Optimizing Tenofovir Alafenamide Intermediate Production via Novel Halogenation Strategies
The global demand for effective antiviral therapies, particularly for the treatment of chronic Hepatitis B, has placed Tenofovir Alafenamide (TAF) at the forefront of pharmaceutical innovation. As the prodrug of tenofovir, TAF offers improved plasma stability and targeted delivery to lymphoid tissues, necessitating highly efficient and pure synthetic routes for its key intermediates. Patent CN111484528A introduces a significant advancement in this domain, detailing a robust preparation method for the critical PMPA monophenyl ester intermediate. This technology addresses long-standing challenges in the industry by optimizing reaction conditions to enhance yield and purity while minimizing the use of hazardous reagents. For R&D directors and procurement specialists, understanding the nuances of this halogenation-based pathway is essential for securing a reliable supply chain for next-generation antiviral medications.
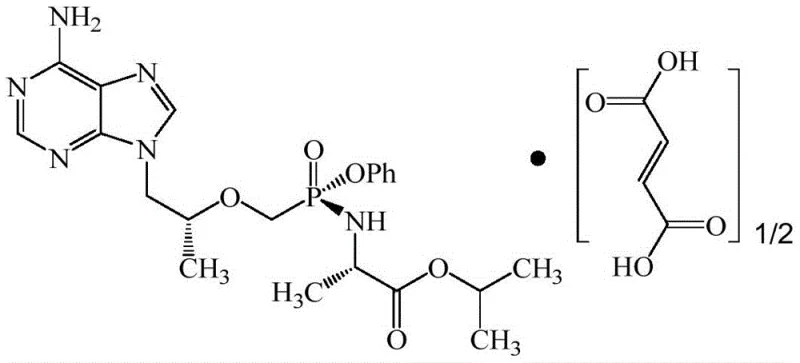
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of TAF intermediates has been plagued by inefficiencies that hinder large-scale commercial viability. Traditional routes often rely on the use of triphenyl phosphite to construct the phenolic ester bond, a reagent that is not only prohibitively expensive but also poses significant safety hazards due to its toxicity and pungent odor. Furthermore, alternative one-pot methods attempting to combine chlorination and amidation steps have struggled with stereochemical control, frequently generating up to fifty percent racemization products which drastically lowers the overall preparation efficiency. Other existing patents require complex water removal steps and the addition of excess alkali, leading to increased reagent consumption, higher waste generation, and the formation of difficult-to-remove impurities that compromise the final product quality.
The Novel Approach
In stark contrast, the methodology disclosed in CN111484528A employs a streamlined two-step strategy that bypasses these historical bottlenecks. By utilizing a controlled halogenation of PMPA to form an activated intermediate (Compound 1), followed by a precise reaction with phenol, the process achieves high conversion rates without the need for expensive condensing agents. This approach allows for the use of cost-effective solvents like toluene and robust halogenating agents such as thionyl chloride. The result is a synthesis pathway that is not only chemically superior in terms of atom utilization but also operationally simpler, making it ideally suited for the rigorous demands of industrial pharmaceutical manufacturing where consistency and safety are paramount.
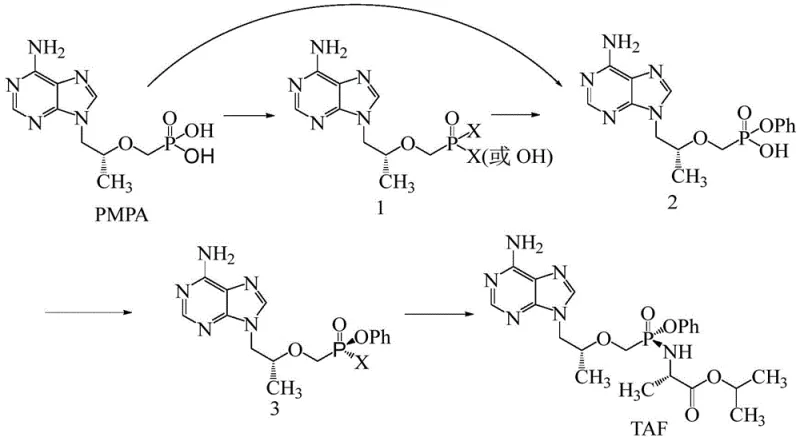
Mechanistic Insights into Halogenation-Activated Phosphonate Esterification
The core of this technological breakthrough lies in the activation of the phosphonic acid moiety of PMPA through halogenation. In the first stage, PMPA reacts with a halogenating reagent, preferably thionyl chloride, in a solvent system such as toluene or acetonitrile. This reaction converts the hydrophilic phosphonic acid groups into a more reactive phosphonic dichloride or mixed anhydride species (Compound 1). The choice of solvent is critical; aromatic solvents like toluene facilitate the reaction at elevated temperatures (70-120°C), ensuring complete conversion of the starting material as monitored by HPLC. This activation step is fundamental because it transforms a relatively stable acid into a highly electrophilic species ready for nucleophilic attack.
Following the formation of Compound 1, the second stage involves the nucleophilic substitution with phenol to establish the P-O-Ph bond. This step is conducted in a second solvent system, such as chloroform or isopropyl acetate, under mild heating. The mechanism ensures that the chiral center adjacent to the purine ring remains intact, avoiding the racemization issues seen in harsher direct methods. The subsequent workup involves a sophisticated pH swing—adjusting to alkaline conditions to remove unreacted phenol and acidic byproducts, followed by acidification to precipitate the pure product. This precise control over the reaction environment minimizes the formation of di-esters or other phosphorus-containing impurities, resulting in a final intermediate with purity levels exceeding 98 percent.
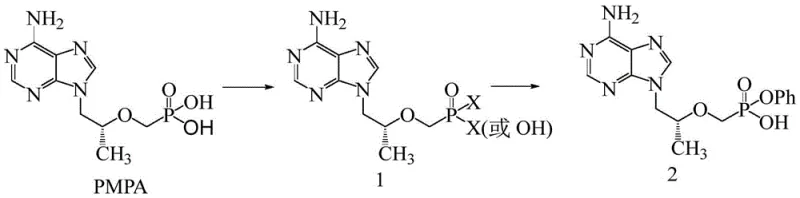
How to Synthesize Tenofovir Alafenamide Intermediate Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for producing high-quality intermediates suitable for downstream coupling with alanine esters. The process begins with the suspension of PMPA in a suitable organic solvent, followed by the controlled addition of the halogenating agent. Critical to success is the thermal management during the exothermic halogenation phase and the rigorous monitoring of reaction completion before proceeding to the phenol coupling step. The detailed standardized synthesis steps, including specific molar ratios and temperature profiles for optimal yield, are provided in the technical guide below.
- React PMPA with a halogenating agent like thionyl chloride in a solvent such as toluene or acetonitrile at elevated temperatures to form the activated Compound 1.
- Monitor the reaction via HPLC to ensure complete conversion of the starting material before removing the solvent.
- React the crude Compound 1 with phenol in a second solvent system (e.g., chloroform or isopropyl acetate) to yield the final phenyl ester intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the shift to this novel halogenation-based route represents a strategic opportunity to optimize both cost structures and supply reliability. By eliminating the dependency on triphenyl phosphite, manufacturers can significantly reduce raw material expenditures and mitigate the risks associated with sourcing hazardous chemicals. The simplified workflow, which removes the need for complex water removal protocols and excessive alkali treatments, translates directly into reduced processing time and lower utility consumption per kilogram of product. These operational efficiencies contribute to a more resilient supply chain capable of meeting the fluctuating demands of the global antiviral market without compromising on quality or delivery timelines.
- Cost Reduction in Manufacturing: The replacement of expensive reagents with commodity chemicals like thionyl chloride and toluene drives down the direct material cost substantially. Additionally, the high atom economy of the reaction reduces the volume of waste solvent and byproducts that require disposal, leading to significant savings in waste management and environmental compliance costs. The ability to isolate the intermediate via simple filtration and pH adjustment further lowers the energy intensity of the purification process compared to traditional chromatographic methods.
- Enhanced Supply Chain Reliability: Utilizing widely available industrial solvents and reagents ensures that production is not bottlenecked by the scarcity of specialized fine chemicals. The robustness of the reaction conditions allows for flexible manufacturing schedules, as the process is less sensitive to minor variations in ambient conditions. This stability is crucial for maintaining continuous production runs, thereby reducing lead times for high-purity pharmaceutical intermediates and ensuring consistent availability for downstream API synthesis.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively from gram scale to multi-kilogram batches in the patent examples. The absence of heavy metal catalysts and the use of recyclable solvents align with modern green chemistry principles, facilitating easier regulatory approval and reducing the environmental footprint of the manufacturing facility. This compliance advantage is increasingly vital for suppliers aiming to partner with top-tier pharmaceutical companies that enforce strict sustainability standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on the practical benefits of adopting this methodology for commercial production.
Q: What are the primary advantages of the halogenation route over triphenyl phosphite methods?
A: The halogenation route eliminates the need for expensive and toxic triphenyl phosphite, significantly reducing raw material costs and hazardous waste generation while simplifying the purification process.
Q: How does this method address chirality concerns in TAF synthesis?
A: By avoiding harsh racemization conditions found in some one-pot methods, this stepwise approach preserves the chiral integrity of the PMPA backbone, ensuring high optical purity essential for antiviral efficacy.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the use of common industrial solvents like toluene and robust reagents like thionyl chloride, combined with simple workup procedures like filtration and pH adjustment, makes it highly scalable for commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tenofovir Alafenamide Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving antiviral therapies. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to full-scale manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of Tenofovir Alafenamide intermediate adheres to the highest international standards for pharmaceutical applications.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your production volume. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project requirements, ensuring a partnership built on transparency, quality, and mutual success.
