Scalable Zolpidem Production: Advanced Catalytic Reduction for Commercial Supply
Introduction to Advanced Zolpidem Manufacturing Technologies
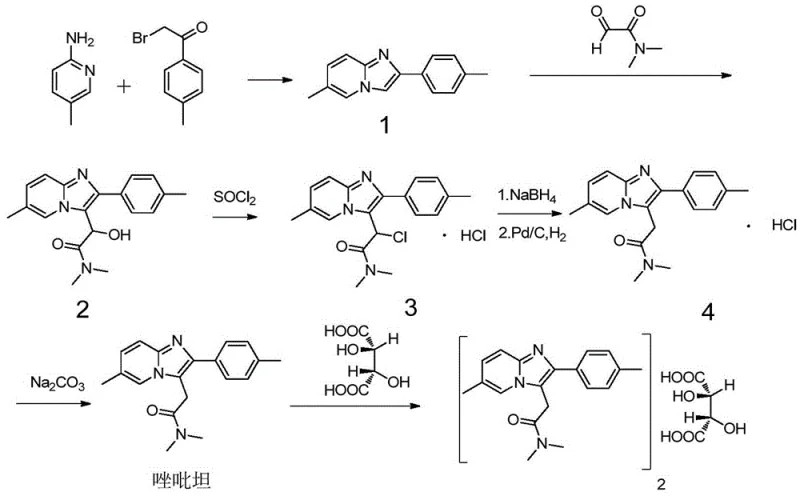
The pharmaceutical industry continuously seeks robust, scalable, and safe methodologies for the production of critical active pharmaceutical ingredients (APIs) and their intermediates. Patent CN116003407A represents a significant technological leap in the large-scale synthesis of Zolpidem, a widely prescribed non-benzodiazepine sedative-hypnotic agent. This innovation addresses longstanding challenges in the chemical synthesis of imidazopyridine derivatives by introducing a novel composite catalytic system. Unlike traditional routes that rely on hazardous hydrogenation or inefficient stoichiometric reductions, this patented approach utilizes a synergistic combination of Phosphorus Tribromide ($PBr_3$) and Lewis Acids. The technical breakthrough lies in its ability to achieve commercial-grade purity exceeding 99.5% while maintaining high yields and operational safety. For global procurement and R&D teams, this development signals a shift towards more sustainable and cost-effective manufacturing paradigms for high-purity pharmaceutical intermediates.
The significance of this patent extends beyond mere academic interest; it offers a tangible solution for supply chain resilience. By optimizing reaction conditions and catalyst loading, the method ensures consistent quality output suitable for stringent regulatory environments. The process avoids the use of precious metal catalysts like Palladium on Carbon, which are subject to volatile market pricing and supply constraints. Furthermore, the elimination of high-pressure hydrogenation steps reduces the capital expenditure required for specialized reactor infrastructure. As a reliable zolpidem intermediate supplier, understanding these underlying technological shifts is crucial for forecasting production capabilities and ensuring continuity of supply for downstream drug manufacturers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the commercial synthesis of Zolpidem has been plagued by inefficiencies and safety concerns inherent in older synthetic routes. For instance, the method disclosed in US4794185A involves multiple reaction steps starting from 5-methyl-2-pyridinamine, leading to a prolonged production cycle and cumulative yield losses. A critical bottleneck in such traditional pathways is the reduction step, often employing Sodium Borohydride ($NaBH_4$) in acidic media. This reaction generates hydrogen gas rapidly, posing significant explosion risks and requiring specialized venting and safety protocols that increase operational costs. Additionally, alternative routes utilizing Palladium-Carbon (Pd/C) catalyzed hydrogenation, while effective, introduce the risk associated with one of the eighteen dangerous controlled reactions recognized by safety authorities. The reliance on expensive noble metals also creates a vulnerability in cost reduction in pharmaceutical intermediates manufacturing, as fluctuations in palladium prices directly impact the final cost of goods.
The Novel Approach
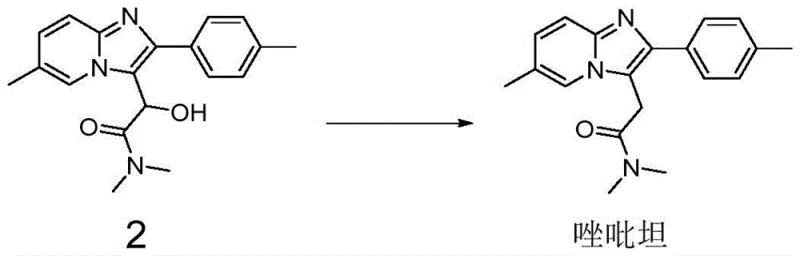
In stark contrast, the methodology outlined in CN116003407A streamlines the synthesis by focusing on the efficient reduction of the key hydroxy-amide precursor (Compound 2) directly to Zolpidem. This approach bypasses the need for high-pressure equipment and precious metal catalysts entirely. The core innovation is the use of a composite catalyst system comprising Phosphorus Tribromide and a Lewis Acid, such as Aluminum Chloride ($AlCl_3$), Ferric Chloride ($FeCl_3$), or Zinc Chloride ($ZnCl_2$). This dual-catalyst strategy activates the hydroxyl group for substitution more effectively than $PBr_3$ alone, which historically suffered from low conversion rates and difficult separation of unreacted starting materials. The result is a robust process that operates under mild conditions, typically involving a controlled dropwise addition at temperatures between -20°C and 0°C, followed by reflux. This simplification not only enhances safety but also drastically shortens the production cycle, making it highly suitable for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into FeCl3-Catalyzed Cyclization and Reduction
The efficacy of this novel synthesis route can be attributed to the sophisticated interplay between the Phosphorus Tribromide and the Lewis Acid co-catalyst. Mechanistically, the Lewis Acid coordinates with the oxygen atom of the hydroxyl group in Compound 2, increasing its electrophilicity and facilitating the nucleophilic attack by the bromide ion generated from $PBr_3$. This activation lowers the energy barrier for the substitution reaction, ensuring that the conversion proceeds to completion even at relatively mild thermal conditions. The optimization of the molar ratio between Compound 2 and the Lewis Acid, specifically within the range of 1:0.4 to 1:1.2, is critical for maximizing this catalytic effect without generating excessive acidic byproducts that could degrade the sensitive imidazopyridine core. Furthermore, the subsequent reflux step ensures that any intermediate bromo-species are fully reduced to the final methylene bridge found in Zolpidem, locking in the structural integrity of the molecule.
Impurity control is another paramount aspect of this mechanism, particularly for R&D directors focused on purity and impurity profiles. The data indicates that the composite catalyst system significantly suppresses the formation of unknown impurities (labeled as Impurities 1-4 in the patent data) compared to using $PBr_3$ alone. When $PBr_3$ is used in isolation, incomplete conversion often leads to a complex mixture of starting material and partially reduced species that are difficult to separate. However, the presence of the Lewis Acid promotes a cleaner reaction pathway, resulting in a crude product with Zolpidem content often exceeding 99.5%. This high level of chemical fidelity minimizes the burden on downstream purification processes, such as recrystallization or chromatography, thereby preserving overall yield and reducing solvent consumption. The ability to consistently achieve such high purity standards is essential for meeting the rigorous specifications required by global regulatory bodies for API intermediates.
How to Synthesize Zolpidem Efficiently
The practical implementation of this synthesis route involves a carefully sequenced addition of reagents to maintain thermal control and reaction homogeneity. The process begins with the dissolution of the precursor in a suitable organic solvent, followed by the introduction of the Lewis Acid catalyst under cooling. The critical step involves the slow, dropwise addition of the Phosphorus Tribromide solution, which must be managed precisely to prevent exothermic runaway. Detailed standardized operating procedures regarding stirring rates, addition times, and quenching protocols are essential for reproducibility. For a comprehensive guide on the exact operational parameters, please refer to the technical documentation below.
- Dissolve the hydroxy-amide precursor (Compound 2) in an organic solvent such as tetrahydrofuran and add a Lewis acid catalyst like aluminum chloride.
- Cool the reaction mixture to between -20°C and 0°C, then slowly add a solution of phosphorus tribromide while maintaining strict temperature control.
- Heat the mixture to reflux to complete the reduction, then quench with sodium carbonate solution and purify via extraction and drying to obtain Zolpidem.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this catalytic system offers substantial advantages in terms of cost stability and supply chain reliability. The most immediate benefit is the elimination of Palladium on Carbon, a catalyst that not only carries a high price tag but also requires complex recovery and recycling processes to meet environmental standards. By replacing this with inexpensive and readily available Lewis Acids like Aluminum Chloride or Ferric Chloride, manufacturers can achieve significant cost savings in raw material procurement. Furthermore, the avoidance of high-pressure hydrogenation removes the need for specialized autoclaves and the associated safety inspections, lowering the barrier to entry for contract manufacturing organizations (CMOs) and reducing the overall capital intensity of the production line. This translates to a more flexible supply base capable of responding quickly to market demand fluctuations.
- Cost Reduction in Manufacturing: The shift away from precious metal catalysts and high-pressure equipment fundamentally alters the cost structure of Zolpidem production. Without the need for expensive Pd/C or the infrastructure to handle hydrogen gas safely, the operational expenditure (OpEx) is drastically simplified. The use of commodity chemicals like $PBr_3$ and common Lewis acids ensures that raw material costs remain stable and predictable, shielding the supply chain from the volatility often seen in the precious metals market. Additionally, the high conversion rates reduce the volume of waste generated per kilogram of product, leading to lower waste disposal costs and improved overall process economics.
- Enhanced Supply Chain Reliability: Dependence on a single source for specialized catalysts or dangerous gases can create bottlenecks in the supply chain. This new method utilizes reagents that are widely produced and available from multiple global suppliers, thereby diversifying the supply risk. The robustness of the reaction, demonstrated by its success in 1000L reactors, indicates that the process is not fragile and can withstand the variances inherent in large-scale industrial operations. This reliability is crucial for reducing lead time for high-purity pharmaceutical intermediates, ensuring that downstream API manufacturers receive their materials on schedule without unexpected delays caused by catalyst shortages or safety shutdowns.
- Scalability and Environmental Compliance: The mild reaction conditions and the absence of heavy metal residues simplify the environmental compliance landscape. Traditional methods involving heavy metals often require extensive wastewater treatment to remove trace catalyst residues to ppm levels. In contrast, the Lewis Acid/$PBr_3$ system generates byproducts that are easier to manage and neutralize, such as phosphoric acid derivatives and metal salts, which can be treated with standard effluent protocols. This environmental friendliness facilitates smoother regulatory approvals and supports the growing industry demand for green chemistry practices, making the commercial scale-up of complex pharmaceutical intermediates more sustainable in the long term.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear picture of what partners can expect when adopting this methodology. Understanding these nuances helps in aligning technical expectations with commercial realities.
Q: What are the safety advantages of this new Zolpidem synthesis method?
A: This method eliminates the need for high-pressure hydrogenation reactions and expensive palladium catalysts, significantly reducing operational risks and explosion hazards associated with traditional reduction pathways.
Q: How does the composite catalyst improve product purity?
A: The synergistic effect of Phosphorus Tribromide and Lewis Acids ensures high conversion rates and minimizes side reactions, consistently achieving purity levels above 99.5% without complex purification steps.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent demonstrates successful scaling in 1000L reactors with high yields (over 84%), proving its viability for commercial manufacturing and reliable supply chain integration.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Zolpidem Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in patent examples can be replicated with consistency and precision on an industrial scale. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to verify that every batch of Zolpidem intermediate meets the highest global standards. Our commitment to quality assurance means that we do not just supply chemicals; we deliver confidence in your supply chain.
We invite procurement leaders and technical directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific production volumes and quality requirements. By leveraging our optimized catalytic processes, we can help you navigate the complexities of modern pharmaceutical manufacturing while achieving your cost and efficiency targets. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us collaborate to build a more resilient and efficient supply chain for your critical therapeutic programs.
