Advanced Suzuki Coupling Technology for High-Purity Biphenyl Pharmaceutical Intermediates
The pharmaceutical industry continuously seeks robust synthetic methodologies that ensure high purity and scalability for complex active pharmaceutical ingredient (API) intermediates. Patent CN1219756C, filed in 2005, presents a significant technological advancement in the preparation of biphenyl compounds, specifically targeting the synthesis of complex structures like Formula I. This patent addresses a critical challenge in palladium-catalyzed cross-coupling reactions: the formation of symmetric biaryl byproducts, known as homocoupling dimers, which are notoriously difficult to separate from the desired cross-coupled product. The invention introduces a novel process utilizing alkali metal formates as additives to suppress these impurities at the source, thereby streamlining the purification workflow. 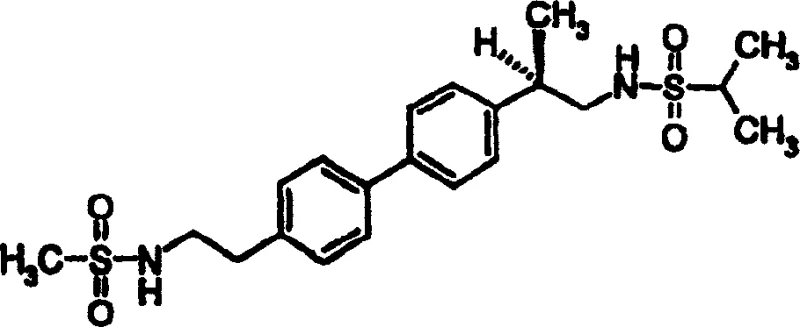 . For R&D directors and procurement specialists, understanding this methodology is crucial as it directly impacts the cost of goods sold (COGS) and the reliability of the supply chain for high-value metabolic drug candidates.
. For R&D directors and procurement specialists, understanding this methodology is crucial as it directly impacts the cost of goods sold (COGS) and the reliability of the supply chain for high-value metabolic drug candidates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional Suzuki-Miyaura coupling reactions, while powerful, often suffer from the competitive formation of homocoupling byproducts when synthesizing unsymmetrical biphenyls. In the context of complex pharmaceutical intermediates, the presence of achiral dimers and chiral dimers poses a severe purification bottleneck. Conventional methods typically rely on extensive chromatographic separation or multiple recrystallization steps to achieve the necessary purity levels, which drastically reduces overall yield and increases solvent consumption. Furthermore, without specific additives, the ratio of these symmetric byproducts can fluctuate based on minor variations in catalyst activity or oxygen levels, leading to inconsistent batch quality. This variability creates significant risks for supply chain heads who require predictable timelines and consistent specifications for commercial scale-up. The difficulty in removing achiral dimers, which often possess solubility profiles similar to the target molecule, means that conventional processes may fail to meet stringent regulatory impurity thresholds without substantial process optimization.
The Novel Approach
The methodology described in CN1219756C fundamentally alters the reaction environment by introducing specific additives, such as potassium formate, into the coupling mixture. This approach effectively suppresses the formation of achiral dimers derived from the self-coupling of the phenylboronic acid derivative. By incorporating approximately 5 equivalents of potassium formate alongside a palladium catalyst like palladium black, the process achieves a marked reduction in impurity generation before purification even begins. 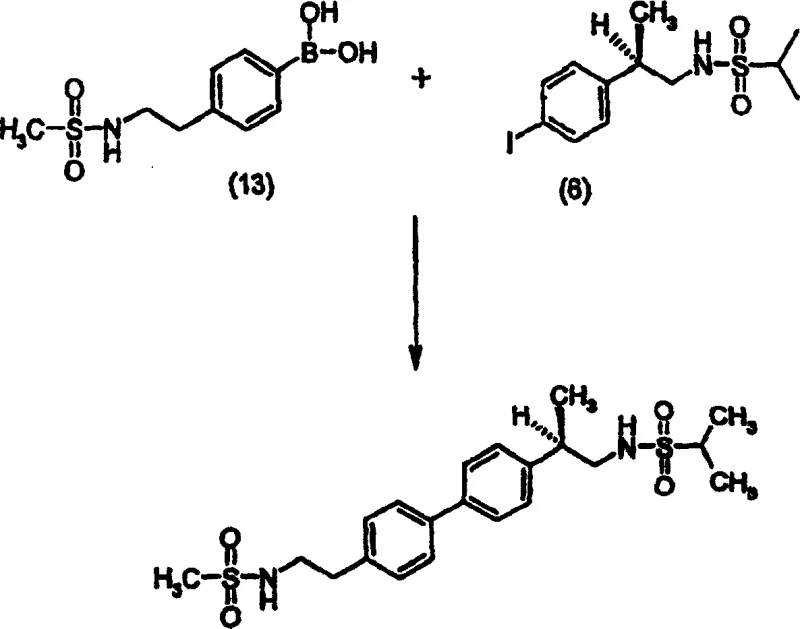 . Experimental data within the patent demonstrates that this additive strategy reduces the achiral dimer content from 0.14% in standard deoxygenated reactions down to 0.07%. This pre-emptive impurity control simplifies the downstream processing requirements, allowing for a more direct path to high-purity material. Additionally, the process utilizes a specific recrystallization regimen involving acetone and water, which not only polishes the chemical purity but also controls the physical properties of the solid, yielding a product with a desirable particle size distribution without the need for mechanical milling.
. Experimental data within the patent demonstrates that this additive strategy reduces the achiral dimer content from 0.14% in standard deoxygenated reactions down to 0.07%. This pre-emptive impurity control simplifies the downstream processing requirements, allowing for a more direct path to high-purity material. Additionally, the process utilizes a specific recrystallization regimen involving acetone and water, which not only polishes the chemical purity but also controls the physical properties of the solid, yielding a product with a desirable particle size distribution without the need for mechanical milling.
Mechanistic Insights into Formate-Assisted Suzuki Coupling
The core of this innovation lies in the interaction between the formate additive and the palladium catalytic cycle. In standard Suzuki couplings, the transmetallation step between the organoboron species and the palladium complex is sensitive to the reaction conditions. The presence of formate ions is believed to modify the electronic environment of the catalyst or act as a mild reducing agent that maintains the active palladium(0) species, thereby favoring the cross-coupling pathway over the homocoupling pathway. The mechanism involves the oxidative addition of the palladium catalyst into the carbon-iodine bond of the halogenated benzene derivative, followed by transmetallation with the phenylboronic acid derivative. The formate additive likely helps to scavenge reactive intermediates that would otherwise lead to the symmetric biaryl byproduct. This mechanistic nuance is critical for R&D teams aiming to replicate the process, as it highlights the importance of maintaining strict stoichiometric control over the additive. The use of potassium formate, prepared in situ from potassium hydroxide and formic acid or added as a commercial salt, ensures a consistent source of formate ions throughout the reaction duration at elevated temperatures around 88°C.
Furthermore, the patent details a sophisticated understanding of impurity solubility dynamics during the purification phase. The distinction between chiral and achiral dimers is vital; while chiral dimers are more soluble in organic solvents and easier to remove, achiral dimers present a greater challenge due to their lower solubility. The invention leverages this difference by employing a binary solvent system of acetone and water for recrystallization. This specific solvent combination creates a solubility gradient where the target biphenyl compound precipitates efficiently while retaining the impurities in the mother liquor. The process is optimized to remove approximately 33% of the achiral dimer and 28% of the chiral dimer in a single crystallization step. This level of control over the solid-state chemistry ensures that the final product meets rigorous purity specifications, typically exceeding 99% purity with minimal residual impurities, which is essential for subsequent pharmaceutical formulation steps.
How to Synthesize {(2R)-2-[4-(4-{2-[(methylsulfonyl)amino]ethyl}phenyl)phenyl]propyl}[(methylethyl)sulfonyl]amine Efficiently
The synthesis of this complex biphenyl intermediate requires precise adherence to the patented protocol to maximize yield and minimize impurity carryover. The process begins with the preparation of the key coupling partners: the iodinated sulfonamide derivative and the functionalized phenylboronic acid. These precursors are combined in a reaction vessel under an inert atmosphere to prevent catalyst deactivation. The addition of the formate additive is the critical differentiator in this workflow, distinguishing it from generic Suzuki protocols. Operators must ensure thorough deoxygenation of the reaction mixture, typically achieved through vacuum-nitrogen cycles, to maintain the integrity of the palladium catalyst. Following the coupling reaction, the workup involves filtration to remove the heterogeneous catalyst and concentration of the organic phase. The final and most crucial step is the controlled recrystallization from acetone and water, which dictates both the chemical purity and the physical morphology of the product. Detailed standardized synthesis steps are provided in the guide below.
- Prepare the reaction mixture by combining the phenylboronic acid derivative (Compound 13) and the halogenated benzene derivative (Compound 6) in a suitable organic solvent such as 1-propanol with an aqueous base.
- Add a critical amount of alkali metal formate additive, specifically potassium formate (approximately 5 equivalents), along with a palladium catalyst like palladium black to the deoxygenated mixture.
- Heat the reaction mixture to approximately 88°C under nitrogen atmosphere for 8 to 16 hours, followed by purification via recrystallization from acetone/water to remove chiral and achiral dimers.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this formate-assisted coupling technology offers tangible strategic benefits beyond simple chemical yield. The primary advantage lies in the drastic simplification of the purification train. By suppressing the formation of difficult-to-remove achiral dimers at the reaction stage, the process eliminates the need for resource-intensive chromatographic separations or multiple iterative recrystallizations. This reduction in processing steps translates directly into lower manufacturing costs, as it decreases solvent consumption, reduces waste disposal volumes, and shortens the overall production cycle time. The ability to achieve high purity through a single, optimized recrystallization step significantly enhances the economic viability of producing this intermediate on a commercial scale. Furthermore, the robustness of the reaction conditions, utilizing common reagents like potassium formate and palladium black, ensures that the supply chain is not dependent on exotic or unstable catalysts that could introduce sourcing risks.
- Cost Reduction in Manufacturing: The implementation of the formate additive strategy leads to substantial cost savings by minimizing material loss during purification. Traditional methods often suffer from significant yield erosion when attempting to purge homocoupling byproducts, whereas this novel approach maintains high recovery rates while achieving superior purity. The elimination of downstream grinding operations, facilitated by the controlled particle size generated during crystallization, further reduces energy consumption and equipment wear. This holistic efficiency gain allows for a more competitive pricing structure for the final API, providing a clear margin advantage for manufacturers adopting this technology.
- Enhanced Supply Chain Reliability: The use of stable, commercially available reagents such as potassium formate and 1-propanol ensures a resilient supply chain that is less susceptible to market fluctuations. Unlike processes relying on sensitive ligands or air-sensitive catalysts that require specialized handling and storage, this method utilizes robust conditions that are easier to manage in a multi-purpose manufacturing facility. The consistency of the impurity profile from batch to batch reduces the risk of failed quality control tests, ensuring reliable delivery schedules to downstream customers. This predictability is invaluable for supply chain heads managing just-in-time inventory for critical drug development programs.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively in multi-gram to kilogram scales within the patent examples. The reduction in solvent usage and the avoidance of complex separation techniques align with green chemistry principles, reducing the environmental footprint of the manufacturing process. The ability to produce the intermediate with a specific particle size distribution (29 to 34 microns) without mechanical milling not only saves energy but also reduces the generation of fine particulate dust, improving workplace safety and environmental compliance. This makes the technology highly attractive for facilities aiming to meet increasingly stringent regulatory and sustainability standards.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this biphenyl synthesis technology. These insights are derived directly from the experimental data and claims presented in patent CN1219756C, providing a factual basis for evaluating the process feasibility. Understanding these details is essential for technical teams assessing the transferability of this method to their own production environments. The answers highlight the specific advantages of the formate additive system and the recrystallization protocol.
Q: How does the addition of potassium formate improve the purity of the biphenyl product?
A: The addition of potassium formate acts as a specialized additive that significantly suppresses the formation of achiral dimers, which are symmetric biaryl byproducts formed via homocoupling of the boronic acid derivative. Patent data indicates this reduces achiral dimer content from 0.14% to 0.07% prior to recrystallization.
Q: What is the preferred method for removing chiral and achiral dimer impurities?
A: The patent specifies a recrystallization process using a solvent system of acetone and water. This specific solvent combination effectively removes approximately 33% of achiral dimers and 28% of chiral dimers while simultaneously controlling particle size to between 29 and 34 microns.
Q: Why is particle size control important for this intermediate?
A: Controlling the particle size to the 29-34 micron range during crystallization eliminates the need for downstream grinding or milling processes. This facilitates easier formulation and enhances oral absorption characteristics for the final pharmaceutical product.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable {(2R)-2-[4-(4-{2-[(methylsulfonyl)amino]ethyl}phenyl)phenyl]propyl}[(methylethyl)sulfonyl]amine Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom pharmaceutical manufacturing, possessing the technical expertise to translate complex patent methodologies like CN1219756C into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the nuances of the formate-assisted Suzuki coupling are perfectly preserved during scale-up. We understand that maintaining stringent purity specifications and rigorous QC labs are non-negotiable requirements for API intermediates, and our infrastructure is designed to meet these demands with precision. By leveraging our advanced process chemistry capabilities, we can deliver this high-value biphenyl compound with the consistent quality and particle size characteristics required for your formulation needs.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can optimize your supply chain. Contact us today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. Our experts are ready to provide specific COA data and route feasibility assessments to demonstrate how partnering with NINGBO INNO PHARMCHEM can accelerate your project timelines and reduce overall manufacturing costs.
