Advanced 5-Step Synthesis of Methoxamine Hydrochloride: Enhancing Purity and Scalability for Global Pharma Supply Chains
Advanced 5-Step Synthesis of Methoxamine Hydrochloride: Enhancing Purity and Scalability for Global Pharma Supply Chains
The pharmaceutical industry continuously seeks robust synthetic routes that balance high purity with operational safety, particularly for cardiovascular agents like Methoxamine Hydrochloride. Patent CN101417956B introduces a transformative 5-step synthesis method that fundamentally alters the traditional approach to constructing this critical vasoconstrictor intermediate. By replacing hazardous methylating agents with safer alternatives and optimizing reaction conditions across acylation, oximation, and reduction stages, this technology offers a compelling value proposition for reliable pharmaceutical intermediate suppliers. The process initiates with a green methylation of hydroquinone, achieving an impressive 94% yield, and proceeds through rigorous purification steps to ensure the final API meets stringent quality standards. This technical breakthrough not only addresses historical toxicity concerns but also streamlines the manufacturing workflow, providing a stable foundation for commercial scale-up of complex pharmaceutical intermediates in a regulated global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of methoxamine derivatives has relied heavily on dimethyl sulfate as the primary methylating agent, a chemical notorious for its extreme toxicity and carcinogenic properties. The use of such hazardous reagents imposes severe restrictions on manufacturing facilities, requiring specialized containment equipment, extensive personal protective gear, and complex waste treatment protocols that drastically inflate operational expenditures. Furthermore, traditional routes often suffer from inconsistent yields and difficult purification profiles due to the formation of stubborn by-products that co-elute with the desired intermediate. These legacy methods frequently necessitate harsh reaction conditions that can degrade sensitive functional groups, leading to lower overall throughput and increased batch-to-batch variability. For procurement teams, these factors translate into higher costs and significant supply chain risks, as any safety incident or regulatory non-compliance can halt production entirely, disrupting the availability of high-purity pharmaceutical intermediates essential for downstream drug formulation.
The Novel Approach
In stark contrast, the methodology outlined in CN101417956B pioneers the use of methyl p-toluenesulfonate as a benign yet highly effective methylating reagent, effectively eliminating the acute toxicity risks associated with dimethyl sulfate. This strategic substitution allows the reaction to proceed under much milder conditions, specifically controlling temperatures between 15°C and 83°C, which enhances process stability and reduces the energy load required for heating and cooling cycles. The novel route integrates a seamless sequence of Friedel-Crafts acylation, oximation, and catalytic hydrogenation, each step optimized to maximize atom economy and minimize waste generation. By employing common solvents like acetonitrile and ethanol, the process simplifies solvent recovery and recycling, contributing to a more sustainable manufacturing footprint. This approach not only safeguards worker health but also delivers consistent high yields, with the initial methylation step alone achieving 94% efficiency, thereby establishing a new benchmark for cost reduction in API manufacturing through improved safety and efficiency.
Mechanistic Insights into Green Methylation and Sequential Functionalization
The core innovation of this synthesis lies in the initial nucleophilic substitution where hydroquinone reacts with methyl p-toluenesulfonate in the presence of potassium carbonate within an acetonitrile medium. This green methylation step is meticulously controlled, starting at a low temperature of 15°C±3°C to manage the exotherm before ramping to a vigorous reflux at 83°C to drive the reaction to completion over 22 hours. The mechanism avoids the formation of toxic methylating by-products, instead generating p-toluenesulfonic acid salts which are easily removed during the aqueous workup, ensuring a clean reaction profile. Following this, the resulting p-dimethoxybenzene undergoes Friedel-Crafts acylation with propionyl chloride and aluminum trichloride at sub-zero temperatures (-10°C to 0°C), a critical parameter that prevents poly-acylation and ensures regioselectivity. The subsequent oximation utilizes methyl nitrite generated in situ, allowing for precise control over the introduction of the nitrogen functionality without requiring unstable pre-formed reagents.
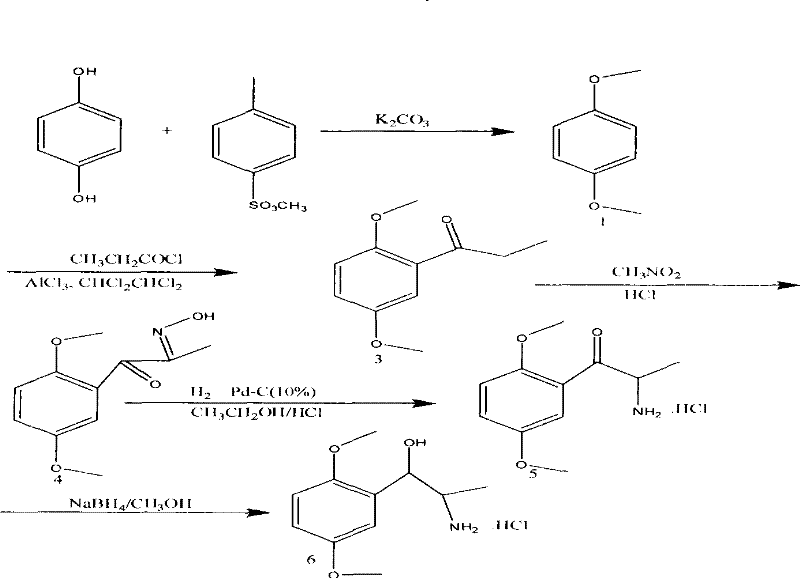
Following the formation of the oxime intermediate, the process employs a sophisticated two-stage reduction strategy to install the final amine and alcohol functionalities with high stereochemical integrity. The oxime group is first reduced via catalytic hydrogenation using 10% Pd-C in ethanol under a hydrogen atmosphere, a step that requires careful monitoring of pressure and temperature (45-55°C) to prevent over-reduction or catalyst poisoning. The final transformation involves the reduction of the ketone moiety using sodium borohydride at strictly controlled low temperatures (0-10°C) to avoid side reactions and ensure the formation of the desired amino-alcohol structure. Throughout these stages, impurity control is maintained through rigorous TLC monitoring and specific washing protocols, such as using NaOH solutions to remove acidic impurities and activated carbon treatments to decolorize the product. This mechanistic precision ensures that the final high-purity pharmaceutical intermediates meet the rigorous specifications required for clinical applications, minimizing the risk of genotoxic impurities often associated with older synthetic routes.
How to Synthesize Methoxamine Hydrochloride Efficiently
The execution of this synthesis requires strict adherence to the patented parameters to replicate the high yields and purity profiles reported. Operators must begin by establishing an inert nitrogen atmosphere to prevent oxidation of the phenolic starting materials, followed by the precise addition of reagents to manage exothermic events. The detailed standard operating procedures involve specific stoichiometric ratios, such as the use of 33.6 grams of potassium carbonate for every 16.7 grams of hydroquinone, and precise thermal ramps that are critical for success. While the general workflow is robust, the devil is in the details regarding workup procedures, such as the specific pH adjustments and solvent exchanges required to isolate the crystalline product. For a comprehensive understanding of the exact operational parameters, the detailed standardized synthesis steps are provided in the guide below.
- Perform green methylation of hydroquinone with methyl p-toluenesulfonate in acetonitrile at 15°C to 83°C to obtain p-dimethoxybenzene.
- Conduct Friedel-Crafts acylation using propionyl chloride and aluminum trichloride to form p-dimethoxypropiophenone.
- Execute oximation, followed by catalytic hydrogenation of the oxime and final reduction with sodium borohydride to yield the target amine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers profound strategic advantages that extend far beyond simple chemical yield improvements. By eliminating the need for dimethyl sulfate, facilities can drastically reduce their insurance premiums and regulatory compliance costs, as the handling of Schedule 1 toxic substances is no longer required. This shift simplifies the logistics of raw material sourcing, as methyl p-toluenesulfonate is more readily available and easier to transport than highly restricted methylating agents, thereby enhancing supply chain resilience against regulatory shocks. Furthermore, the high efficiency of the initial methylation step means that less raw material is wasted, directly lowering the cost of goods sold (COGS) and improving the overall margin profile of the manufactured intermediate. The stability of the process under mild conditions also reduces equipment wear and tear, extending the lifecycle of reactor vessels and lowering capital expenditure requirements for maintenance and replacement.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents like dimethyl sulfate removes the need for specialized scrubbing systems and costly waste disposal protocols, leading to substantial operational savings. Additionally, the high yields achieved in each of the five steps, particularly the 94% yield in the first step, minimize the volume of starting materials required per kilogram of final product, optimizing raw material utilization. The use of recoverable solvents like acetonitrile and ethanol further contributes to cost efficiency, as these can be distilled and reused in subsequent batches, reducing the recurring expense of solvent procurement. Overall, the streamlined process reduces the number of unit operations required for purification, saving both time and energy resources in the production facility.
- Enhanced Supply Chain Reliability: By relying on commercially abundant and less regulated starting materials, manufacturers can secure a more stable supply of inputs, reducing the risk of production stoppages due to vendor shortages or transportation bans on hazardous chemicals. The robustness of the reaction conditions, which tolerate slight variations without significant yield loss, ensures consistent batch output, allowing supply chain planners to forecast inventory levels with greater accuracy. This reliability is crucial for maintaining continuous production schedules for downstream API manufacturers who depend on timely deliveries of pharmaceutical intermediates to meet their own production targets. Consequently, partners adopting this technology can offer more reliable lead times and stronger service level agreements to their global customer base.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard reactor configurations and avoiding extreme pressures or temperatures that would require custom engineering solutions for scale-up. The environmental profile is significantly improved due to the absence of sulfur-based toxic waste streams, facilitating easier permitting and compliance with increasingly strict global environmental regulations. The ability to recycle catalysts, such as the palladium on carbon used in the hydrogenation step, further reduces the environmental footprint and aligns with green chemistry principles valued by modern pharmaceutical buyers. This alignment not only future-proofs the manufacturing asset against tightening regulations but also enhances the brand reputation of the supplier as a responsible and sustainable partner in the global healthcare value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The responses cover aspects ranging from reagent selection to final product quality assurance.
Q: Why is methyl p-toluenesulfonate preferred over dimethyl sulfate in this synthesis?
A: Methyl p-toluenesulfonate is selected to replace highly toxic dimethyl sulfate, significantly improving operator safety and reducing environmental compliance burdens while maintaining high methylation yields of 94%.
Q: What is the overall purity achievable with this 5-step route?
A: The patented process demonstrates robust impurity control, achieving a final product purity of 98.5% for Methoxamine Hydrochloride through optimized crystallization and washing steps.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the process utilizes mild reaction conditions (e.g., 15°C to 83°C) and common solvents like acetonitrile and ethanol, making it stable and easily scalable for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Methoxamine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering stringent purity specifications for all our intermediates, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. By leveraging technologies like the one described in CN101417956B, we can offer clients a superior balance of cost, quality, and safety, positioning us as a strategic partner for long-term supply agreements. Our dedication to continuous improvement means we are constantly evaluating new patents and processes to enhance our portfolio of cardiovascular and specialty chemical intermediates.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain requirements. Request a Customized Cost-Saving Analysis today to understand the potential economic impact of switching to this greener, more efficient manufacturing process. Our experts are ready to provide specific COA data and route feasibility assessments tailored to your volume needs, ensuring that you have all the information necessary to make informed sourcing decisions. Contact us now to secure a reliable supply of high-quality Methoxamine Hydrochloride and experience the NINGBO INNO PHARMCHEM difference in service and technical excellence.
