The first major innovation is the high-temperature ketone making section, where benzoic acid and isobutyric acid are directly condensed over a metal salt catalyst at temperatures ranging from 300°C to 500°C. This direct decarboxylative condensation elegantly bypasses the need for acyl chloride intermediates and Lewis acid catalysts like aluminum trichloride, effectively eliminating the generation of aluminum wastewater and the consumption of benzene solvent.  As illustrated in the reaction scheme, this single catalytic step achieves what previously required two distinct and dangerous chemical transformations. Furthermore, the subsequent processing stage utilizes a one-pot alkali chloride hydrolysis method employing carbon tetrachloride and sodium hydroxide under phase transfer catalysis. This modification removes the necessity for handling gaseous chlorine, replacing it with a safer liquid-phase reagent system that simplifies reactor design and operational protocols. The consolidation of these steps not only enhances the overall atom economy of the process but also significantly reduces the physical footprint required for production, offering a compelling value proposition for facilities looking to modernize their asset base while mitigating regulatory risks associated with hazardous chemical inventories.
As illustrated in the reaction scheme, this single catalytic step achieves what previously required two distinct and dangerous chemical transformations. Furthermore, the subsequent processing stage utilizes a one-pot alkali chloride hydrolysis method employing carbon tetrachloride and sodium hydroxide under phase transfer catalysis. This modification removes the necessity for handling gaseous chlorine, replacing it with a safer liquid-phase reagent system that simplifies reactor design and operational protocols. The consolidation of these steps not only enhances the overall atom economy of the process but also significantly reduces the physical footprint required for production, offering a compelling value proposition for facilities looking to modernize their asset base while mitigating regulatory risks associated with hazardous chemical inventories.
Mechanistic Insights into High-Temperature Catalytic Condensation
From a mechanistic perspective, the high-temperature ketone making section represents a sophisticated application of heterogeneous catalysis designed to maximize throughput while minimizing side reactions. The process utilizes a composite catalyst system where zinc oxide and magnesium oxide are supported on a high-surface-area carrier such as aluminum oxide or silicon dioxide. This specific formulation is critical because the basic sites provided by the zinc and magnesium oxides facilitate the deprotonation and activation of the carboxylic acid substrates, while the thermal stability of the support ensures longevity under the rigorous conditions of 300°C to 500°C. The reaction mechanism likely proceeds through a surface-bound anhydride intermediate or a direct decarboxylative coupling, where the evolution of carbon dioxide and water drives the equilibrium forward. For technical teams, understanding the catalyst preparation is vital; the patent specifies a soaking and calcination protocol that ensures optimal dispersion of the active metal species, which directly correlates to the observed high selectivity of over 96% for the isobutyrophenone intermediate. This level of selectivity is paramount for downstream purification, as it minimizes the formation of heavy ends or polymeric byproducts that could foul distillation columns, thereby ensuring a consistent quality of feedstock for the subsequent functionalization steps.
The second mechanistic pillar of this invention is the one-pot chlorination and hydrolysis sequence, which leverages phase transfer catalysis to overcome solubility barriers between organic and aqueous phases. In this system, tetrabutylammonium bromide acts as the phase transfer agent, shuttling hydroxide ions from the aqueous sodium hydroxide phase into the organic phase containing the ketone and carbon tetrachloride. 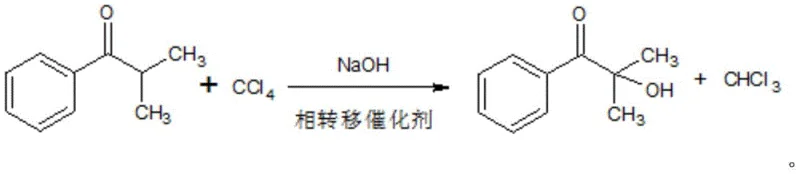 This intimate contact allows for the rapid alpha-chlorination of the ketone followed immediately by nucleophilic substitution to install the hydroxyl group, all within a single reactor vessel. This tandem transformation is kinetically favorable because the intermediate chloroketone is consumed as soon as it is formed, preventing the accumulation of potentially unstable or hazardous chlorinated species. The use of carbon tetrachloride as both a solvent and a chlorinating source is a strategic choice that simplifies reagent logistics, although it requires careful recovery and recycling protocols to maintain environmental standards. For R&D directors, this mechanism offers a robust platform for impurity control, as the homogeneous nature of the reaction environment (facilitated by the phase transfer catalyst) ensures uniform reaction rates throughout the bulk solution, leading to a narrower impurity profile compared to heterogeneous gas-liquid chlorination methods.
This intimate contact allows for the rapid alpha-chlorination of the ketone followed immediately by nucleophilic substitution to install the hydroxyl group, all within a single reactor vessel. This tandem transformation is kinetically favorable because the intermediate chloroketone is consumed as soon as it is formed, preventing the accumulation of potentially unstable or hazardous chlorinated species. The use of carbon tetrachloride as both a solvent and a chlorinating source is a strategic choice that simplifies reagent logistics, although it requires careful recovery and recycling protocols to maintain environmental standards. For R&D directors, this mechanism offers a robust platform for impurity control, as the homogeneous nature of the reaction environment (facilitated by the phase transfer catalyst) ensures uniform reaction rates throughout the bulk solution, leading to a narrower impurity profile compared to heterogeneous gas-liquid chlorination methods.
How to Synthesize Photoinitiator 1173 Efficiently
Implementing this advanced synthesis route requires precise adherence to the thermal and catalytic parameters defined in the patent to ensure optimal yield and safety performance. The process begins with the meticulous preparation of the fixed-bed catalyst, involving the impregnation of metal oxides onto a refractory support followed by high-temperature calcination to activate the surface sites. Operators must then manage the feed rates of the liquefied benzoic and isobutyric acid mixture carefully, maintaining a steady flow through the heated reactor zone to prevent hot spots that could lead to catalyst deactivation or thermal runaway. The detailed standardized synthesis steps, including specific molar ratios, temperature gradients, and workup procedures, are critical for reproducing the high selectivity reported in the examples.
- Prepare a fixed-bed catalyst by impregnating zinc oxide and magnesium oxide onto an aluminum oxide support, then calcine at high temperature.
- Pass a preheated mixture of benzoic acid and isobutyric acid through the fixed-bed reactor at 300-500°C to generate isobutyrophenone.
- React the crude isobutyrophenone with carbon tetrachloride and sodium hydroxide using a phase transfer catalyst in a one-pot system to yield the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel process translates into tangible strategic advantages that extend far beyond simple unit cost calculations. The most immediate impact is the drastic simplification of the raw material portfolio, as the process eliminates the need for purchasing and storing highly regulated and volatile commodities such as phosphorus trichloride, aluminum trichloride, and elemental chlorine gas. These substances often command premium pricing due to their hazardous classification and require specialized logistics providers, so removing them from the bill of materials significantly de-risks the supply chain against market fluctuations and transportation disruptions. Furthermore, the shift to commodity acids like benzoic acid and isobutyric acid ensures a stable and abundant supply base, as these chemicals are produced at massive scales globally for various industries, providing a buffer against localized shortages. This stability allows for more accurate long-term forecasting and contract negotiation, empowering procurement teams to secure better terms and ensure uninterrupted production schedules for their downstream customers.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the elimination of expensive waste treatment protocols associated with traditional methods. By avoiding the generation of high-phosphorus wastewater and aluminum-containing sludge, manufacturers can significantly reduce their expenditure on effluent treatment plants and hazardous waste disposal fees, which often constitute a hidden but substantial portion of operating costs. Additionally, the removal of benzene as a solvent eliminates the costs associated with solvent recovery systems and occupational health monitoring programs required for carcinogen handling. The continuous nature of the fixed-bed reactor also implies higher energy efficiency and labor productivity compared to batch processes, further driving down the conversion cost per kilogram of finished product without compromising on quality or purity specifications.
- Enhanced Supply Chain Reliability: From a logistics perspective, the simplified reagent list enhances the resilience of the supply chain by reducing dependency on single-source suppliers of specialty hazardous chemicals. Traditional reagents like phosphorus trichloride are often subject to strict transportation regulations that can delay shipments during adverse weather or regulatory inspections, whereas the carboxylic acids used in this new process are classified as general cargo in many jurisdictions, facilitating smoother and faster inbound logistics. This reliability is crucial for maintaining just-in-time inventory levels and meeting tight delivery windows for global clients. Moreover, the robustness of the solid catalyst system means that production campaigns can run for extended periods without frequent catalyst change-outs, ensuring a consistent output rate that supports reliable fulfillment of large-volume orders.
- Scalability and Environmental Compliance: The architectural design of this process is inherently scalable, making it ideally suited for commercial expansion without the exponential increase in capital expenditure typically seen with batch reactors. The fixed-bed configuration allows for capacity increases simply by adding parallel reactor trains or increasing tube diameter, a linear scaling path that preserves the reaction kinetics and selectivity established at the pilot scale. Environmentally, the process aligns perfectly with increasingly stringent global regulations regarding volatile organic compounds (VOCs) and toxic emissions. By generating significantly less hazardous waste and avoiding the release of corrosive gases, facilities can operate with a smaller environmental footprint, reducing the risk of regulatory fines and enhancing the corporate sustainability profile which is increasingly important for winning contracts with eco-conscious multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this patented synthesis route for Photoinitiator 1173. These insights are derived directly from the experimental data and process descriptions found in the patent documentation, providing a factual basis for evaluating the technology's fit within your existing manufacturing framework. Understanding these nuances is essential for stakeholders who need to balance technical feasibility with commercial viability when considering process upgrades or new supplier qualifications.
Q: How does the new process improve safety compared to traditional methods?
A: The novel process eliminates the use of highly hazardous reagents such as phosphorus trichloride, aluminum trichloride, and chlorine gas, significantly reducing operational risks and the need for specialized containment infrastructure.
Q: What are the environmental benefits of this synthesis route?
A: By avoiding the generation of high-phosphorus wastewater and aluminum-containing waste liquids, the process drastically lowers the burden on three-waste treatment facilities and aligns with stricter environmental compliance standards.
Q: Can this method be scaled for industrial production?
A: Yes, the use of a continuous fixed-bed catalytic reactor for the ketone-making step facilitates easier scale-up and continuous operation compared to batch-wise Friedel-Crafts reactions, enhancing production efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Photoinitiator 1173 Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to greener and more efficient chemical processes is not just a regulatory requirement but a strategic imperative for maintaining competitiveness in the global market. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the one described in patent CN108892605B can be seamlessly translated from the laboratory to full-scale manufacturing. Our state-of-the-art facilities are equipped with the necessary corrosion-resistant reactors and continuous flow units required to handle high-temperature catalytic processes safely, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the exacting standards of the coatings and electronics industries. We are committed to delivering high-purity Photoinitiator 1173 that enables our clients to formulate superior UV-curing products with confidence.
We invite you to engage with our technical procurement team to discuss how this advanced manufacturing route can be tailored to your specific volume requirements and cost targets. By leveraging our expertise, you can access a Customized Cost-Saving Analysis that quantifies the potential operational efficiencies available through this technology. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our commitment to innovation and quality can become a cornerstone of your supply chain strategy.
