Advanced Liquid Phase Cyclization Technology for High-Purity Carbetocin Manufacturing
Advanced Liquid Phase Cyclization Technology for High-Purity Carbetocin Manufacturing
The pharmaceutical industry continuously seeks robust manufacturing pathways for complex polypeptide therapeutics, particularly for oxytocin analogues like Carbetocin. A significant technological breakthrough in this domain is detailed in patent CN114591403A, which discloses a novel preparation method of Carbetocin based on liquid phase cyclization. This innovation addresses critical bottlenecks in traditional synthesis, specifically targeting the reduction of oxidation impurities and the enhancement of overall synthesis yield. By shifting the cyclization step from the solid phase to the liquid phase, the process achieves a remarkable balance between operational simplicity and chemical efficiency. The method utilizes Rink Amide MBHA resin for the initial assembly of the linear peptide chain, followed by a strategic cleavage and a room-temperature condensation cyclization in the presence of reducing agents. This approach not only streamlines the production workflow but also ensures the structural integrity of the sensitive disulfide bridge, which is paramount for the biological activity of the final API. For R&D directors and procurement specialists, understanding this shift is crucial for evaluating supply chain resilience and cost structures in peptide manufacturing.
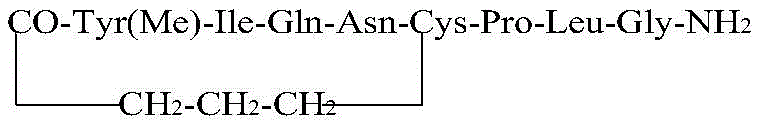
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Carbetocin has been plagued by significant technical hurdles associated with both solid-phase and early liquid-phase methodologies. Conventional solid-phase synthesis often involves performing the cyclization reaction while the peptide is still attached to the resin. This approach frequently results in incomplete cyclization due to steric hindrance and limited mobility of the reactive groups within the resin matrix. Furthermore, the purification of the final product from solid-phase cyclization mixtures is notoriously difficult, often requiring extensive chromatographic separation to remove truncated sequences and deletion mutants. Earlier liquid-phase attempts, such as those utilizing BOP/HOBt coupling reagents, introduced their own set of complications, including complicated operational procedures and the generation of hazardous by-products that are difficult to remove. Additionally, some prior art methods relied on palladium catalysts for deprotection steps, which are highly susceptible to poisoning by free thiol groups exposed during the synthesis, leading to reaction failures and inconsistent batch quality. These inefficiencies translate directly into higher production costs and longer lead times, creating substantial friction for large-scale commercial manufacturing.
The Novel Approach
The methodology outlined in CN114591403A represents a paradigm shift by decoupling the chain assembly from the cyclization event. In this novel approach, the linear peptide is first assembled on the resin using standard Fmoc chemistry, capped with 4-chlorobutyric acid at the N-terminus, and then fully cleaved into the solution phase before cyclization occurs. This liquid-phase cyclization strategy offers superior kinetic control, allowing the reactive thiol and chloroacetyl groups to find each other more freely than they would on a solid support. Crucially, the process incorporates a reducing agent directly into the cyclization mixture, which actively scavenges dissolved oxygen and prevents the oxidation of the cysteine residue to sulfoxides. This dual strategy of liquid-phase reaction dynamics combined with chemical protection against oxidation results in a crude product with significantly higher purity and yield compared to traditional methods. The elimination of complex solid-phase cyclization steps simplifies the workflow, making it inherently more scalable and suitable for industrial-level production of high-purity pharmaceutical intermediates.
Mechanistic Insights into Liquid Phase Condensation Cyclization
The core of this technological advancement lies in the precise control of the cyclization environment. After the linear peptide (4-chlorobutyric acid-Tyr(Me)-Ile-Gln-Asn-Cys-Pro-Leu-Gly-NH2) is cleaved from the resin and precipitated, it is dissolved in an acetonitrile-water solution. The reaction is conducted at room temperature, typically around 25°C ± 2°C, for a duration of 20 to 35 hours. The pH is meticulously adjusted to a range of 8.5 to 9.5, with 9.0 being the optimal point, using alkaline reagents such as DIEA or triethylamine. This specific pH window is critical for deprotonating the thiol group of the cysteine residue to generate the nucleophilic thiolate anion without causing racemization or hydrolysis of other sensitive amide bonds. The intramolecular nucleophilic attack of the thiolate on the terminal chloroacetyl group forms the stable thioether bridge, mimicking the disulfide bond found in natural oxytocin but with enhanced metabolic stability. The use of mild conditions avoids the thermal degradation often seen in harsher cyclization protocols, preserving the stereochemical integrity of the chiral centers within the peptide backbone.
Impurity control is achieved through the strategic addition of reducing agents such as Vitamin C, sodium sulfite, DTT, or TCEP. In the absence of these agents, the free thiol group is highly prone to oxidation by atmospheric oxygen, leading to the formation of sulfoxide impurities (Oxidation Impurity 1 and 2) which are structurally similar to the target molecule and difficult to separate. The patent data demonstrates that maintaining a molar ratio of reducing agent to linear peptide of approximately 1:1 to 2:1 effectively neutralizes oxidative species in the solution. For instance, the use of TCEP or sodium sulfite has been shown to keep the content of oxidized impurities below 2% in the crude mixture. This mechanistic intervention is vital for downstream processing, as it drastically reduces the load on the preparative HPLC purification step, thereby increasing the overall recovery rate of the final API and ensuring that the stringent purity specifications required for injectable polypeptide drugs are met with greater consistency.
How to Synthesize Carbetocin Efficiently
The synthesis of Carbetocin via this liquid phase cyclization route involves a sequence of well-defined unit operations that transition from solid-phase assembly to solution-phase modification. The process begins with the swelling and deprotection of Rink Amide MBHA resin, followed by the sequential coupling of Fmoc-protected amino acids using DIC and HOBt as activators. Once the linear sequence is complete and capped with 4-chlorobutyric acid, the peptide is cleaved using a TFA-based cocktail containing scavengers like thioanisole and EDT. The resulting crude linear peptide is then subjected to the critical cyclization step in an aqueous-organic solvent system under basic conditions with a reducing agent. Finally, the crude cyclic peptide is purified using reverse-phase HPLC and freeze-dried to obtain the final high-purity product. For a detailed breakdown of the specific reagent quantities, reaction times, and workup procedures, please refer to the standardized protocol below.
- Synthesize the linear peptide resin using Fmoc chemistry on Rink Amide MBHA resin, capping with 4-chlorobutyric acid.
- Cleave the linear peptide from the resin using a TFA-based cocktail and precipitate with methyl tert-butyl ether.
- Perform liquid-phase cyclization in acetonitrile/water with a reducing agent (e.g., TCEP or Vitamin C) at pH 9.0 and room temperature.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this liquid phase cyclization technology offers profound advantages for procurement managers and supply chain heads looking to optimize the sourcing of Carbetocin intermediates. The primary benefit stems from the drastic simplification of the manufacturing process, which eliminates the need for specialized solid-phase cyclization equipment and the associated complex washing protocols. By moving the cyclization to the liquid phase, the process becomes more amenable to standard reactor setups found in most fine chemical facilities, thereby reducing capital expenditure requirements for specialized infrastructure. Furthermore, the improved yield of the crude product means that less starting material is required to produce the same amount of final API, directly impacting the cost of goods sold. The robustness of the method against oxidation also translates to fewer failed batches and less waste, contributing to a more sustainable and predictable supply chain.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts, such as the palladium tetratriphenylphosphine used in some prior art methods, removes a significant cost driver from the bill of materials. Palladium catalysts are not only expensive to purchase but also require rigorous and costly removal steps to meet heavy metal limits in pharmaceutical products. By utilizing organic reducing agents like Vitamin C or TCEP, the process avoids these costs entirely. Additionally, the higher crude purity achieved through this method reduces the consumption of chromatography packing materials and solvents during the purification stage, leading to substantial operational savings. The overall efficiency gains allow for a more competitive pricing structure for the final pharmaceutical intermediate without compromising on quality standards.
- Enhanced Supply Chain Reliability: The reliance on readily available and stable reagents enhances the reliability of the supply chain. Unlike methods that depend on sensitive catalysts prone to deactivation or specialized resins with long lead times, the reagents used in this liquid phase cyclization (such as DIC, HOBt, and common reducing agents) are commodity chemicals with robust global supply networks. This reduces the risk of production delays caused by raw material shortages. Moreover, the room-temperature reaction conditions eliminate the need for energy-intensive heating or cooling systems, making the process less vulnerable to utility fluctuations and easier to operate in diverse manufacturing environments. This operational flexibility ensures a consistent flow of high-purity Carbetocin to downstream drug manufacturers.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, moving seamlessly from gram-scale laboratory synthesis to multi-kilogram commercial production. The liquid phase nature of the cyclization step allows for better heat and mass transfer control in larger reactors compared to solid-phase methods, which often suffer from mixing issues at scale. From an environmental standpoint, the reduction in solvent usage and the avoidance of heavy metal waste streams align with increasingly stringent green chemistry regulations. The simplified workup procedure, involving precipitation and centrifugation rather than multiple complex extractions, minimizes the volume of hazardous waste generated. This compliance with environmental standards mitigates regulatory risks and supports the long-term sustainability goals of modern pharmaceutical supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Carbetocin using this advanced liquid phase cyclization method. These answers are derived directly from the experimental data and technical specifications provided in the underlying patent literature. They are intended to provide clarity on the process capabilities, impurity profiles, and scalability potential for stakeholders evaluating this technology for integration into their manufacturing portfolios. Understanding these details is essential for making informed decisions about vendor qualification and process validation.
Q: How does the liquid phase cyclization method control oxidation impurities?
A: By introducing specific reducing agents such as Vitamin C, sodium sulfite, DTT, or TCEP during the cyclization step, the method effectively neutralizes oxygen in the solution, preventing the formation of sulfoxide impurities.
Q: What is the expected purity of Carbetocin produced via this method?
A: The crude peptide typically achieves a purity of over 70% to 85%, and after preparative HPLC purification, the final product reaches approximately 99.9% purity.
Q: Why is this method superior to traditional solid-phase cyclization?
A: Traditional solid-phase cyclization often suffers from incomplete reactions and difficult purification. This liquid-phase approach allows for better monitoring, higher yields (around 60% for crude), and easier removal of by-products.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbetocin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthesis technologies to meet the evolving demands of the global pharmaceutical market. Our technical team has extensively analyzed the liquid phase cyclization pathway for Carbetocin and possesses the expertise to implement this method with precision and efficiency. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of high-quality intermediates. Our facility is equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, including the precise quantification of oxidation impurities and related substances using advanced HPLC techniques. We are committed to delivering Carbetocin that meets the highest international pharmacopoeia standards.
We invite pharmaceutical companies and contract manufacturers to collaborate with us to leverage this cost-effective and robust synthesis route. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data, route feasibility assessments, and samples for your evaluation. Let us help you secure a sustainable and economical supply chain for your Carbetocin-based therapeutic programs.
