Optimizing Methoxamine Hydrochloride Production: A Safer, Scalable Synthetic Route for Global API Manufacturers
The pharmaceutical industry continuously seeks robust synthetic pathways that balance efficiency with stringent safety standards, particularly for cardiovascular agents like Methoxamine Hydrochloride. Patent CN102976961A introduces a transformative methodology that redefines the production landscape for this critical alpha-receptor stimulant. By shifting away from traditional, hazard-prone oximation chemistry toward a controlled bromination and amination sequence, this technology offers a compelling solution for manufacturers aiming to modernize their API intermediate supply chains. The core innovation lies in the strategic use of di-tert-butyl iminodicarboxylate to install the amine functionality, bypassing the generation of toxic methyl nitrite gas entirely. This report analyzes the technical merits and commercial implications of adopting this refined synthetic route, providing actionable insights for R&D directors and procurement strategists alike.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Methoxamine Hydrochloride has relied heavily on a pathway originating from resorcinol, involving methylation, acylation, and a problematic oximation step. As illustrated in the traditional reaction scheme below, this legacy process necessitates the use of dimethyl sulfate, a potent carcinogen, and generates methyl nitrite gas during the oximation phase.
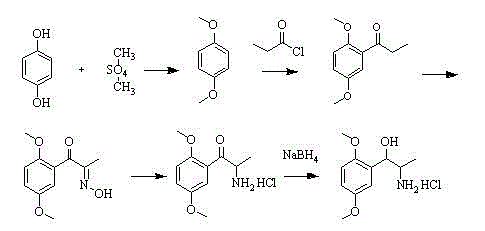
The management of methyl nitrite and hydrogen chloride gas evolution presents severe engineering challenges, requiring specialized corrosion-resistant equipment and extensive gas scrubbing systems to meet environmental regulations. Furthermore, the gas-liquid reaction kinetics in the oximation step are notoriously difficult to control, often leading to fluctuating yields and inconsistent impurity profiles that complicate downstream purification. The reliance on these hazardous reagents not only inflates operational costs due to safety compliance measures but also introduces significant supply chain vulnerabilities associated with the procurement of controlled toxic substances.
The Novel Approach
In stark contrast, the methodology disclosed in CN102976961A employs a sophisticated bromination-amination strategy that fundamentally alters the reaction mechanism to enhance safety and reproducibility. The new route begins with the alpha-bromination of a dimethoxy-substituted propiophenone derivative, followed by a nucleophilic substitution with a protected amine source, and concludes with a mild reduction.
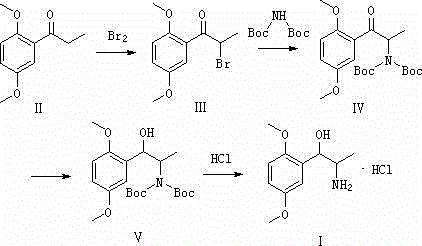
This approach effectively decouples the carbon-nitrogen bond formation from hazardous gas-generating reactions, replacing them with liquid-phase transformations that are far easier to monitor and control. By utilizing di-tert-butyl iminodicarboxylate, the process ensures that the amine group is introduced in a protected form, preventing unwanted side reactions and simplifying the isolation of intermediates. The elimination of high-risk reagents like methyl nitrite creates a inherently safer manufacturing environment, reducing the regulatory burden and facilitating smoother technology transfer from laboratory to commercial scale.
Mechanistic Insights into Alpha-Bromination and Protected Amination
The success of this synthetic route hinges on the precise execution of the alpha-bromination step, where molecular bromine reacts with the enolizable ketone to form the alpha-bromo ketone intermediate (Compound III). This electrophilic substitution is conducted at low temperatures, typically between 0°C and 5°C, to suppress poly-bromination and ensure high regioselectivity for the mono-bromo species. The use of ester solvents such as butyl acetate provides an optimal medium that balances solubility with reaction stability, allowing for the crystallization of the intermediate upon workup, which serves as a critical purification point to remove unreacted starting material.
Subsequently, the introduction of the nitrogen atom is achieved through a nucleophilic displacement of the bromide by the nitrogen anion generated from di-tert-butyl iminodicarboxylate under basic conditions. This step is pivotal as it installs the amine functionality with the Boc protecting groups already in place, shielding the nitrogen from oxidation or other degradation pathways during the subsequent reduction. The final reduction of the ketone to the secondary alcohol using sodium borohydride is chemoselective, leaving the ether linkages and the Boc groups intact, thereby preserving the structural integrity of the molecule until the final deprotection and salt formation with hydrochloric acid yields the pure Methoxamine Hydrochloride.
How to Synthesize Methoxamine Hydrochloride Efficiently
Implementing this synthesis requires careful attention to stoichiometry and temperature control, particularly during the exothermic bromination and the base-mediated amination steps. The process is designed to be operationally simple, utilizing standard reactor setups without the need for high-pressure hydrogenation equipment often seen in alternative reduction methods. For a comprehensive breakdown of the specific reaction parameters, solvent choices, and workup procedures validated in the patent examples, please refer to the standardized protocol below.
- Perform alpha-bromination of 2,5-dimethoxypropiophenone using bromine in an ester solvent at low temperatures (0-5°C) to form the alpha-bromo ketone intermediate.
- Conduct nucleophilic substitution using di-tert-butyl iminodicarboxylate under basic conditions to introduce the protected amine functionality.
- Reduce the ketone group to a secondary alcohol using sodium borohydride, followed by acid-mediated deprotection and salt formation to yield the final hydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this bromination-based route offers substantial strategic advantages for procurement managers and supply chain leaders focused on cost optimization and risk mitigation. By removing the dependency on methyl nitrite and dimethyl sulfate, manufacturers can significantly reduce the capital expenditure associated with specialized safety infrastructure and hazardous waste treatment facilities. The simplified operational profile translates directly into lower overhead costs and a more resilient supply chain that is less susceptible to regulatory disruptions regarding toxic chemical transport and storage.
- Cost Reduction in Manufacturing: The elimination of toxic gas handling systems and the associated scrubbing technologies results in a drastic simplification of the production facility requirements. Furthermore, the ability to crystallize intermediates improves overall yield consistency, reducing the loss of valuable raw materials and minimizing the cost per kilogram of the final API intermediate. The use of commodity chemicals like sodium borohydride and common solvents further drives down the variable costs compared to specialized catalytic hydrogenation or exotic reagents.
- Enhanced Supply Chain Reliability: Sourcing di-tert-butyl iminodicarboxylate and bromine is generally more stable and predictable than managing the supply of highly regulated toxic gases like methyl nitrite. This shift reduces the administrative burden of compliance reporting and permits, ensuring uninterrupted production schedules. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, enhancing the reliability of supply for downstream API manufacturers.
- Scalability and Environmental Compliance: The mild reaction temperatures and ambient pressure operations make this route exceptionally scalable from pilot plant to multi-ton commercial production without significant re-engineering. The waste stream is significantly cleaner, devoid of nitrite salts and sulfur-containing byproducts, which simplifies effluent treatment and aligns with increasingly stringent global environmental standards. This 'green chemistry' aspect not only lowers disposal costs but also enhances the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this novel synthetic pathway. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on the feasibility and benefits of the technology.
Q: How does the new bromination-amination route improve safety compared to traditional oximation?
A: The novel route eliminates the use of methyl nitrite and hydrogen chloride gas required for oximation, significantly reducing the risk of toxic gas leaks and removing the need for complex gas scrubbing infrastructure.
Q: What are the critical quality attributes controlled in this synthesis?
A: The process utilizes crystallization steps after bromination and final salt formation to strictly control impurities, ensuring the removal of unreacted starting materials and side-products like dibromo-derivatives.
Q: Is this method suitable for large-scale commercial manufacturing?
A: Yes, the reaction conditions are mild (0-40°C), use common solvents like ethyl acetate and ethanol, and avoid hazardous high-pressure hydrogenation, making it highly scalable for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Methoxamine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes like the one described in CN102976961A to maintain competitiveness in the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale innovation to industrial reality is seamless and efficient. We are committed to delivering Methoxamine Hydrochloride and related cardiovascular intermediates with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical techniques to verify every batch.
We invite forward-thinking pharmaceutical partners to collaborate with us on optimizing their supply chains through superior chemistry. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and detailed route feasibility assessments to demonstrate how our manufacturing capabilities can drive value and security for your API production needs.
