Revolutionizing Cinacalcet HCl Production: A Scalable 2-Step Synthetic Route via Novel Intermediates
Revolutionizing Cinacalcet HCl Production: A Scalable 2-Step Synthetic Route via Novel Intermediates
The pharmaceutical landscape for calcimimetics has been significantly advanced by the disclosure in patent CN109535006B, which introduces a groundbreaking intermediate and method for preparing Cinacalcet Hydrochloride. As the first drug in its class to activate calcium receptors in the parathyroid gland, Cinacalcet is critical for managing secondary hyperparathyroidism, yet its manufacturing has historically been plagued by complex, multi-step syntheses involving hazardous reagents. This new intellectual property addresses these critical bottlenecks by proposing a concise, two-step pathway that begins with readily available aldehydes and amines. The core innovation lies in the identification of a specific halo-amine intermediate, structurally defined as (R)-3-bromo-N-(1-(naphthalen-1-yl)ethyl)propan-1-amine, which serves as a robust precursor for the final carbon-carbon bond formation. By shifting the synthetic logic from traditional amide reductions to a Lewis acid-catalyzed alkylation, this technology offers a transformative approach to producing high-purity active pharmaceutical ingredients (APIs) with enhanced safety profiles.
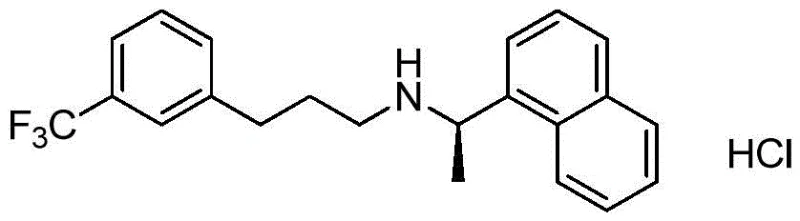
For R&D directors and process chemists, the structural elegance of this new route represents a paradigm shift away from the cumbersome methodologies of the past. The patent explicitly details how this intermediate facilitates a direct connection between the naphthyl-ethyl amine fragment and the trifluoromethylphenyl ring, bypassing the need for protecting groups or harsh reduction conditions typically required in legacy processes. This not only streamlines the workflow but also inherently reduces the generation of difficult-to-remove impurities, thereby simplifying downstream purification. As we delve deeper into the technical specifics, it becomes evident that this method is not merely an incremental improvement but a fundamental re-engineering of the supply chain for this vital therapeutic agent, promising substantial benefits in both cost efficiency and operational safety for global manufacturers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Cinacalcet Hydrochloride has relied on several distinct pathways, each carrying significant drawbacks that hinder efficient large-scale production. One prevalent method involves a reductive amination reaction between m-trifluoromethyl phenylpropyl aldehyde and R-1-(1-naphthyl) ethylamine, utilizing titanium tetraisopropoxide as a catalyst and sodium cyanoborohydride as the reducing agent. While chemically feasible, this route suffers from the formation of high levels of reducing impurities, which compromises the long-term stability of the final drug product. Furthermore, the reliance on titanium tetraisopropoxide introduces high material costs, and the use of sodium cyanoborohydride poses severe toxicity risks, necessitating specialized handling and waste disposal protocols that inflate operational expenditures.
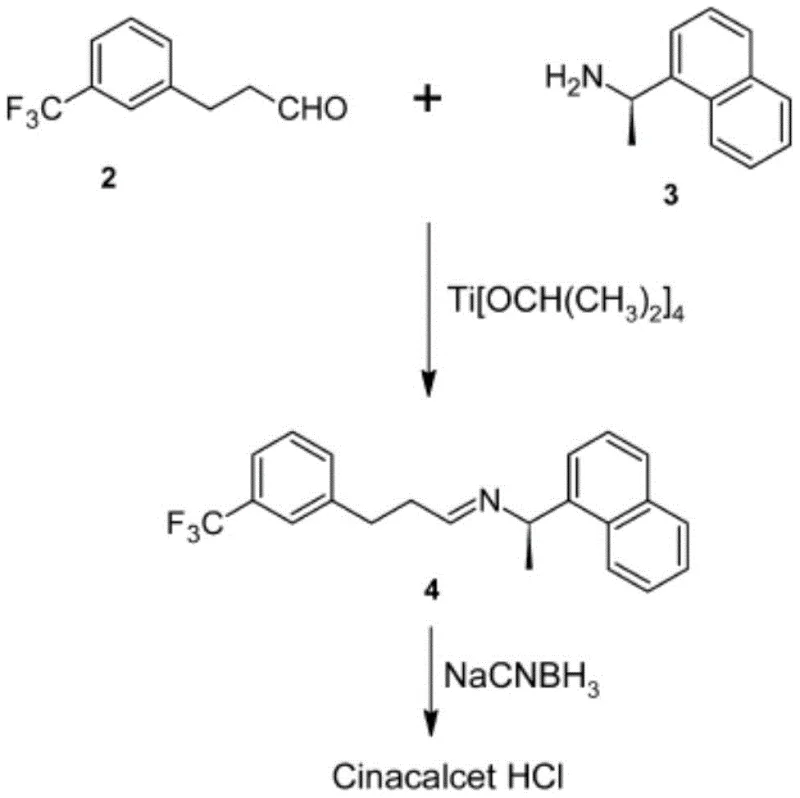
Alternative strategies have attempted to circumvent these issues through amide reduction pathways or Heck coupling reactions, but these too are fraught with challenges. For instance, routes involving the reduction of amide intermediates often require aggressive reducing agents such as lithium aluminum hydride, borane-THF complexes, or Red-Al. These reagents are notoriously unstable, pyrophoric, or highly toxic, creating unsafe working environments and complicating the scale-up process. Additionally, methods employing Heck coupling to construct the carbon skeleton often involve carcinogenic and unstable starting materials like ethyl acrylate, resulting in excessively long reaction sequences that are economically unviable for commercial manufacturing. The cumulative effect of these limitations is a supply chain vulnerable to regulatory scrutiny, safety incidents, and inconsistent product quality.
The Novel Approach
In stark contrast to these convoluted legacy methods, the technology disclosed in CN109535006B presents a streamlined, two-step synthesis that dramatically simplifies the manufacturing landscape. The novel approach centers on the preparation of a specific intermediate, (R)-3-bromo-N-(1-(naphthalen-1-yl)ethyl)propan-1-amine, via a mild reductive amination of 3-halopropionaldehyde and (R)-1-naphthylethylamine. This intermediate is then subjected to a Friedel-Crafts alkylation with benzotrifluoride in the presence of anhydrous aluminum trichloride. This strategic pivot eliminates the need for expensive titanium catalysts and toxic cyanoborohydrides in the critical coupling step, replacing them with more manageable Lewis acid chemistry. The result is a process that operates under mild conditions, typically between 0°C and 10°C, ensuring high selectivity and minimizing side reactions.
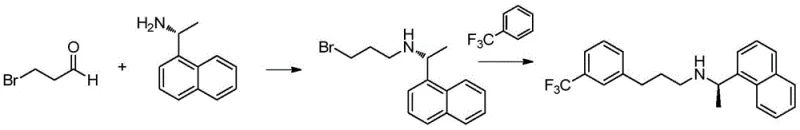
The superiority of this new route is further evidenced by its exceptional yield and purity metrics reported in the patent examples, where final product purity reaches up to 99.9% with yields exceeding 95%. By decoupling the construction of the amine backbone from the introduction of the trifluoromethylphenyl group, the process allows for better control over impurity profiles at each stage. The use of 3-halopropionaldehyde as a starting material is particularly advantageous due to its commercial availability and low cost compared to the complex aldehydes required in prior art. This method effectively resolves the stability issues associated with reducing impurities and offers a robust platform for the consistent production of high-quality Cinacalcet Hydrochloride, making it an ideal candidate for modern, compliant pharmaceutical manufacturing facilities.
Mechanistic Insights into Friedel-Crafts Alkylation and Reductive Amination
The chemical elegance of this new synthesis lies in the precise orchestration of two fundamental organic transformations: reductive amination and electrophilic aromatic substitution. The first step involves the condensation of 3-halopropionaldehyde with (R)-1-naphthylethylamine to form an imine intermediate, which is subsequently reduced by sodium cyanoborohydride. Crucially, the patent specifies a controlled addition of the reducing agent in three portions with a specific molar ratio (2:2:1), a technique designed to manage the exotherm and prevent over-reduction or side reactions. This careful stoichiometry ensures that the chiral center at the benzylic position remains intact, preserving the optical purity essential for the biological activity of the final API. The resulting halo-amine intermediate possesses a reactive alkyl halide tail, primed for nucleophilic attack or, in this specific case, serving as the electrophile in the subsequent step.
The second and most critical mechanistic step is the Lewis acid-catalyzed alkylation of benzotrifluoride. Here, anhydrous aluminum trichloride acts as a potent Lewis acid, coordinating with the halogen atom of the intermediate to generate a highly reactive carbocation or a tight ion pair. This electrophilic species then attacks the electron-rich aromatic ring of benzotrifluoride. The regioselectivity of this substitution is governed by the electronic effects of the trifluoromethyl group, which directs the incoming alkyl chain to the meta-position, perfectly aligning with the target structure of Cinacalcet. The reaction is conducted at low temperatures (0-5°C) to suppress poly-alkylation and rearrangement of the carbocation, which are common pitfalls in Friedel-Crafts chemistry. Following the coupling, the reaction mixture is quenched carefully to hydrolyze the aluminum complexes, and the free base is converted to the hydrochloride salt, yielding the final stable crystalline form of the drug.
How to Synthesize Cinacalcet Hydrochloride Efficiently
Implementing this novel synthetic route requires strict adherence to the optimized conditions outlined in the patent to maximize yield and safety. The process begins with the preparation of the key intermediate in anhydrous methanol, utilizing an ice-water bath to maintain thermal control during the exothermic reduction. Once the intermediate is isolated, it is dissolved in dichloromethane and added dropwise to a suspension of anhydrous aluminum trichloride, ensuring that the concentration of the reactive electrophile remains low to favor mono-alkylation. The detailed standardized synthesis steps, including specific solvent volumes, stirring times, and workup procedures, are critical for reproducibility and are summarized in the guide below for technical teams evaluating this methodology.
- Prepare the key intermediate (R)-3-bromo-N-(1-(naphthalen-1-yl)ethyl)propan-1-amine by reacting 3-bromopropanal with (R)-1-naphthylethylamine using sodium cyanoborohydride reduction in methanol.
- Perform a Friedel-Crafts alkylation by reacting the intermediate with benzotrifluoride in the presence of anhydrous aluminum trichloride at controlled low temperatures (0-10°C).
- Quench the reaction, isolate the free base, and convert it to the hydrochloride salt using concentrated hydrochloric acid to obtain high-purity Cinacalcet HCl.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers compelling economic and logistical advantages that directly impact the bottom line. By transitioning away from the legacy methods that rely on exotic catalysts and hazardous reductants, manufacturers can achieve significant cost reductions in raw material procurement and waste management. The elimination of titanium tetraisopropoxide and sodium cyanoborohydride from the final coupling step removes the need for expensive specialty chemicals and the rigorous safety infrastructure required to handle them. Furthermore, the shortened reaction sequence reduces the overall processing time and energy consumption, leading to a more lean and efficient manufacturing operation that can respond rapidly to market demand fluctuations.
- Cost Reduction in Manufacturing: The new process leverages commodity chemicals such as 3-bromopropanal and benzotrifluoride, which are widely available and priced significantly lower than the complex precursors used in traditional Heck coupling or amide reduction routes. By avoiding the use of stoichiometric amounts of expensive metal catalysts and pyrophoric reducing agents like lithium aluminum hydride, the direct material cost per kilogram of API is drastically lowered. Additionally, the simplified workup procedure reduces solvent usage and disposal costs, contributing to a more sustainable and economically viable production model that enhances profit margins without compromising quality.
- Enhanced Supply Chain Reliability: Dependence on a limited number of suppliers for specialized reagents often creates bottlenecks in the pharmaceutical supply chain. This novel method mitigates such risks by utilizing robust, commercially abundant starting materials that are sourced from a broad global supplier base. The stability of the intermediate and the mild reaction conditions also reduce the likelihood of batch failures due to reagent degradation or handling errors. This reliability ensures a consistent flow of high-quality intermediates and final API, safeguarding against production delays and enabling manufacturers to maintain steady inventory levels to meet contractual obligations with downstream partners.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental challenges, particularly when dealing with toxic or unstable reagents. The proposed route is inherently safer, operating at near-ambient temperatures and avoiding the generation of heavy metal waste or toxic gas byproducts associated with borane or cyanoborohydride treatments. This alignment with green chemistry principles simplifies regulatory compliance and environmental permitting, facilitating smoother technology transfer from pilot plant to commercial scale. The ability to run this process in standard stainless steel reactors without specialized lining or containment systems further lowers the barrier to entry for contract manufacturing organizations looking to expand their Cinacalcet production capacity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this technology outperforms existing standards in terms of safety, purity, and efficiency. Understanding these nuances is essential for technical teams assessing the feasibility of adopting this route for their own manufacturing portfolios.
Q: What are the primary advantages of this new intermediate route over traditional amide reduction methods?
A: Unlike traditional methods that require hazardous reducing agents like lithium aluminum hydride or borane complexes, this novel route utilizes a milder Friedel-Crafts alkylation strategy. This eliminates the safety risks associated with pyrophoric reagents and simplifies the purification process, leading to significantly higher product stability and purity.
Q: How does this method ensure the preservation of chiral integrity during synthesis?
A: The process initiates with enantiomerically pure (R)-1-naphthylethylamine. The subsequent reductive amination and electrophilic aromatic substitution steps are designed to proceed under mild conditions that do not racemize the chiral center, ensuring the final API meets stringent stereochemical specifications without the need for complex resolution steps.
Q: Is this synthetic route suitable for large-scale industrial manufacturing?
A: Yes, the route is highly scalable. It avoids the use of expensive titanium catalysts and toxic cyanoborohydrides in the final coupling step. The reaction conditions are moderate (0-10°C), and the raw materials, such as 3-halopropionaldehyde and benzotrifluoride, are commercially abundant, making it ideal for cost-effective mass production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cinacalcet Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic route described in CN109535006B and are fully equipped to leverage this technology for our global partners. As a premier CDMO specializing in complex pharmaceutical intermediates, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are designed to handle the specific requirements of Lewis acid catalysis and sensitive amine chemistry, ensuring that every batch meets stringent purity specifications. With our rigorous QC labs and commitment to process optimization, we guarantee the delivery of high-purity Cinacalcet intermediates and API that adhere to the highest international regulatory standards.
We invite forward-thinking pharmaceutical companies to collaborate with us to capitalize on the efficiencies of this novel manufacturing process. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and supply chain needs. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise can drive down your costs and secure your supply of this critical calcimimetic agent.
