Advanced Zinc-Mediated Synthesis of Biphenyl Intermediates for Sartan Drugs
Advanced Zinc-Mediated Synthesis of Biphenyl Intermediates for Sartan Drugs
The pharmaceutical industry continuously seeks robust and economically viable pathways for synthesizing critical scaffolds, particularly biphenyl structures which are ubiquitous in angiotensin II receptor blockers (ARBs) known as Sartans. Patent CN101735106B introduces a transformative preparation method for biphenyl compounds that addresses long-standing inefficiencies in traditional bromination and coupling workflows. This technology leverages a zinc-mediated reductive debromination strategy to not only synthesize target intermediates but also to recover valuable materials from waste streams. By converting over-brominated byproducts and bromomethyl precursors directly into the desired methyl-substituted biphenyls, this process significantly enhances atom economy. The method is particularly relevant for the production of key intermediates such as 2-cyano-4'-methylbiphenyl (OTBN) and various tetrazole derivatives, which serve as the foundational building blocks for major antihypertensive medications. This technical insight explores the mechanistic advantages and commercial implications of adopting this circular chemistry approach.
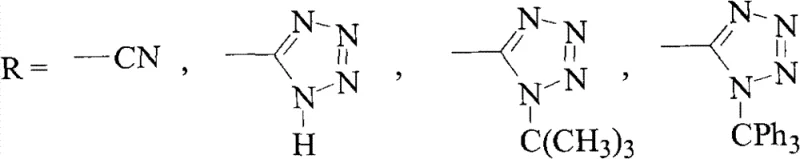
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for biphenyl intermediates often rely on palladium-catalyzed cross-coupling reactions, such as Suzuki or Grignard couplings, which, while effective, impose significant economic and operational burdens. These methods typically require expensive transition metal catalysts, strictly anhydrous conditions, and sophisticated purification steps to remove trace metal residues to meet pharmaceutical standards. Furthermore, the preceding bromination steps required to generate coupling partners frequently suffer from selectivity issues, leading to the formation of dibromo impurities (Formula III) alongside the desired monobromo products (Formula II). In conventional processing, these dibromo byproducts are often treated as waste or require complex, yield-depleting recrystallization processes to separate, resulting in substantial material loss. The disposal of halogenated organic waste also presents environmental compliance challenges, increasing the overall cost of goods sold (COGS) for manufacturers relying on these legacy technologies.
The Novel Approach
The methodology disclosed in the patent data offers a paradigm shift by utilizing a reductive debromination strategy that turns waste into value. Instead of discarding the crystallization mother liquor containing dibromo impurities, this novel approach subjects the crude mixture to zinc powder reduction in an alkaline aqueous-organic solvent system. This single operation effectively strips the excess bromine atoms from the dibromo species (Formula III) and reduces the bromomethyl group in Formula II to a methyl group, yielding the target compound (Formula I) with high purity. The process eliminates the need for precious metal catalysts and operates under mild thermal conditions, typically between 15°C and 30°C. By integrating waste recovery directly into the synthesis loop, the method drastically simplifies the workflow, reduces raw material consumption, and mitigates the environmental impact associated with halogenated waste disposal, representing a significant advancement in green chemistry for pharmaceutical intermediates.
Mechanistic Insights into Zinc-Mediated Reductive Debromination
The core of this technology lies in the electron transfer capability of activated zinc powder in a biphasic system comprising a water-miscible aprotic solvent (such as tetrahydrofuran or dioxane) and aqueous sodium hydroxide. Mechanistically, the zinc surface acts as an electron donor, facilitating the reductive cleavage of the carbon-bromine bonds present in both the benzylic bromide (Formula II) and the gem-dibromide (Formula III) impurities. The presence of the alkaline solution is critical, as it likely assists in activating the zinc surface and neutralizing the hydrogen bromide generated during the reduction, driving the equilibrium towards the formation of the stable methyl group. This reductive environment ensures that even sterically hindered or electronically deactivated bromine atoms are efficiently removed. The reaction proceeds through radical or organozinc intermediates that are rapidly protonated by the solvent system to yield the final hydrocarbon product, avoiding the formation of complex coupling byproducts often seen in metal-catalyzed cycles.
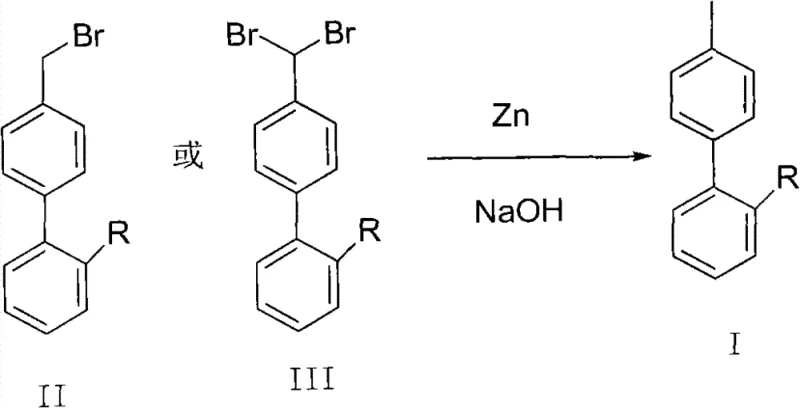
Impurity control is inherently built into this mechanism due to the chemoselectivity of the reduction conditions. While the carbon-bromine bonds are susceptible to zinc-mediated reduction, the aromatic biphenyl core and functional groups such as nitriles or protected tetrazoles remain intact under these specific alkaline conditions. This orthogonality is vital for maintaining the structural integrity of complex Sartan intermediates. Furthermore, because the process can accept crude feeds containing up to 35% dibromo impurities (as noted in the experimental data) and convert them into the desired product, the burden on upstream purification is significantly lowered. The work-up procedure involves simple phase separation followed by concentration, which effectively removes inorganic zinc salts and residual base, resulting in a product profile that requires minimal downstream polishing to achieve high-purity specifications suitable for subsequent drug synthesis steps.
How to Synthesize Biphenyl Compounds Efficiently
The synthesis protocol described in the patent provides a standardized framework for implementing this reductive debromination at scale. The process begins by dissolving the brominated raw material, which can be either pure 4'-bromomethyl-2-cyanobiphenyl or a crude recovered solid from bromination mother liquor, into a solvent system consisting of tetrahydrofuran and 15-20% aqueous sodium hydroxide. The detailed standardized synthesis steps see the guide below.
- Dissolve the brominated raw material (Formula II or crude mother liquor containing Formula III) in a mixture of water-miscible organic solvent (e.g., THF) and aqueous sodium hydroxide.
- Add zinc powder in batches to the reaction mixture while maintaining the temperature between 15°C and 30°C to control the exothermic debromination reaction.
- After completion, allow the mixture to stand for phase separation, concentrate the organic layer, and purify to obtain the target biphenyl compound (Formula I).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this zinc-mediated process translates into tangible strategic benefits centered around cost stability and resource efficiency. By enabling the recycling of bromination mother liquors that were previously considered waste, the method effectively increases the overall yield of the manufacturing campaign without requiring additional starting materials. This circular approach reduces the volume of hazardous waste requiring disposal, thereby lowering environmental compliance costs and simplifying waste management logistics. The reliance on commodity chemicals like zinc powder and sodium hydroxide, rather than volatile precious metals like palladium, insulates the supply chain from the price fluctuations often associated with rare earth and noble metal markets. Additionally, the mild reaction conditions reduce energy consumption for heating and cooling, contributing to a lower carbon footprint and reduced utility costs per kilogram of produced intermediate.
- Cost Reduction in Manufacturing: The elimination of expensive palladium catalysts and ligands removes a significant cost driver from the bill of materials. Furthermore, the ability to convert dibromo impurities back into saleable product maximizes the utility of every gram of raw material purchased, effectively lowering the cost per unit of the active intermediate. The simplified work-up procedure, which avoids complex chromatography or multiple recrystallizations, reduces labor hours and solvent usage, leading to substantial operational expenditure savings.
- Enhanced Supply Chain Reliability: Zinc and sodium hydroxide are globally available commodity chemicals with robust supply chains, minimizing the risk of production stoppages due to reagent shortages. The process tolerance for crude feedstocks means that variations in the quality of upstream bromination outputs can be accommodated without rejecting entire batches, ensuring consistent production throughput. This resilience is critical for maintaining continuous supply to downstream API manufacturers who depend on just-in-time delivery schedules for their own production lines.
- Scalability and Environmental Compliance: The reaction is exothermic but manageable under controlled batch addition of zinc, making it highly scalable from pilot plant to multi-ton commercial production without significant engineering hurdles. The aqueous work-up generates zinc salts that are easier to treat and dispose of compared to heavy metal catalyst residues, facilitating easier adherence to increasingly stringent environmental regulations regarding heavy metal discharge in pharmaceutical manufacturing zones.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this biphenyl synthesis technology. These answers are derived directly from the experimental data and process descriptions found in the underlying patent literature, providing a factual basis for evaluating process feasibility.
Q: How does this method handle over-brominated impurities?
A: The process utilizes zinc-mediated reduction to convert dibromo impurities (Formula III) found in crystallization mother liquors directly back into the desired mono-substituted product (Formula I), effectively recycling waste.
Q: What are the typical reaction conditions for this debromination?
A: The reaction operates under mild conditions, typically between 15°C and 30°C, using zinc powder as the reducing agent in a THF/water/NaOH system, avoiding the need for high pressure or temperature.
Q: Is this process suitable for large-scale manufacturing of Sartan intermediates?
A: Yes, the use of inexpensive reagents like zinc and sodium hydroxide, combined with simple work-up procedures like phase separation and concentration, makes it highly scalable and economically viable for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Biphenyl Compound Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating advanced academic and patent innovations into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this zinc-mediated reduction are fully realized in a GMP-compliant environment. Our rigorous QC labs and stringent purity specifications guarantee that every batch of biphenyl intermediate meets the exacting standards required for Sartan drug synthesis, providing our partners with absolute confidence in material quality and consistency.
We invite global pharmaceutical partners to leverage our technical expertise for their next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how this innovative process can optimize your supply chain and reduce overall manufacturing costs.
